Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
5<br />
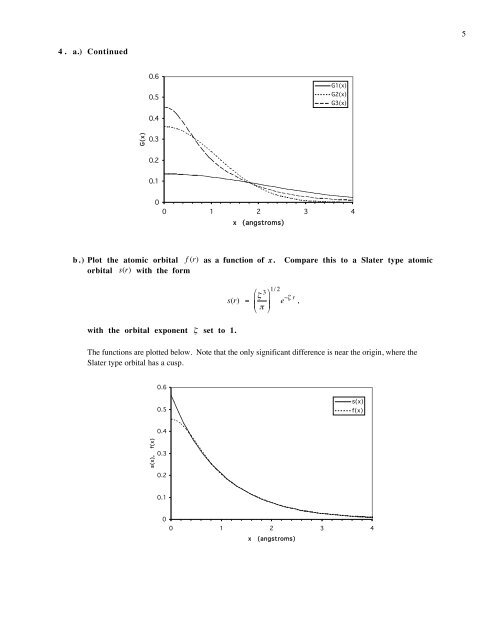
4. a.) Continued<br />
0.6<br />
0.5<br />
G1(x)<br />
G2(x)<br />
G3(x)<br />
0.4<br />
G(x)<br />
0.3<br />
0.2<br />
0.1<br />
0<br />
0 1 2 3 4<br />
x (angstroms)<br />
b.) Plot the atomic orbital f (r) as a function of x.<br />
orbital s(r) with the form<br />
Compare this to a Slater type atomic<br />
€<br />
€<br />
s(r) = ζ 3 1/ 2<br />
⎛⎛ ⎞⎞<br />
⎜⎜<br />
⎜⎜<br />
⎟⎟<br />
⎝⎝ π ⎟⎟ e −ζ r ,<br />
⎠⎠<br />
with the orbital exponent ζ set to 1.<br />
€<br />
The functions are plotted below. Note that the only significant difference is near the origin, where the<br />
Slater type orbital has € a cusp.<br />
0.6<br />
0.5<br />
s(x)<br />
f(x)<br />
0.4<br />
s(x), f(x)<br />
0.3<br />
0.2<br />
0.1<br />
0<br />
0 1 2 3 4<br />
x (angstroms)