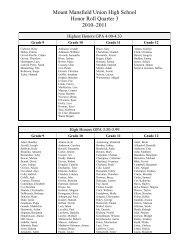
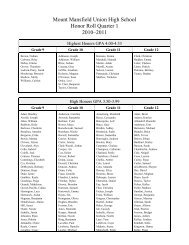
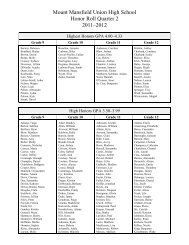
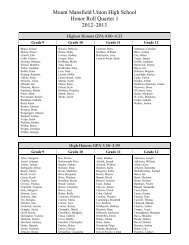
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
11. LiOH(aq) + H3PO4(aq) ----> H2O(l) + Li3PO4(aq)<br />
12. NH4Cl(s) + Mg(OH)2(s) -----> NH3(g) + H2O(l) + MgCl2(s)<br />
13. H2SO4(aq) + Ca(OH)2(s) -----> __________ + __________<br />
14. (NH4) 3PO4(s) + NaOH(s) -----> ________ + ________ + ________<br />
15. CaCO3(s) + HI(aq) -----> ________ + ________ + ________<br />
16. HCN(aq) + Mg(s) -----> ________ + ________<br />
17. Using any combination of the reagents listed here, write balanced equations showing how you could form the<br />
compounds in a - e.<br />
HCl Ca(OH)2 MgO Mg K2CO3 NH4Cl<br />
a. MgCl2<br />
b. H2<br />
c. NH3<br />
d. CaCl2<br />
e. CO2<br />
2