Neuromuscular Blocking Agents in the Adult ICU
Neuromuscular Blocking Agents in the Adult ICU
Neuromuscular Blocking Agents in the Adult ICU
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
INTRODUCTION<br />
<strong>Neuromuscular</strong> block<strong>in</strong>g agents (NMBAs) should be considered an <strong>in</strong>tervention of last resort due to <strong>the</strong><br />
multiple complications associated with <strong>the</strong>ir use. These agents are classified based upon <strong>the</strong>ir structure,<br />
mechanism of action, and pharmacok<strong>in</strong>etic properties. Structurally, NMBAs have ei<strong>the</strong>r an<br />
am<strong>in</strong>osteroidal or benzylisoqu<strong>in</strong>ol<strong>in</strong>ium nucleus. Mechanistically, <strong>the</strong>y are classified as ei<strong>the</strong>r<br />
depolariz<strong>in</strong>g or non-depolariz<strong>in</strong>g. Depolariz<strong>in</strong>g agents b<strong>in</strong>d to and activate nicot<strong>in</strong>ic acetylchol<strong>in</strong>e<br />
receptors result<strong>in</strong>g <strong>in</strong> depolarization of <strong>the</strong> postsynaptic membrane of striated muscle. Succ<strong>in</strong>ylchol<strong>in</strong>e is<br />
<strong>the</strong> only depolariz<strong>in</strong>g agent. Outside of rapid sequence <strong>in</strong>tubation (RSI), it has limited application <strong>in</strong> <strong>the</strong><br />
<strong>ICU</strong> sett<strong>in</strong>g due to its short half-life. Non-depolariz<strong>in</strong>g agents also b<strong>in</strong>d to acetylchol<strong>in</strong>e receptors, but<br />
<strong>in</strong>stead of activat<strong>in</strong>g <strong>the</strong>m <strong>the</strong>y act as competitive antagonists. Pharmacok<strong>in</strong>etically, NMBAs differ <strong>in</strong> <strong>the</strong>ir<br />
duration of action and route of elim<strong>in</strong>ation. Additionally, differences exist <strong>in</strong> <strong>the</strong> degree of histam<strong>in</strong>e<br />
release, vagal block, risk of prolonged blockade, and cost.<br />
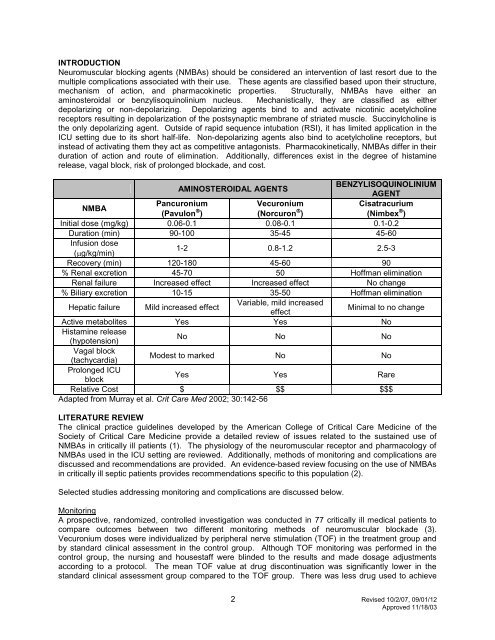
AMINOSTEROIDAL AGENTS<br />
BENZYLISOQUINOLINIUM<br />
AGENT<br />
NMBA<br />
Pancuronium<br />
Vecuronium<br />
Cisatracurium<br />
(Pavulon )<br />
(Norcuron )<br />
(Nimbex )<br />
Initial dose (mg/kg) 0.06-0.1 0.08-0.1 0.1-0.2<br />
Duration (m<strong>in</strong>) 90-100 35-45 45-60<br />
Infusion dose<br />
(g/kg/m<strong>in</strong>)<br />
1-2 0.8-1.2 2.5-3<br />
Recovery (m<strong>in</strong>) 120-180 45-60 90<br />
% Renal excretion 45-70 50 Hoffman elim<strong>in</strong>ation<br />
Renal failure Increased effect Increased effect No change<br />
% Biliary excretion 10-15 35-50 Hoffman elim<strong>in</strong>ation<br />
Hepatic failure Mild <strong>in</strong>creased effect<br />
Variable, mild <strong>in</strong>creased<br />
effect<br />
M<strong>in</strong>imal to no change<br />
Active metabolites Yes Yes No<br />
Histam<strong>in</strong>e release<br />
(hypotension)<br />
No No No<br />
Vagal block<br />
(tachycardia)<br />
Modest to marked No No<br />
Prolonged <strong>ICU</strong><br />
block<br />
Yes Yes Rare<br />
Relative Cost $ $$ $$$<br />
Adapted from Murray et al. Crit Care Med 2002; 30:142-56<br />
LITERATURE REVIEW<br />
The cl<strong>in</strong>ical practice guidel<strong>in</strong>es developed by <strong>the</strong> American College of Critical Care Medic<strong>in</strong>e of <strong>the</strong><br />
Society of Critical Care Medic<strong>in</strong>e provide a detailed review of issues related to <strong>the</strong> susta<strong>in</strong>ed use of<br />
NMBAs <strong>in</strong> critically ill patients (1). The physiology of <strong>the</strong> neuromuscular receptor and pharmacology of<br />
NMBAs used <strong>in</strong> <strong>the</strong> <strong>ICU</strong> sett<strong>in</strong>g are reviewed. Additionally, methods of monitor<strong>in</strong>g and complications are<br />
discussed and recommendations are provided. An evidence-based review focus<strong>in</strong>g on <strong>the</strong> use of NMBAs<br />
<strong>in</strong> critically ill septic patients provides recommendations specific to this population (2).<br />
Selected studies address<strong>in</strong>g monitor<strong>in</strong>g and complications are discussed below.<br />
Monitor<strong>in</strong>g<br />
A prospective, randomized, controlled <strong>in</strong>vestigation was conducted <strong>in</strong> 77 critically ill medical patients to<br />
compare outcomes between two different monitor<strong>in</strong>g methods of neuromuscular blockade (3).<br />
Vecuronium doses were <strong>in</strong>dividualized by peripheral nerve stimulation (TOF) <strong>in</strong> <strong>the</strong> treatment group and<br />
by standard cl<strong>in</strong>ical assessment <strong>in</strong> <strong>the</strong> control group. Although TOF monitor<strong>in</strong>g was performed <strong>in</strong> <strong>the</strong><br />
control group, <strong>the</strong> nurs<strong>in</strong>g and housestaff were bl<strong>in</strong>ded to <strong>the</strong> results and made dosage adjustments<br />
accord<strong>in</strong>g to a protocol. The mean TOF value at drug discont<strong>in</strong>uation was significantly lower <strong>in</strong> <strong>the</strong><br />
standard cl<strong>in</strong>ical assessment group compared to <strong>the</strong> TOF group. There was less drug used to achieve<br />
2 Revised 10/2/07, 09/01/12<br />
Approved 11/18/03