Anodic Behavior of Lithium in Aqueous Electrolytes I. Transient Passivation First lithium-water Lithium manifests a transient passivation when it is anodically polarized batteries E. L. Littauer* and K. C. Tsai* Lockheed Missiles and Space Company, Incorporated, Lockheed Palo Alto Research Laboratories, Palo Alto, California 94304 ABSTRACT to ~ --2.66 NHE in LiOH electrolytes. The duration of the passivation ranges from seconds to hours. The occurrence of the passivation is independent of electrolyte concentration, flow velocity, anode-cathode contact pressure, and of the polarization technique used. The duration of the transient is proportional to electrolyte concentration, the more dilute the solution the shorter the time. The passivation is believed due to the formation of an insulating, but unstable aggregate of LifO which nucleates at active Li sites at the base of the pores in the protective LiOH film. The recovery of the surface to the active state is due to the conversion of the Li20 to LiOH in the presence of water at the Li surface. The recent discovery of ways to electro<strong>chem</strong>ically harness the lithium-water reaction (1) has prompted basic studies of the electro<strong>chem</strong>ical reactions of lithium in aqueous electrolytes. Attempts to minimize polarization effects and to attain high current efficiency have been the subject of an ongoing program. Electro<strong>chem</strong>ical characteristics of the lithium-water cell have been treated in a recent paper (2). The <strong>chem</strong>istry of the lithium-water reaction is relatively simple, but the electro<strong>chem</strong>istry JUNE of the system is complex because (i) the reaction rate between lithium and water is very fast and the 1976 major portion of the lithium electrode is always covered with thick oxide films Li + H20 ~ LiOH + 1/2 tI2 [1] JOURNAL DF THE ELECTROCHEMICAL SOCIETY ELECTROCHEMICAL ---- AND TECHNOLOGY SCIENCE Anodic Behavior of Lithium in Aqueous Electrolytes I. Transient Passivation and (ii) the hydrogen evolution at the metal surface always creates pores within the film and this in turn complicates the electro<strong>chem</strong>ical reaction Li + X- ~--- LiX + e- [2] E. L. Littauer* and K. C. Tsai* where X- = anions in the aqueous electrolyte. These simultaneous processes result in a porous electrode Lockheed Missiles and Space Company, Incorporated, comprising randomly formed and moving pores with Lockheed Palo Alto Research Laboratories, Palo Alto, California active 94304 reaction sites existing only at the extremity of the pores at the metal-film interface. The anodic oxide film is of such a form that it per- ABSTRACT mits the inert cathode st<strong>ru</strong>cture to be pressed against it without shorting. The oxide film acts, in principle, Lithium manifests a transient passivation when it is anodically as a polarized battery separator and serves as a conducting to ~ --2.66 NHE in LiOH electrolytes. The duration of the passivation medium ranges for the mass transport of ionic species to and from seconds to hours. The occurrence of the passivation is independent from the anode of surface. Through control of the anodeelectrolyte concentration, flow velocity, anode-cathode contact cathode pressure, applied and pressure, exceptionally reproducible of the polarization technique used. The duration of the transient is anodic proportional polarization curves can be obtained. to electrolyte concentration, the more dilute the solution the shorter In the the lithium-water time. system, the rate-limiting proc- The passivation is believed due to the formation of an insulating, but unstable ess is the oxidation of lithium rather than the reducaggregate of LifO which nucleates at active Li sites at the base of the pores tion of water at the cathode. It has been found that in the protective LiOH film. The recovery of the surface to the active state is due to the conversion of the Li20 to LiOH in the presence of water * Electro<strong>chem</strong>ical at the Society Active Member. Li surface. Key words: transient passivation, mechanical passivation, lithium, lithiura-water cell, ~H-potential diagrara. under anodic polarization, within certain ranges of hydroxyl ion concentration and "electrolyte flow, two types of passivation are manifested. First, transient passivation is observed at a specific electrode potential. As is described in this paper, it is apparently related to the formation of an intermediate oxide. Second, actual mechanical passivation occurs and is presented in Part II of this subject title. Part III deals with the influence of electrolyte flow velocity on the surface coverage of the lithium anode. Experimental A general setup of the electro<strong>chem</strong>ical cell is shown in Fig, 1. It was designed with the following requirements: rapid assembly, minimum electrical contact resistance between anode holder and the lithium anode, precise control of anode-cathode contact pressures, a Fig. 1. General setup of the electro<strong>chem</strong>ical cell The recent discovery of ways to electro<strong>chem</strong>ically harness the lithium-water reaction (1) has prompted basic studies of the electro<strong>chem</strong>ical reactions of lithium in aqueous electrolytes. Attempts to minimize polarization effects and to attain high current efficiency have been the subject of an ongoing program. Electro<strong>chem</strong>ical characteristics of the lithium-water cell have been treated in a recent paper (2). The <strong>chem</strong>istry of the lithium-water reaction is relatively simple, but the electro<strong>chem</strong>istry of the system is complex because (i) the reaction rate between lithium and water is very fast and the major portion of the lithium electrode is always covered with thick oxide films under anodic polarization, within certain ranges of hydroxyl ion concentration and "electrolyte flow, two types of passivation are manifested. First, transient passivation is observed at a specific electrode potential. As is described in this paper, it is apparently related to the formation of an intermediate oxide. Second, actual mechanical passivation occurs and is presented in Part II of this subject title. Part III deals with the influence of electrolyte flow velocity on the surface coverage of the lithium anode. Experimental A general setup of the electro<strong>chem</strong>ical cell is shown in Fig, 1. It was designed with the following require- 771 2
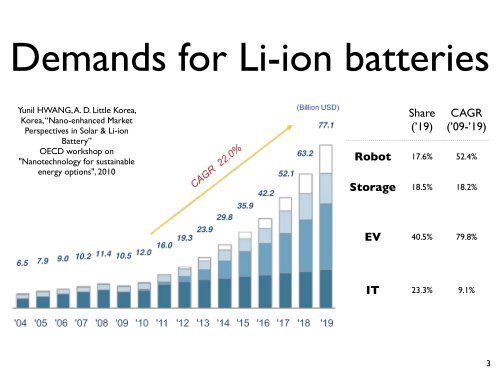
Demands for Li-ion batteries Yunil HWANG, A. D. Little Korea, Korea, “Nano-enhanced Market Perspectives in Solar & Li-ion Battery” OECD workshop on "Nanotechnology for sustainable energy options", 2010 Share (’19) CAGR (’09-’19) Robot 17.6% 52.4% Storage 18.5% 18.2% EV 40.5% 79.8% IT 23.3% 9.1% 3