Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
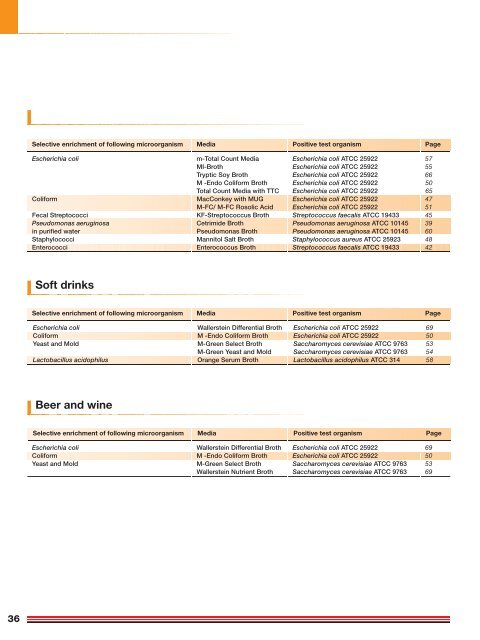
Selective enrichment of following microorganism Media Positive test organism Page<br />
Escherichia coli m-Total Count Media Escherichia coli ATCC 25922<br />
MI-<strong>Broth</strong> Escherichia coli ATCC 25922<br />
Tryptic Soy <strong>Broth</strong> Escherichia coli ATCC 25922<br />
M -Endo Coliform <strong>Broth</strong> Escherichia coli ATCC 25922<br />
Total Count Media with TTC Escherichia coli ATCC 25922<br />
65<br />
Coliform MacConkey with MUG Escherichia coli ATCC 25922 47<br />
M-FC/ M-FC Rosolic Acid Escherichia coli ATCC 25922 51<br />
Fecal Streptococci KF-Streptococcus <strong>Broth</strong> Streptococcus faecalis ATCC 19433 45<br />
Pseudomonas aeruginosa <strong>Cetrimide</strong> <strong>Broth</strong> Pseudomonas aeruginosa ATCC 10145 39<br />
in purified water Pseudomonas <strong>Broth</strong> Pseudomonas aeruginosa ATCC 10145 60<br />
Staphylococci Mannitol Salt <strong>Broth</strong> Staphylococcus aureus ATCC 25923 48<br />
Enterococci Enterococcus <strong>Broth</strong> Streptococcus faecalis ATCC 19433 42<br />
57<br />
55<br />
66<br />
50<br />
Soft drinks<br />
Selective enrichment of following microorganism Media Positive test organism Page<br />
Escherichia coli Wallerstein Differential <strong>Broth</strong> Escherichia coli ATCC 25922 69<br />
Coliform M -Endo Coliform <strong>Broth</strong> Escherichia coli ATCC 25922 50<br />
Yeast and Mold M-Green Select <strong>Broth</strong> Saccharomyces cerevisiae ATCC 9763 53<br />
M-Green Yeast and Mold Saccharomyces cerevisiae ATCC 9763 54<br />
Lactobacillus acidophilus Orange Serum <strong>Broth</strong> Lactobacillus acidophilus ATCC 314 58<br />
Beer and wine<br />
Selective enrichment of following microorganism Media Positive test organism Page<br />
Escherichia coli Wallerstein Differential <strong>Broth</strong> Escherichia coli ATCC 25922 69<br />
Coliform M -Endo Coliform <strong>Broth</strong> Escherichia coli ATCC 25922 50<br />
Yeast and Mold M-Green Select <strong>Broth</strong> Saccharomyces cerevisiae ATCC 9763 53<br />
Wallerstein Nutrient <strong>Broth</strong> Saccharomyces cerevisiae ATCC 9763 69<br />
36
Products<br />
Media<br />
Quick Media Selection Guide<br />
Quick Media Selection Guide<br />
Dairy products<br />
Selective enrichment of following microorganism Media Positive test organism Page<br />
Escherichia coli Wallerstein Differential <strong>Broth</strong> Escherichia coli ATCC 25922 69<br />
Coliform MacConkey with MUG Escherichia coli ATCC 25922 47<br />
Yeast and Mold Potato Dextrose <strong>Broth</strong> Saccharomyces cerevisiae ATCC 9763 59<br />
Lactobacillus MRS <strong>Broth</strong> Lactobacillus plantarum ATCC 8014 56<br />
Food<br />
Selective enrichment of following microorganism Media Positive test organism Page<br />
Escherichia coli Tryptic Soy <strong>Broth</strong> Escherichia coli ATCC 25922 66<br />
MIBlue Escherichia coli ATCC 25922<br />
55<br />
Coliform MacConkey with MUG Escherichia coli ATCC 25922 47<br />
Fecal Streptococci KF-Streptococcus <strong>Broth</strong> Streptococcus faecalis ATCC 19433 45<br />
Yeast and Mold Potato Dextrose <strong>Broth</strong> Saccharomyces cerevisiae ATCC 9763 59<br />
Lactobacillus MRS <strong>Broth</strong> Lactobacillus plantarum ATCC 8014 56<br />
Pharmaceutical and cosmetics<br />
Selective enrichment of following microorganism Media Positive test organism Page<br />
Escherichia coli Tryptic Soy <strong>Broth</strong> Escherichia coli ATCC 25922 66<br />
M-Endo Coliform <strong>Broth</strong> Escherichia coli ATCC 25922 50<br />
Coliform MacConkey with MUG Escherichia coli ATCC 25922 47<br />
Fecal Streptococci KF-Streptococcus <strong>Broth</strong> Streptococcus faecalis ATCC 19433 45<br />
Yeast and Mold Sabouraud Dextrose <strong>Broth</strong> Saccharomyces cerevisiae ATCC 9763 62<br />
Staphylococci Mannitol Salt <strong>Broth</strong> Staphylococcus aureus ATCC 25923 48<br />
Pseudomonas aeruginosa <strong>Cetrimide</strong> <strong>Broth</strong> Pseudomonas aeruginosa ATCC 10145 39<br />
Pseudomonas <strong>Broth</strong> Pseudomonas aeruginosa ATCC 10145 60<br />
37
Media<br />
Brilliant Green Bile <strong>Broth</strong> 2 %<br />
Products<br />
Organisms<br />
Characteristics<br />
E. coli ATCC 25922 Growth / gas<br />
E. aerogenes ATCC 13048 Growth / gas<br />
E. faecalis ATCC 29212 Inhibited<br />
S. aureus ATCC 25923 inhibited<br />
Additional information:<br />
To prevent the growth of accompanying microorganisms in this media an<br />
increased concentration of brilliant green should be added.<br />
Salmonella, for example, are not able to ferment either lactose or<br />
sucrose. For this reason the lactose contained in this medium allows<br />
identification of accompanying, weakly lactose-positive or lactose-negative<br />
organisms.<br />
Order information Brilliant Green Bile Media<br />
Product Description Qty/Pkg Order No<br />
Bottled broth 9 ml vial, Durham tube 20 10 496 710<br />
38
Products<br />
Media<br />
<strong>Cetrimide</strong> <strong>Broth</strong><br />
<strong>Cetrimide</strong> <strong>Broth</strong><br />
Used for the isolation and determination<br />
of Pseudomonas aeruginosa.<br />
<strong>Cetrimide</strong> <strong>Broth</strong> complies with the<br />
recommendations of the United States<br />
Pharmacopeia and also European<br />
Pharmacopeia. The formulation of<br />
this medium corresponds to the<br />
specifications in the DIN Norm 38411.<br />
Description:<br />
Pseudomonas aeruginosa is characterized<br />
by the production of pyocyanin (a<br />
blue green, water soluble, nonflourescent,<br />
phenazine pigment), which is<br />
stimulated by the inclusion of magnesium<br />
chloride and potassium sulfate in<br />
the broth.<br />
<strong>Cetrimide</strong> (N-cetyl-NNN-trimethylammonium<br />
bromide) is added to inhibit<br />
bacteria other than Pseudomonas aeruginosa.<br />
Its action as a quaternary ammonium<br />
cationic detergent causes nitrogen<br />
and phosphorus to be released from<br />
bacterial cells other than Pseudomonas<br />
aeruginosa.<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Pseudomonas aeruginosa ATCC 10145,<br />
incubated 24–48 hours at 35º C.<br />
Negative control:<br />
Escherichia coli ATCC 25922,<br />
24–48 hours at 35º C.<br />
Sterility:<br />
7 days plated sterility test.<br />
<strong>Cetrimide</strong> <strong>Broth</strong>: Pure Culture of Pseudomonas<br />
aeruginosa ATCC 10145.<br />
Formulation:<br />
Per liter of Water adjusted<br />
to pH 7.2 ± 0.2<br />
Peptone<br />
Magnesium chloride<br />
Potassium sulfate<br />
<strong>Cetrimide</strong><br />
Glycerol<br />
20.0 g<br />
1.4 g<br />
10.0 g<br />
0.3 g<br />
10.0 ml<br />
Interpretation:<br />
Pyocyanin blue-green pigmentation<br />
surrounding growth is positive for<br />
Pseudomonas aeruginosa. No color<br />
development is negative for Pseudomonas<br />
aeruginosa.<br />
Organisms<br />
Characteristics Coloring<br />
P. aeruginosa ATCC 10145 Growth Blue/green<br />
E. coli ATCC 25922 inhibited<br />
S. aureus ATCC 25923 inhibited<br />
Historical background:<br />
Harper and Canton followed by Hood described the use of cetrimide (cetyl-trimethylammonium<br />
bromide) for selective isolation of Pseudomonas aeruginosa.<br />
Lawbury used cetrimide in a 0.1 % concentration for clinical application.<br />
Sawbury and Collins later reported a modification of the concentration of<br />
cetrimide required for selectivity. The introduction of ”Cetevlon“ (cetrimide)<br />
stimulated a new study to determine the minimum concentration of cetrimide<br />
required for selective isolation of Pseudomonas aeruginosa for mixed clinical<br />
flora. The new experiments demonstrated that a concentration of 0.03 % using<br />
the much improved Cetavlon was sufficient for selectivity.<br />
Brown and Sawbury introduced the use of a new improved cetrimide agar in<br />
1965. By combining the Medium B of King, Ward and Raney with the 0.03 %<br />
cetrimide concentration previously introduced, they developed a medium that<br />
would support the grow of most desired organisms.<br />
Order information <strong>Cetrimide</strong> <strong>Broth</strong><br />
Product Description Qty/Pkg Order No<br />
Ampouled Media 2 ml 50 10 496 146<br />
Bottled broth 50 ml 8 10 496 856*<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
39
Media<br />
EC <strong>Broth</strong><br />
Products<br />
Organisms<br />
Growth at 44.5º C<br />
E. coli ATCC 25922 Growth / gas<br />
E. aerogenes ATCC 13048 Growth / no gas<br />
E. faecalis ATCC 29212 inhibited<br />
Organisms<br />
Growth at 37º C<br />
E. coli ATCC 25922 Growth / gas<br />
E. aerogenes ATCC 13048 Growth / gas<br />
E. faecalis ATCC 29212 inhibited<br />
Historical background:<br />
EC <strong>Broth</strong> was developed by Hajna and Perry for use in the detection of<br />
coliform bacteria at 37° C and Escherichia coli at 44.5° C. This buffered<br />
lactose broth was designed to improve the methods of detection of<br />
contamination of waters, milk and shellfish. Bile salts were incorporated to<br />
inhibit the growth of gram-positive cocci and sporeformers which<br />
frequently were responsible for false-positive results obtained when using<br />
lactose broth or lauryl tryptose broth. The EC <strong>Broth</strong> formula conforms to<br />
that recommended by the APHA for use in determining the source of water<br />
pollution. Through employment of the elevated temperature confirmatory<br />
test procedure, differentiation can be made between coliforms of fecal<br />
origin (intestine of warm-blooded animals) and coliforms from other<br />
sources<br />
Order information EC Media<br />
Product Description Qty/Pkg Order No<br />
Bottled broth 9 ml, vials, Durham tubes 20 10 496 714<br />
40
Products<br />
Media<br />
EC <strong>Broth</strong> with MUG<br />
EC <strong>Broth</strong> with MUG<br />
Used for the detection of Escherichia<br />
coli in water and food samples by a<br />
fluorogenic procedure.<br />
Interpretation:<br />
The presence of Escherichia coli is<br />
detected by the appearance of<br />
fluorescence throughout the tube.<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Escherichia coli ATCC 25922,<br />
incubated 24 hours at 35–37º C<br />
Negative control:<br />
Enterobacter aerogenes ATCC 13048,<br />
incubated 24 hours at 35–37º C.<br />
Sterility:<br />
7 days plated sterility test.<br />
EC-<strong>Broth</strong>: Vial left: Control; Vial right: <strong>Broth</strong> inoculated<br />
with Escherichia coli ATCC 25922.<br />
Formulation:<br />
Per liter of water adjusted<br />
to pH 6.9 ± 0.2<br />
Description:<br />
The presence of the fluorescence using<br />
a long-wave UV light source confirms<br />
the presence of Escherichia coli, and<br />
any further confirmation is not required.<br />
MUG detects anaerogenic strains,<br />
which may not be detected in the<br />
conventional procedure.<br />
Lactose is a source of energy. Casein<br />
peptone provides additional nutrients.<br />
The mixture of bile salts is inhibiting for<br />
gram-positive bacteria, particularly<br />
bacilli and fecal streptococci.<br />
The substrate 4-methylumbelliferyl-β-<br />
D-glucuronide is hydrolyzed by an<br />
enzyme, β-glucuronidase, possessed<br />
by most Escherichia coli and a few<br />
strains of Salmonella, Shigella and<br />
Yersinia, to produce a fluorescent end<br />
product, 4-methylumbelliferone.<br />
Pancreatic Digest of Casein<br />
Lactose<br />
Bile Salts Mixture<br />
Dipotassium Phosphate<br />
Monopotassium Phosphate<br />
Sodium Chloride<br />
4-Methylumbelliferylβ-D-glucuronide<br />
20.0 g<br />
5.0 g<br />
1.5 g<br />
4.0 g<br />
1.5 g<br />
5.0 g<br />
50 mg<br />
Organisms<br />
Growth at 44,5° C<br />
E. coli ATCC 25922 Growth/gas/ fluorescence<br />
E. aerogenes ATCC 13048 inhibited fluorescence<br />
E. faecalis ATCC 29212 inhibited<br />
Organisms<br />
Growth at 35° C<br />
E. coli ATCC 25922 Growth/gas/ fluorescence<br />
E. aerogenes ATCC 13048 Growth/gas/ no fluorescence<br />
E. faecalis ATCC 29212 inhibited<br />
Order information EC Media with MUG<br />
Product Description Qty/Pkg Order No<br />
Bottled broth 9 ml, vial 20 10 496 709<br />
Bottled broth 9 ml vial, no Durham Tubes 20 10 496 332<br />
41
Media<br />
Enterococcus <strong>Broth</strong><br />
Products<br />
Enterococcus <strong>Broth</strong><br />
Selected media for use in membrane<br />
filtration procedures for the isolation<br />
and enumeration of enterococci in<br />
food, water and other materials.<br />
Description:<br />
Enterococcus broth is a modified version<br />
of the improved media described<br />
by Slanetz and Bartley with TTC. The<br />
membrane filtration method is simple to<br />
perform, does not require confirmation<br />
and permits a direct count of enterococci<br />
in 48 hours.<br />
Interpretation:<br />
Enterococci appear as pink to dark<br />
maroon colonies from 0.5–3 mm in diameter.<br />
Organism<br />
Growth/Coloring<br />
E. faecalis ATCC 19433 Pink to red colonies<br />
E. coli ATCC 25922 Inhibited<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control.<br />
Enterococcus faecalis ATCC 19433,<br />
incubated at 35º C for 24 hours.<br />
Negative control:<br />
Escherichia coli ATCC 25922<br />
incubated at 35º C for 24–48 hours.<br />
Sterility test:<br />
14 days plated sterility test.<br />
Enterococcus <strong>Broth</strong>: A pure culture of Enterococcus<br />
faecalis ATCC 19433 appears pink to red on<br />
this media.<br />
Formulation:<br />
Per liter of water adjusted<br />
to pH 7.2 ± 0.2<br />
Yeast extract<br />
Casein<br />
Dextrose<br />
Papaic digest of soybean meal<br />
Potassium phosphate<br />
Sodium azide<br />
Triphenyltetrazoliumchloride1%<br />
5.0 g<br />
15.0 g<br />
2.0 g<br />
5.0 g<br />
4.0 g<br />
0.4 g<br />
10 ml<br />
Additional informations:<br />
The presence of sodium azide function as inhibitor for the growth of the<br />
entire accompanying Gram-negative microbial flora. As described above<br />
Enterococci reduce TTC and therefore, their colonies appear pink to dark<br />
maroon in color. Furthermore an improved selectivity for enterococci can<br />
be obtained by adding additives like carbonate and Tween 80 ® to the<br />
media (Lachica and Hartman,1968).<br />
Order information Enterococcus Media<br />
Product Description Qty/Pkg Order No<br />
Ampouled Media 2 ml 50 10 496 120<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
42
Products<br />
Media<br />
Eugon <strong>Broth</strong><br />
Eugon <strong>Broth</strong><br />
Used for the cultivation of a wide<br />
variety of microorganisms, including<br />
fastidious bacterial species.<br />
Description:<br />
Eugon media was developed to obtain<br />
eugonic (luxuriant) growth of fastidious<br />
microorganisms. The unenriched media<br />
supports rapid growth of lactobacilli<br />
associated with cured meat products,<br />
dairy products and other food.<br />
The high concentration of Dextrose is<br />
the energy source for rapid growth of<br />
bacteria. L-cystine and sodium sulfite<br />
are added to stimulate growth. Sodium<br />
chloride maintains the osmotic balance<br />
of the media. The high carbohydrate<br />
content along with high sulfur (cystine)<br />
content improves growth with chromogenicity.<br />
Interpretation:<br />
Colony morphology and color are species<br />
specific.<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Escherichia coli ATCC 25922,<br />
24–48 hours at 35–37º C<br />
Candida albicans ATCC 10231,<br />
48 hours at 25–30º C<br />
Negative control:<br />
Not undertaken.<br />
Sterility:<br />
7 days plated sterility test.<br />
Eugon <strong>Broth</strong>: A pure culture of Escherichia coli<br />
ATCC 25922 appears on Eugon <strong>Broth</strong> with typical<br />
white-creamy to opaque colonies.<br />
Formulation:<br />
Per liter of water adjusted<br />
to pH 7.0 ± 0.2<br />
Pancreatic Digest of Casein<br />
Papaic Digest of Soybean Meal<br />
Sodium Chloride<br />
l-Cystine<br />
Sodium Sulfite<br />
Dextrose<br />
15.0 g<br />
5.0 g<br />
4.0 g<br />
0.3 g<br />
0.2 g<br />
5.5 g<br />
Organism<br />
Coloring<br />
E. coli ATCC 25922 White,<br />
cream to<br />
opaque<br />
colonies<br />
Order information Eugon Media<br />
Product Description Qty/Pkg Order No<br />
Ampouled Media 2 ml 50 10 496 126<br />
Bottled broth 50 ml 8 10 496 703*<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
43
Media<br />
HPC <strong>Broth</strong> with TTC<br />
Products<br />
HPC <strong>Broth</strong> with TTC<br />
Used for heterotrophic plate counts<br />
in the examination, for example, of<br />
potable water, dairy products and<br />
swimming pools.<br />
Description:<br />
HPC is used to determine total count at<br />
incubation temperatures of 35º C. All<br />
bacteria develop on HPC with indicator<br />
media and produce a red color as a<br />
result of the precipitation of formazan<br />
following the reduction of 2,3,5-<br />
triphenyltetrazolium chloride (TTC) by<br />
bacteria.<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Escherichia coli ATCC 25922,<br />
incubate for 24–48 hours at 35º C.<br />
Negative control:<br />
Not undertaken.<br />
Sterility:<br />
14 days plated sterility test.<br />
HPC with Indicator: Escherichia coli ATCC 25922<br />
shows a typical red to pink coloring.<br />
Formulation:<br />
Per liter of water adjusted<br />
to pH 7.1 ± 0.2<br />
Interpretation:<br />
Colonies that develop on HPC are<br />
counted as total heterotrophic counts.<br />
Specific colony identification should be<br />
done using standard microbiological<br />
identification techniques.<br />
Peptone<br />
Gelatin<br />
Glycerol<br />
TTC, 1% solution<br />
30.0 g<br />
15.0 g<br />
15.0 ml<br />
10.0 ml<br />
Organisms<br />
Characteristics Coloring<br />
E. coli ATCC 25922 Growth Pink to red<br />
E. faecalis ATCC 29212 Growth Pink to red<br />
S. aureus ATCC 25923 Growth Pink to red<br />
Order information HPC with TTC<br />
Product Description Qty/Pkg Order No<br />
Ampouled Media 2 ml 50 10 496 151<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
44
Products<br />
Media<br />
KF-Streptococcus <strong>Broth</strong><br />
KF-Streptococcus <strong>Broth</strong><br />
Selected media for the isolation and<br />
enumeration of fecal streptococci<br />
Description:<br />
KF-Streptococcus <strong>Broth</strong> is selective for<br />
the determination of fecal streptococci<br />
in polluted surface waters. Maltose and<br />
lactose are fermentable carbohydrates,<br />
sodium azide is the selective agent and<br />
brom cresol purple is the indicator dye.<br />
Interpretation:<br />
Identification of fecal streptococci has<br />
to be undertaken using conventional<br />
microbiology techniques.<br />
Organisms<br />
Characteristics Coloring<br />
E. faecalis ATCC 29212 Growth Red<br />
E. faecalis ATCC 19433 Growth Red<br />
E. aerogenes ATCC 13048 inhibited<br />
E. coli ATCC 25922 inhibited<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Enterococcus faecalis ATCC 19433,<br />
incubated 24–48 hours at 35° C.<br />
Negative control:<br />
Escherichia coli ATCC 25922,<br />
incubated 24– 48 hours at 35° C.<br />
Sterility:<br />
14 days plated sterility test.<br />
KF-Streptococcus <strong>Broth</strong>: Pure culture of Enterococcus<br />
faecalis ATCC 19433 develops on this<br />
media a typical red colony coloring.<br />
Formulation:<br />
Per liter of water adjusted<br />
to pH 7.2 ± 0.2<br />
Peptone<br />
Yeast extract<br />
Sodium chloride<br />
Sodium glycerophosphate<br />
Maltose<br />
Lactose<br />
Sodium azide<br />
Brom cresol purple<br />
TTC 1% solution<br />
10.0 g<br />
10.0 g<br />
5.0 g<br />
10.0 g<br />
20.0 g<br />
1.0 g<br />
0.4 g<br />
15 mg<br />
10.0 ml<br />
Order information KF-Streptococcus Media<br />
Product Description Qty/Pkg Order No<br />
Ampouled Media 2 ml 50 10 496 125<br />
Bottled broth 50 ml 8 10 496 753*<br />
100 ml 1 10 496 754*<br />
500 ml 1 10 496 755*<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
45
Media<br />
Lauryl Sulfate or Lauryl Tryptose <strong>Broth</strong><br />
Products<br />
Lauryl Sulfate or Lauryl Tryptose <strong>Broth</strong><br />
Used for the detection of coliform<br />
organisms in water, wastewater and<br />
foods.<br />
Description:<br />
This media was developed for the<br />
detection of coliform organisms by the<br />
American Public Health Association<br />
(APHA). It is now the standard medium<br />
of choice in the presumptive phase of<br />
the standard coliform MPN test for the<br />
microbiological examination of water.<br />
Interpretation:<br />
Lactose acts as a source of fermentable<br />
carbohydrates for coliforms. The<br />
fermentation of lactose with gas formation<br />
is a presumptive test for coliforms.<br />
Sodium lauryl sulfate inhibits the<br />
growth of organisms other than coliforms.<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Escherichia coli ATCC 29522<br />
incubation for 24–48 hours at 35º C.<br />
Negative control:<br />
Enterococcus faecalis ATCC 19433<br />
incubated at 35º C for 24–48 hours =<br />
no growth.<br />
Sterility test:<br />
7 days plated sterility test.<br />
Lauryl Sulfate <strong>Broth</strong>: Pure culture of Escherichia<br />
coli ATCC 25922 cultivated on this media shows<br />
normal growth with gas formation.<br />
Formulation:<br />
Per liter of water adjusted<br />
to pH 6.8 ± 0.2<br />
Sodium lauryl sulfate<br />
Pancreatic Digest of Casein<br />
Lactose<br />
Dipotassium phosphate<br />
Monopotassium phosphate<br />
Sodium chloride<br />
0.1 g<br />
20.0 g<br />
5.0 g<br />
2.75 g<br />
2.75 g<br />
5.0 g<br />
Organisms<br />
Characteristics<br />
E. coli ATCC 25922 Growth/gas<br />
E. aerogenes ATCC 13048 Growth/gas<br />
S. faecalis ATCC 29212 inhibited<br />
Order information Lauryl Sulfate or Lauryl Tryptose Media<br />
Product Description Qty/Pkg Order No<br />
Lauryl Sulfate or Lauryl Tryptose <strong>Broth</strong> 9 ml vials 20 10 496 722<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
46
Products<br />
Media<br />
MacConkey with MUG<br />
MacConkey with MUG<br />
Used in the presumptive phase for<br />
the presence of coliform bacteria in<br />
water and foods. Selective to the<br />
detection of gram-negative lactose<br />
fermenting bacilli. This medium<br />
largely complies with the European<br />
Pharmacopeia.<br />
Description:<br />
MacConkey <strong>Broth</strong> was originally<br />
developed by MacCONKEY and HILL<br />
(1901) and the formulation for the agar<br />
was modified by MacConkey in 1950.<br />
MacConkey developed this medium for<br />
the cultivation of enteric pathogens and<br />
coliforms. It contains bile salts, which<br />
inhibit certain gram-negative bacteria,<br />
and crystal violet, which inhibits grampositive<br />
organisms such as enterococci<br />
and staphylococci. Combination of the<br />
lactose and neutral red indicator to<br />
produce pink to red colored colonies<br />
indicates an isolate with the ability to<br />
ferment lactose. Non-lactose fermenting<br />
organisms are colorless. The addition<br />
of MUG (4-Methylumbellifryl-β-Dglucuronide),<br />
which is a fluorogenic<br />
enzyme, allows the media to selectively<br />
identify Escherichia coli. MUG is<br />
hydrolyzed by the Escherichia coli<br />
specific enzyme β-glucuronidase to release<br />
4-Methylumbellifone which fluoresces<br />
under ultraviolet light.<br />
Interpretation:<br />
Fluorescence under UV light is specific<br />
for the presence of Escherichia coli.<br />
Lactose fermenting organisms grow as<br />
pink to red colonies.<br />
Organisms Characteristics Coloring<br />
E. faecalis ATCC 29212 inhibited<br />
S. typhimurium ATCC 14028 Growth Clear<br />
E. coli ATCC 25922 Growth/fluorescence<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Escherichia coli ATCC 25922,<br />
incubated at 35º C for 24 hours.<br />
Checked for florescence at 366 nm.<br />
Negative control:<br />
Not undertaken.<br />
Sterility.<br />
7 days plated sterility test.<br />
MacConkey with MUG: Escherichia coli ATCC<br />
25922 forms as lactose-fermenting organisms pink<br />
to red colonies.<br />
Formulation:<br />
Make to 1 liter and adjust pH to 7.1<br />
Casein Peptone<br />
17.0 g<br />
Proteose Peptone<br />
3.0 g<br />
Lactose 10.0g<br />
Bile Salts #3<br />
1.5 g<br />
Sodium Chloride<br />
5.0 g<br />
Neutral Red<br />
30 mg<br />
Crystal Violet<br />
0.1 mg<br />
MUG (4-Methylumbelliferylβ-D-glucuronide)<br />
0.1 g<br />
Additional information:<br />
MacConkey <strong>Broth</strong> is a modification of the original bile salt broth where bromo<br />
cresol purple is used in place of neutral red or litmus as the indicator. Bile salts<br />
replace 0.5 % sodium taurocholate in the origin formulation. The addition of MUG<br />
(4-Methylumbellifryl-(-D-glucuronide), which is a fluorogenic enzyme, allows the<br />
media to selectively identify Escherichia coli. MUG is hydrolyzed by the<br />
Escherichia coli specific enzyme (β-glucuronidase to release 4-Methylumbellifone<br />
which fluoresces under ultraviolet light).<br />
Historical background:<br />
MacConkey <strong>Broth</strong> is a modification of the formula given by MacConkey which<br />
corresponds to the alternative formulation recommended by the World Health<br />
Organization. The medium is used for the presumptive determination of the<br />
presence of coliform organisms (gram-negative, lactose – fermenting bacilli) in<br />
water and milk as well as other materials. Presence of lactose – fermenting<br />
organisms is detected by the change of color of the medium (from purple to<br />
yellow) after inoculation and incubation.The original formula of MacConkey called<br />
for use of litmus as an indicator of acid reaction. In later investigations neutral red<br />
was found to be a more suitable indicator. More recently, Childe and Allen<br />
demonstrated an inhibitory effect on the growth of Escherichia coli by neutral red<br />
in this medium. Bromcresol purple is not only less inhibitory, but also gives a<br />
more clear – cut indication of acid reaction.<br />
Order information MacConkey with MUG<br />
Product Description Qty/Pkg Order No<br />
Ampouled Media 2 ml 50 10 496 118<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
47
Media<br />
Mannitol Salt <strong>Broth</strong><br />
Products<br />
Mannitol Salt <strong>Broth</strong><br />
Used for the selective isolation and<br />
enumeration of staphylococci. In<br />
addition the medium complies with<br />
the recommendations in the United<br />
States Pharmacopeia.<br />
Description:<br />
Because of the amount of peptones<br />
and beef extract, Mannitol Salt is a<br />
nutrient rich medium. Most bacteria<br />
(other than staphylococci) are inhibited<br />
by the high concentration of sodium<br />
chloride. Organisms capable of fermenting<br />
mannitol e.g. Staphylococcus<br />
aureus, cause a pH change in the<br />
media. With phenol red as the pH<br />
indicator the colonies appear with a<br />
yellow coloration.<br />
Interpretation:<br />
Typical pathogenic staphylococci<br />
ferment mannitol and form yellow<br />
colonies with yellow zones, while<br />
typical non-pathogenic staphylococci<br />
do not ferment mannitol and form red<br />
colonies.<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Staphylococcus aureus ATCC 25923,<br />
24–48 hours at 35º C.<br />
Negative control:<br />
Escherichia coli ATCC 25922,<br />
24–48 hours at 35º C.<br />
Sterility:<br />
7 days plated sterility test.<br />
Appearance of Colonies<br />
Surrounded by bright<br />
yellow zones, abundant<br />
growth<br />
pink to red colonies<br />
growth is usually poorer<br />
Microorganisms<br />
Mannitol-positive:<br />
S. aureus<br />
Mannitol-negative:<br />
S. epidermis and others<br />
Mannitol Salt <strong>Broth</strong>: Staphylococcus aureus ATCC<br />
25923 forms typical yellow colonies with zones on<br />
this media. Indication for Mannitol positive<br />
organisms.<br />
Formulation:<br />
Per liter of water adjusted<br />
to pH 7.4 ± 0.2<br />
Beef Extract<br />
Pancreatic Digest of Casein<br />
Peptic Digest of Animal Tissue<br />
Sodium Chloride<br />
d-Mannitol<br />
Phenol Red<br />
1.0 g<br />
5.0 g<br />
5.0 g<br />
75.0 g<br />
10.0 g<br />
25 mg<br />
Organisms<br />
Characteristics Coloring<br />
S. aureus ATCC 25923 Growth Yellow with<br />
yellow zones<br />
S. epidermidis ATCC 12228 Growth Red with<br />
no zone<br />
E. coli ATCC 25922 inhibited<br />
E. aerogenes ATCC 13048 inhibited<br />
Historical background:<br />
The tolerance of Staphylococcus aureus to high concentrations of sodium<br />
chloride was reported by Koch in 1942. In 1945, Chapman described a<br />
formulation which incorporated 7.5 % sodium chloride in phenol red<br />
mannitol agar to successfully cultivate pathogenic staphylococci. These<br />
coagulase positive organisms grew in large colonies surrounded by yellow<br />
zones. Non-pathogenic staphylococci are usually less luxuriant after the 36<br />
hour incubation period recommended by Chapman.<br />
Mannitol Salt <strong>Broth</strong> and Agar is recommended for the enumeration of<br />
staphylococci in food and dairy products by the American Public Health<br />
Association.<br />
Order information Mannitol Salt Media<br />
Product Description Qty/Pkg Order No<br />
Ampouled Media 2 ml 50 10 496 121<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
48
Products<br />
Media<br />
Membrane Lauryl Sulfate <strong>Broth</strong><br />
Membrane Lauryl Sulfate <strong>Broth</strong><br />
For the presumptive identification of<br />
coliforms and Escherichia coli in water<br />
and drinking water. Prepared to the<br />
formula published in Journal of<br />
Hygiene (PHLS/SCA).<br />
Description:<br />
This media was developed for the<br />
detection of coliform organisms and is<br />
now the media of choice for the<br />
enumeration of total coliforms and<br />
Escherichia coli in the United Kingdom.<br />
This media replaced membrane enriched<br />
broth containing 0.4 % Teepol 610.<br />
To use membrane lauryl sulphate broth<br />
for the identification of thermotolerant<br />
coliforms incubate at 30º C for 4 hours<br />
then incubate for further 14 hours at<br />
44º C. Yellow colonies are counted as<br />
presumptive coliforms which require<br />
confirmation.<br />
Organisms<br />
Characteristics Coloring<br />
37 °C<br />
E. coli ATCC 25922 Growth Yellow<br />
E. aerogenes ATCC 13048 Growth Yellow<br />
E. faecalis ATCC 29212 Growth Pink or<br />
colorless<br />
This picture shows a mix culture on Membrane<br />
Lauryl Sulphate <strong>Broth</strong> incubated at 37° C. Organisms<br />
like Escherichia coli ATCC 25922 and Enterobacter<br />
aerogenes ATCC 13048 form yellow<br />
colonies whereas Enterococcus faecalis ATCC<br />
29212 appears as pink colonies.<br />
Interpretation:<br />
Lactose acts as a source of fermentable<br />
carbohydrates for coliforms. Phenol<br />
red acts as an indicator of acidity as a<br />
result of coliform metabolism. Incubate<br />
for 4 hours at 30º C then increase the<br />
temperature to 37º C and incubate for<br />
further 14 hours. Yellow colonies are<br />
counted as presumptive coliforms which<br />
require confirmation. Pink or colorless<br />
colonies are not counted as coliforms.<br />
Organisms<br />
Characteristics Coloring<br />
44 °C<br />
E. coli ATCC 25922 Growth Yellow<br />
E. aerogenes ATCC 13048 Inhibited Inhibited<br />
E. faecalis ATCC 29212 Inhibited Inhibited<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Escherichia coli ATCC 25922<br />
incubation for 4 hours at 30º C<br />
then for 14 hours at 37º C.<br />
Formulation:<br />
Per liter of water adjusted<br />
to pH 7.4 ± 0.2<br />
Peptone<br />
Yeast extract<br />
Lactose<br />
Phenol red (0.4% solution)<br />
Sodium lauryl sulfate<br />
40.0 g<br />
6.0 g<br />
30.0 g<br />
50.0 ml<br />
1.0 g<br />
Negative control:<br />
Not undertaken.<br />
Sterility:<br />
7 days plated sterility test.<br />
Order information Membrane Lauryl Sulfate Media<br />
Product Description Qty/Pkg Order No<br />
Ampouled Media 2 ml 50 10 496 187<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
49
Media<br />
M-Endo Coliform <strong>Broth</strong><br />
Products<br />
M-Endo Coliform <strong>Broth</strong><br />
M-Endo <strong>Broth</strong> is used for the<br />
enumeration of coliforms by membrane<br />
filtration. It is used to differentiate<br />
lactose from non-lactose<br />
fermenting intestinal organisms and<br />
as a presumptive test for coliforms.<br />
Description:<br />
M-Endo is a red colored media, which<br />
needs to be stored in the dark to prevent<br />
discoloration of the media. Gram-positive<br />
bacteria are inhibited on this media by<br />
the desoxycholate and lauryl sulfate. The<br />
addition of ethanol increases the<br />
antibacterial nature of the formulation.<br />
Lactose fermenting organisms form<br />
aldehydes, which react with Schiffs<br />
reagent (basic fuchsin and sodium sulfite)<br />
to give red colored zones around the<br />
colonies. Coliform colonies are therefore<br />
red with a characteristic metallic sheen.<br />
Interpretation:<br />
Production of both acid and aldehyde<br />
by lactose fermenters, such as<br />
Escherichia coli, produce deep red<br />
colonies that color the surrounding<br />
medium and have a green metallic<br />
sheen. Non-lactose fermenters form<br />
colorless, translucent colonies.<br />
Organisms<br />
Characteristics Coloring<br />
E. coli ATCC 25922 Growth Red with<br />
green<br />
metallic<br />
sheen<br />
E. aerogenes ATCC 13048 Growth Red colonies<br />
with or<br />
without<br />
green<br />
metallic<br />
sheen<br />
P. aeruginosa ATCC 10145 Growth Pink to<br />
colorless<br />
S. aureus ATCC 25923 Marked to<br />
complete inhibition<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Escherichia coli ATCC 25922,<br />
24 hours at 35º C.<br />
Negative control:<br />
Pseudomonas aeruginosa ATCC 10145,<br />
24 hours at 35º C.<br />
Sterility:<br />
7 days plated sterility test.<br />
M-Endo <strong>Broth</strong>: Mixed culture of Escherichia coli<br />
ATCC 25922: (red colonies with green metallic<br />
sheen) and Pseudomonas aeruginosa ATCC 10145<br />
(pink to colorless).<br />
Formulation:<br />
Per liter of water adjusted<br />
to pH 7.2 ± 0.2<br />
Peptones<br />
10.0 g<br />
(equal parts digests of animal tissue<br />
and casein)<br />
Peptic Digest of Animal Tissue 5.0 g<br />
Pancreatic Digest of Casein 5.0 g<br />
Yeast extract<br />
1.5 g<br />
Lactose<br />
12.5 g<br />
Sodium chloride<br />
5.0 g<br />
Dipotassium phosphate 4.375 g<br />
Monopotassium phosphate 1.375 g<br />
Sodium lauryl sulfate<br />
50 mg<br />
Sodium desoxycholate<br />
0.1 g<br />
Sodium sulfite<br />
2.1 g<br />
Basic fuchsin<br />
1.05 g<br />
95 % Ethanol 30.0 ml<br />
Order information M-Endo<br />
Product Description Qty/Pkg Order No<br />
Ampouled Media 2 ml 50 10 496 103<br />
Bottled broth 50 ml, Screw cap 8 10 496 700<br />
50 ml, Septa cap 8 10 496 701<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
50
Products<br />
Media<br />
M-FC <strong>Broth</strong><br />
M-FC <strong>Broth</strong><br />
M-FC media (membrane Fecal<br />
Coliform media) is used for the detection<br />
of fecal coliforms as an index<br />
of water pollution.<br />
Description:<br />
Allows the development of fecal<br />
coliforms at elevated temperatures<br />
(44.5° C).<br />
Interpretation:<br />
Bile salts included in the medium inhibit<br />
the growth of gram-positive bacteria.<br />
Fecal coliforms ferment lactose at<br />
elevated temperatures and produce<br />
blue colonies. Other organisms form<br />
grey to cream colonies.<br />
Organisms<br />
Characteristics Coloring<br />
E. coli ATCC 25922 Growth Blue<br />
E. aerogenes ATCC 13048 Growth Gray to<br />
cream<br />
E. faecalis ATCC 29212 inhibited<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Escherichia coli ATCC 25922,<br />
incubated 24 hours at 44.5º C.<br />
Negative control:<br />
Enterobacter aerogenes ATCC 13048,<br />
incubated 24 hours at 44.5º C.<br />
Sterility:<br />
7 days plated sterility test.<br />
M-FC-Media: A pure culture of Escherichia coli<br />
ATCC 25922 shows a typical blue coloring on<br />
m-FC <strong>Broth</strong>.<br />
Formulation:<br />
Per liter of water adjusted<br />
to pH 7.4 ± 0.2<br />
Tryptose<br />
Peptone No. 3<br />
Yeast extract<br />
Sodium chloride<br />
Lactose<br />
Bile salts<br />
Aniline blue<br />
10.0 g<br />
3 5.0 g<br />
3.0 g<br />
5.0 g<br />
12.5 g<br />
1.5 g<br />
0.1 g<br />
Order information M-FC-Media<br />
M-FC-Media: A cultivated mix culture indicates<br />
lactose fermenters as blue colonies whereas nonlactose<br />
fermenters form grey to creamy colonies<br />
e.g. Enterobacter aerogenes ATCC 13048.<br />
Product Description Qty/Pkg Order No<br />
Ampouled Media 2 ml 50 10 496 124<br />
Bottled broth 50 ml 8 10 496 756*<br />
100 ml 1 10 496 757*<br />
500 ml 1 10 496 758*<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
51
Media<br />
M-FC with Rosolic Acid<br />
Products<br />
M-FC with Rosolic Acid<br />
M-FC <strong>Broth</strong> with Rosolic Acid is used<br />
for the detection of fecal coliforms by<br />
the membrane filtration technique.<br />
Description:<br />
M-FC with Rosolic Acid acts and<br />
functions in the same way as m-FC<br />
<strong>Broth</strong>. Rosolic acid inhibits bacterial<br />
growth in general, except for fecal<br />
coliforms.<br />
Interpretation:<br />
Bile salts inhibit non-enteric bacteria.<br />
Aniline blue indicates the ability of fecal<br />
coliforms to ferment lactose to acid<br />
that causes a pH change in the<br />
medium. Other organisms form grey to<br />
cream colonies.<br />
Organisms<br />
Characteristics Coloring<br />
E. coli ATCC 25922 Growth Blue<br />
E. aerogenes ATCC 13048 Growth Gray to<br />
cream<br />
E. faecalis ATCC 29212 inhibited<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Escherichia coli ATCC 25922,<br />
24 hours at 44,5º C.<br />
Negative control:<br />
Enterobacter aerogenes ATCC 13048,<br />
24 hours at 44.5º C.<br />
Sterility:<br />
7 days plated sterility test.<br />
M-FC with Rosolic Acid: Escherichia coli ATCC<br />
25922 forms blue colonies whereas non-lactose<br />
fermenters appears as grey colonies.<br />
Formulation:<br />
Per liter of water adjusted to pH 7.4<br />
Tryptose<br />
Peptone No. 3<br />
Yeast extract<br />
Sodium chloride<br />
Lactose<br />
Bile salts<br />
Aniline blue<br />
Rosolic Acid 1%<br />
10.0 g<br />
5.0 g<br />
3.0 g<br />
5.0 g<br />
12.5 g<br />
1.5 g<br />
0.1 g<br />
10.0 ml<br />
Additional information:<br />
Fecal coliform (FC) Medium for the membrane filtration technique was<br />
described by Geldereich et al. in 1965. It was the first membrane filtration<br />
technique to be incubated at 44.5 ± 0.2.<br />
Order information m-FC with Rosolic Acid<br />
Product Description Qty/Pkg Order No<br />
Ampouled Media 2 ml 50 10 496 114<br />
Bottled broth 50 ml 8 10 496 719*<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
52
Products<br />
Media<br />
M-Green Select <strong>Broth</strong><br />
M-Green Select <strong>Broth</strong><br />
Used for the enumeration of yeasts<br />
and mold in soft drinks and fruit<br />
juices.<br />
Description:<br />
M-Green Select <strong>Broth</strong> is an improved<br />
modification of the liquid medium, m-<br />
Green Yeast and Mold <strong>Broth</strong> and was<br />
developed to improve efficiency of<br />
detection and enumeration of fungi in<br />
sugar based drinks using the membrane<br />
filtration method.<br />
This medium has a low pH, which<br />
inhibits bacterial growth. The addition<br />
of Chloramphenicol further inhibits the<br />
growth of bacteria to allow for the development<br />
and enumeration of yeast<br />
and mold.<br />
The addition of bromocresol green,<br />
which diffuses into fungal colonies as<br />
an alkaline reaction, allows them to be<br />
easily identified. Metabolic by-products<br />
from the developing colonies diffuse<br />
into the surrounding medium, further<br />
reducing the pH which aids in the<br />
inhibition of bacterial growth, but also<br />
produces an acid reaction which<br />
causes residual bromocresol green to<br />
change to yellow.<br />
Interpretation:<br />
Green opaque colonies against a<br />
yellow background indicative of the<br />
growth of yeast. Mold colonies are<br />
green and filamentous.<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Candida albicans ATCC 10231,<br />
incubated 48 hours at 25–30º C.<br />
Negative control:<br />
Escherichia coli ATCC 25922,<br />
incubated 48 hours at 35º C.<br />
Sterility:<br />
7 days plated sterility test.<br />
M-Green Select Media: Ideal for the enumeration<br />
of yeasts & mold.<br />
Formulation:<br />
Per liter of water adjusted<br />
to pH 4.6 ± 0.2<br />
Dipeptone<br />
Yeast extract<br />
Dextrose<br />
Magnesium sulfate<br />
Potassium phosphate<br />
Diastase<br />
Thiamine<br />
Bromocresol green<br />
Chloramphenicol, 1% Solution<br />
10.0 g<br />
9.0 g<br />
50.0 g<br />
2.1 g<br />
2.0 g<br />
50 mg<br />
50 mg<br />
26 mg<br />
8.5 ml<br />
Organisms<br />
Characteristics<br />
E. coli ATCC 25922 Partial to marked inhibition<br />
S. cerevisiae ATCC 4098 Growth<br />
C. albicans ATCC 10231 Growth<br />
Order information M-Green Select Media<br />
Product Description Qty/Pkg Order No<br />
Ampouled Media 2 ml 50 10 496 116<br />
Bottled broth 50 ml 8 10 496 716*<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
53
Media<br />
M-Green Yeast and Mold<br />
Products<br />
M-Green Yeast and Mold<br />
Used for the enumeration of yeast<br />
and mold in soft drinks and fruit<br />
juices.<br />
Description:<br />
M-Green is an improved modification of<br />
the liquid medium, m-Yeast and Mold<br />
<strong>Broth</strong> and was developed to improve<br />
efficiency of detection and enumeration<br />
of fungi in sugar based drinks using the<br />
membrane filtration method.<br />
This medium has a low pH, which<br />
inhibits bacterial growth. The addition<br />
of bromocresol green, which diffuses<br />
into fungal colonies as an alkaline<br />
reaction, allows them to be easily<br />
identified. Metabolic by-products from<br />
the developing colonies diffuse into the<br />
surrounding medium, further reducing<br />
the pH which aids in the inhibition of<br />
bacterial growth, but also produces an<br />
acid reaction which causes residual<br />
bromocresol green to change to yellow.<br />
Interpretation:<br />
Green opaque colonies against a<br />
yellow background are indicative of the<br />
growth of yeast. Mold colonies are<br />
green and filamentous. Examples of<br />
detected organisms:<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Candida albicans ATCC 10231,<br />
48 hours at 25–30º C.<br />
Negative control:<br />
Not undertaken.<br />
Sterility:<br />
7 days plated sterility test.<br />
M-Green Yeast & Mold <strong>Broth</strong>: Typical growth of<br />
Candida albicans ATCC 10231 on a black<br />
membrane.<br />
Formulation:<br />
Per liter of water adjusted<br />
to pH 4.6 ± 0.2<br />
Dipeptone<br />
Yeast extract<br />
Dextrose<br />
Magnesium sulfate<br />
Potassium phosphate<br />
Diastase<br />
Thiamine<br />
Bromocresol green<br />
10.0 g<br />
9.0 g<br />
50.0 g<br />
2.1 g<br />
2.0 g<br />
50 mg<br />
50 mg<br />
26 mg<br />
Order information M-Green Yeast & Mold<br />
Product Description Qty/Pkg Order No<br />
Ampouled Media 2 ml 50 10 496 101<br />
Bottled broth 50 ml 8 10 496 759*<br />
100 ml 1 10 496 760*<br />
500 ml 1 10 496 761*<br />
Bottled agar 100 ml 1 10 496 705<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
54
Products<br />
Media<br />
MI <strong>Broth</strong> and MI Agar<br />
MI <strong>Broth</strong> and MI Agar<br />
Used for the simultaneous detection<br />
of total coliforms and Escherichia<br />
coli in water according to the Surface<br />
Water Treatment Rule (USEPA) and<br />
the Total Coliform Rule (USEPA)<br />
Description:<br />
MI <strong>Broth</strong> detects the presence of<br />
coliform bacteria by the production of<br />
β-galactosidase, which cleaves the<br />
substrate MUGal to produce 4-Methylumbelliferone,<br />
which fluoresces on<br />
exposure to UV light. Non-coliforms do<br />
not produce this enzyme and therefore<br />
do not fluoresce on the medium.<br />
Escherichia coli is detected by the<br />
compound IBDG. The β-glucuronidase<br />
produced by Escherichia coli cleaves<br />
the substrate to produce a blue indigo<br />
color in the colonies. As Escherichia<br />
coli is also a total coliform, and also<br />
produces β-galactosidase it will also<br />
fluoresce.<br />
The antibiotic cefsulodin is added to<br />
inhibit the growth of gram-positive<br />
bacteria and some non-coliform gramnegative<br />
bacteria that can cause false<br />
positive reactions.<br />
MIBlue is specially developed for the<br />
food industry.<br />
Interpretation:<br />
Fluorescent blue colonies are<br />
Escherichia coli.<br />
Colonies that demonstrate blue/white<br />
fluorescence where the colonies are<br />
clear, cream or pale yellow in color are<br />
other coliform organisms. Clear<br />
colonies that do not fluoresce are noncoliforms.<br />
Organisms<br />
Characteristics Coloring<br />
E. coli ATCC 25922 Growth Blue with<br />
fluorescence<br />
E. aerogenes ATCC 13048 Growth Yellow with<br />
fluorescence<br />
P. aeruginosa ATCC 10145 Complete<br />
inhibition<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Escherichia coli ATCC 25922,<br />
18–24 hours at 35º C.<br />
Enterobacter aerogenes ATCC 13048,<br />
18–24 hours at 35º C.<br />
Negative control:<br />
Pseudomonas aeruginosa ATCC 10145,<br />
24 hours at 35º C.<br />
Sterility:<br />
7 days plated sterility test.<br />
MI-Media: Pure Culture of Escherichia coli ATCC<br />
25922 with UV light.<br />
Formulation:<br />
Per liter of Water adjusted<br />
to pH 6.95 ± 0.2<br />
Protease peptone<br />
Yeast extract<br />
β-d-lactose<br />
MUGal<br />
NaCl<br />
K2HPO4<br />
KH2PO4<br />
Sodium lauryl sulfate<br />
Sodium desoxycholate<br />
IBDG<br />
Cefsulodin<br />
(Agar)<br />
5.0 g<br />
3.0 g<br />
1.0 g<br />
0.1 g<br />
7.5 g<br />
3.3 g<br />
1.0 g<br />
0.2 g<br />
0.1 g<br />
0.32 g<br />
5 mg<br />
15.0 g<br />
Order information MI Media<br />
MI-<strong>Broth</strong> Media: Mixed Culture with Escherichia<br />
coli ATCC 25922 and Enterobacter aerogenes<br />
ATCC 13048 without UV.<br />
Product Description Qty/Pkg Order No<br />
Ampouled Media 2 ml 50 10 496 191<br />
Bottled broth 50 ml 1 10 496 851<br />
Bottled agar 50 ml 1 10 496 847<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
MIBlue 10 496 501*<br />
* available on request<br />
55
Media<br />
MRS <strong>Broth</strong><br />
Products<br />
MRS <strong>Broth</strong><br />
Used for the isolation and cultivation<br />
of lactobacilli. MRS <strong>Broth</strong> complies<br />
with the German DIN-Norm 10109<br />
and the International Standard ISO<br />
13721 for the detection of lactosein<br />
meat and to the regulations acc. to §<br />
35 LMBG (06.00/35) for the detection<br />
in meat.<br />
Description:<br />
MRS medium supports luxuriant<br />
growth of all lactobacilli, even the slow<br />
growing species.<br />
Interpretation:<br />
Lactobaccili appear as white colonies.<br />
Pediococcus and Leuconostoc may<br />
also grow on MRS.<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Lactobacillus plantarum ATCC 8014<br />
Lactobacillus fermentum ATCC 9338<br />
incubated at 35º C for 48–72 hours.<br />
Negative control:<br />
Not undertaken.<br />
Sterility:<br />
7 days plated sterility test.<br />
MRS Media; Pure Culture of Lactobacillus plantarum<br />
ATCC 8014.<br />
Formulation:<br />
Per liter of water adjusted<br />
to pH 6.2 ± 0.2<br />
Pancreatic Digest of Casein<br />
Beef extract<br />
Yeast extract<br />
Dextrose<br />
Dipotassium Phosphate<br />
Polysorbate 80<br />
Ammonium citrate<br />
Sodium acetate<br />
Magnesium sulfate<br />
Manganese sulfate<br />
10.0 g<br />
8.0 g<br />
4.0 g<br />
20.0 g<br />
2.0 g<br />
1.0 g<br />
2.0 g<br />
5.0 g<br />
0.2 g<br />
50 mg<br />
Additional information:<br />
The media contains special growth factors for lactobacilli like polysorbate,<br />
acetate, magnesium and manganese. These compounds are known as rich<br />
nutrient base. On a very low degree of selectivity, Pediococcus and Leuconostoc<br />
species and other secondary bacteria can be cultivated.<br />
Historical background:<br />
MRS Agar was developed by de Man, Rogosa & Sharper for the cultivation<br />
of Lactobacillus species. The medium will effectively support the growth of<br />
many strains of lactobacilli that do not generally grow as well as other<br />
media designed for this purpose. Due to the nutritional factors and the<br />
neutral pH other organisms which are not fastidious will grow on the<br />
medium.<br />
Lactobacillus will grow well on the surface of the medium as well as in<br />
deep culture preparations. Although enrichment with carbon dioxide is<br />
unnecessary with this medium, the atmosphere must be kept fairly moist<br />
Order information MRS Media<br />
Product Description Qty/Pkg Order No<br />
Ampouled Media 2 ml 50 10 496 112<br />
Bottled broth 9 ml 20 10 496 737*<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
56
Products<br />
Media<br />
M – TGE Total Count Media<br />
M – TGE Total Count Media<br />
Used for the non-selective development<br />
and enumeration of all aerobic<br />
bacteria.<br />
Description:<br />
All bacteria develop on TGE media and<br />
produce a range of different colored<br />
and sized colonies.<br />
Interpretation:<br />
Identification of bacteria should be<br />
undertaken by using traditional microbiology<br />
techniques following initial<br />
colony development.<br />
For quality control two typical<br />
organisms are detected and enumerated<br />
with m – TGE Total Count Media:<br />
Organisms<br />
Characteristics Coloring<br />
E. coli ATCC 25922 Growth Yellow to<br />
cream<br />
S. aureus ATCC 25923 Growth Yellow to<br />
cream<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Escherichia coli ATCC 25922,<br />
48 hours at 35º C.<br />
Negative control:<br />
Not undertaken.<br />
Sterility:<br />
7 days plated sterility test.<br />
Pure culture of Escherichia coli ATCC 25922 on<br />
m-TGE Total Count Media.<br />
Formulation:<br />
Per liter of water and adjusted<br />
to pH 7.0 ± 0.2<br />
Pancreatic Digest of Casein<br />
Yeast extract<br />
Dextrose<br />
10.0 g<br />
5.0 g<br />
2.0 g<br />
Order information M-TGE Total Count<br />
M-TGE Total Count Media with a mixed culture of<br />
Escherichia coli ATCC 25922 and Staphylococcus<br />
aureus ATCC 25923.<br />
Product Description Qty/Pkg Order No<br />
Ampouled Media 2 ml 50 10 496 102<br />
Bottled broth 50 ml 8 10 496 762*<br />
100 ml 1 10 496 763*<br />
300 ml 1 10 496 764*<br />
500 ml 1 10 496 765*<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
57
Media<br />
Orange Serum Media<br />
Products<br />
Orange Serum Media<br />
Used for the isolation and enumeration<br />
of organisms associated with<br />
the spoilage of citrus juices.<br />
Description:<br />
Organisms known to grow in single<br />
strength and concentrated juices are<br />
lactic acid and acetic acid bacteria and<br />
yeast. Lactobacilli, Leuconostoc and<br />
yeast have all been identified as spoilage<br />
organisms by numerous authors.<br />
Orange serum at pH 5.4 to 5.6 has<br />
been reported to yield maximum<br />
counts of all types of spoilage<br />
organisms in mixed cultures, and in<br />
single culture comparison tests.<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Lactobacillus fermentum ATCC 9338,<br />
48 hours at 35º C.<br />
Candida albicans ATCC 10231,<br />
48 hours at 25–30º C.<br />
Negative control:<br />
Not undertaken.<br />
Sterility:<br />
7 days plated sterility test.<br />
Orange Serum Media agar is especially selective<br />
for microorganisms which prefer low pH conditions<br />
e.g. Lactobacillaceae and some yeast e.g.<br />
Candida.<br />
Formulation:<br />
Per liter of water, adjusted<br />
to pH 5.6 ± 0.2<br />
Interpretation:<br />
The low pH of the test solution usually<br />
prohibits the development of other<br />
microorganisms which cannot survive<br />
low pH conditions. Therefore developing<br />
colonies are presumed to be<br />
problematic organisms.<br />
Orange serum<br />
Yeast extract<br />
Tryptone<br />
Dextrose<br />
Dipotassium phosphate<br />
10.0 g<br />
3.0 g<br />
10.0 g<br />
4.0 g<br />
2.5 g<br />
Order information Orange Serum<br />
Product Description Qty/Pkg Order No<br />
Ampouled Media 2 ml 50 10 496 104<br />
Bottled Agar 100 ml 1 10 496 713<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
58
Products<br />
Media<br />
Potato Dextrose <strong>Broth</strong> and Agar Media<br />
Potato Dextrose <strong>Broth</strong> and Agar Media<br />
Recommended for culturing and<br />
enumerating yeast and mold.<br />
Potato Dextrose <strong>Broth</strong> complies with<br />
the recommendations of the American<br />
Public Health Association for food<br />
and the USP.<br />
Description:<br />
Potato Dextrose <strong>Broth</strong> is recommended<br />
in Standard Methods as the media that<br />
gives the most consistent and highest<br />
counts for the recoveries of yeast and<br />
mold in dairy products. The inclusion of<br />
potato extract encourages the growth<br />
and development of fungi. Sterile<br />
Tartaric Acid may be added to low the<br />
pH to 3.5 ± 0.2 to further inhibit the<br />
growth of conflicting bacteria.<br />
Interpretation:<br />
Yeast, mold and acid tolerant bacteria<br />
grow well on Potato Dextrose <strong>Broth</strong>.<br />
Organisms<br />
Characteristics Coloring<br />
C. albicans ATCC 10231 Growth White,<br />
creamy<br />
S. cerevisiae ATCC 4098 Growth White,<br />
creamy<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Candida albicans ATCC 10231,<br />
incubated at 25–30º C for 48 hours.<br />
Negative control:<br />
Not undertaken.<br />
Sterility:<br />
7 days plated sterility test.<br />
Potato Dextrose Media: Pure culture of Candida<br />
albicans ATCC 10231.<br />
Formulation:<br />
Per liter of water adjusted<br />
to pH 5.1 ± 0.2<br />
Potato infusion<br />
Dextrose<br />
For agar add:<br />
Agar<br />
4.0 g<br />
20.0 g<br />
15.0 g<br />
Additional information:<br />
In general carbohydrate and potato infusion promote the growth of yeast<br />
and mold. Additionally the low pH value has a positive effect by partially<br />
inhibiting the growth of accompanying bacterial flora. If the medium is used<br />
for fungal counts, the pH should be adjusted to approximately 3.5. Fungi<br />
grow on this medium to develop typical morphology.<br />
Historical background:<br />
Potato Dextrose <strong>Broth</strong> is commonly used in slide culture preparations of<br />
fungi to stimulate sporulation and to enhance growth of poorly sporulating<br />
mycelia. It has been used for cultivation and isolation of mold and yeast in<br />
dairy and food products as recommended by the American Public Health<br />
Association and for maintenance of stock cultures of geophilic<br />
dermatophytes.<br />
Order information Potato Dextrose Media<br />
Product Description Qty/Pkg Order No<br />
Ampouled Media 2 ml 50 10 496 138<br />
Bottled broth 50 ml 8 10 496 769*<br />
100 ml 1 10 496 770*<br />
500 ml 1 10 496 771*<br />
Bottled agar 23 ml tube 15 10 496 863*<br />
100 ml 1 10 496 731*<br />
500 ml 1 10 496 767*<br />
1000 ml 1 10 496 768*<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
59
Media<br />
Pseudomonas <strong>Broth</strong><br />
Products<br />
Pseudomonas <strong>Broth</strong><br />
Used for the isolation of Pseudomonas<br />
and differentiating Pseudomonas<br />
aeruginosa from other pseudomonads<br />
based on pigment formation.<br />
Pseudomonas <strong>Broth</strong> complies with<br />
the formulation recommendations of<br />
the United States Pharmacopeia and<br />
to the specifications in the DIN Norm<br />
38411 (examination of water).<br />
Description:<br />
Pseudomonas aeruginosa is characterised<br />
by the production of pyocyanin (a<br />
blue green, water soluble, nonflourescent,<br />
phenazine pigment), which is<br />
stimulated by the inclusion of magnesium<br />
chloride and potassium sulfate in<br />
the broth. Irgasan, an antimicrobial<br />
agent, selectively inhibits gram positive<br />
and gram negative bacteria other than<br />
pseudomonads. Glycerol serves as both<br />
an energy source and helps in the<br />
promotion of pyocyanin.<br />
Interpretation:<br />
Development of a green to blue green<br />
pigmentation surrounding colonies is<br />
positive for Pseudomonas aeruginosa.<br />
Other pseudomonads develop as clear<br />
to amber yellow colonies. Non-pseudomonads<br />
are suppressed.<br />
Organisms<br />
Characteristics Coloring<br />
P. aeroginosa ATCC 10145 Growth Blue to<br />
blue-green<br />
P. aeroginosa ATCC 27853 Growth Blue to<br />
blue-green<br />
E. coli ATCC 25922 inhibited<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Pseudomonas aeruginosa ATCC 10145,<br />
24–48 hours at 35º C.<br />
Negative control:<br />
Escherichia coli ATCC 25922,<br />
24 hours at 35º C.<br />
Sterility:<br />
7 days plated sterility test.<br />
Pseudomonas Media: Typical growth of Pseudomonas<br />
aeroginosa ATCC 10145.<br />
Formulation:<br />
Per liter of Water adjusted<br />
to pH 7.0 ± 0.2<br />
Pancreatic Digest of Casein<br />
Magnesium chloride<br />
Potassium sulfate<br />
Irgasan<br />
Glycerol<br />
20.0 g<br />
1.4 g<br />
10.0 g<br />
0.25 g<br />
20.0 ml<br />
Additional information:<br />
Identification of most Pseudomonas strains can be obtained by their<br />
different pigmentation according to the compounds used in the media.<br />
Some of the strains can only synthesize pyocyanin, some form only<br />
fluorescein and others form both pigments.<br />
Pseudomonas aeruginosa ATCC 27853 is characterised by the production<br />
of pyocyanin (a blue green, water soluble, nonflourescent, phenazine<br />
pigment), which is stimulated by the inclusion of magnesium chloride and<br />
potassium sulfate in the broth. The same Pseudomonas aeruginosa strain<br />
appears on Pseudomonas <strong>Broth</strong> as colonies surrounded by a yellow to<br />
greenish-yellow zone when different types of peptone are added and<br />
magnesium chloride and potassium sulphate are omitted.<br />
BLAZEVIC et al. (1973), noted that some Pseudomonas aeruginosa strains<br />
atypically appear as pyocyanin-negative, fluorescein-positive and for that<br />
reason it is possible to differentiate them from Pseudomonas fluorescens<br />
and Pseudomonas putida. Furthermore BRODSKY and NIXON (1973)<br />
showed that cultivation on MacCONKEY agar can be used for a simple and<br />
rapid differentiation of these strains.<br />
Order information Pseudomonas Media<br />
Product Description Qty/Pkg Order No<br />
Ampouled Media 2 ml 50 10 496 119<br />
Bottled broth 50 ml 8 10 496 775*<br />
100 ml 1 10 496 776*<br />
500 ml 1 10 496 777*<br />
Bottled agar 50 ml 8 10 496 772*<br />
100 m 1 10 496 773*<br />
500 ml 1 10 496 774*<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
60
Products<br />
Media<br />
R2 <strong>Broth</strong> and R2 Agar<br />
R2 <strong>Broth</strong> and R2 Agar<br />
Used for Heterotrophic plate counts<br />
in the examination of potable water.<br />
Description:<br />
R2 broth can be used to determine<br />
heterotrophic plate count at 35ºC.<br />
When incubated at lower temperatures<br />
(25–30º C) for longer periods of 72–96<br />
hours it can also be used to recover<br />
environmentally stressed organisms, or<br />
those that are chlorine tolerant.<br />
Interpretation:<br />
Heterotrophic plate counts are not the<br />
same as standard plate counts and a<br />
media such as TGE should be run in<br />
combination with R2.<br />
Organisms<br />
Characteristics<br />
E. coli ATCC 25922 Growth<br />
E. faecalis ATCC 29212 Growth<br />
S. aureus ATCC 25923 Growth<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Escherichia coli ATCC 25922,<br />
incubation for not less than 72 hours at<br />
35° C.<br />
Negative control:<br />
Not applicable.<br />
Sterility:<br />
7 days plated sterility test.<br />
Sample of tap water on R2 Media.<br />
Formulation:<br />
Per liter of water adjusted<br />
to pH 7.2 ± 0.2<br />
Yeast extract<br />
Peptones<br />
Acid hydrolysate of casein<br />
Dextrose<br />
Soluble starch<br />
Dipotassium phosphate<br />
Magnesium sulfate<br />
(anhydrous)<br />
Sodium pyruvate<br />
0.5 g<br />
0.5 g<br />
0.5 g<br />
0.5 g<br />
0.5 g<br />
0.3 g<br />
24 mg<br />
0.3 g<br />
For agar add:<br />
Agar<br />
15.0 g<br />
Order information R2 Media<br />
Product Description Qty/Pkg Order No<br />
Ampouled Media 2 ml 50 10 496 161<br />
Bottled agar 15 ml tube 15 10 496 724<br />
100 ml 1 10 496 723<br />
500 ml 1 10 496 726*<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
61
Media<br />
Sabouraud Dextrose <strong>Broth</strong><br />
Products<br />
Sabouraud Dextrose <strong>Broth</strong><br />
The medium is used for the quantitative<br />
identification of yeast and<br />
mold.<br />
Description<br />
Peptone in the media is used as a<br />
nitrogen source for the development of<br />
fungi. Dextrose acts as an energy<br />
source for the growth of microorganisms.<br />
The low pH is favorable for<br />
the development of fungi, especially<br />
dermatophytes, but at the same time<br />
inhibits the development of contaminating<br />
bacteria from clinical specimens.<br />
Interpretation:<br />
Growth is interpreted as yeast/mold.<br />
Subculturing and species/group specific<br />
identification is required.<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Candida albicans ATCC 10231<br />
incubated for 72 hours at 25–30º C.<br />
Negative control:<br />
Not undertaken.<br />
Sterility:<br />
7 days plated sterility test.<br />
Sabouraud Dextrose <strong>Broth</strong> is especially selective<br />
for yeast and mold due to the low pH conditions.<br />
Formulation:<br />
Per liter of water adjusted<br />
to pH 5.6 ± 0.2<br />
Peptone<br />
Dextrose<br />
For agar add:<br />
Agar<br />
10.0 g<br />
20.0 g<br />
15.0 g<br />
Organisms<br />
Characteristics Coloring<br />
C. albicans ATCC 10231 Growth Off-white,<br />
creamy<br />
E. coli ATCC 25922 Growth Off-white,<br />
creamy<br />
S. cerevisiae ATCC 9763 Growth Off-white<br />
Historical background:<br />
Sabouraud Dextrose <strong>Broth</strong> is a modification of dextrose agar described by<br />
Sabouraud in 1892 for identification of fungi based on their cultural<br />
characteristics. The medium depended solely upon its acid pH for<br />
suppression of bacteria. Sabouraud Dextrose Agar, Emmons is a<br />
modification with a neutral pH and reduced dextrose concentration.<br />
Historically, the inhibitory action of Sabouraud Dextrose Agar on<br />
microorganisms other than fungi was enhanced by the addition of agents<br />
such as tellurite, copper sulfate, bile salts, dyes and antibiotics.<br />
Sabouraud Dextrose <strong>Broth</strong> (Fluid Sabouraud Medium) is the liquid<br />
counterpart of the agar prepared according to the formulation specified in<br />
the U.S. Pharmacopoeia and National Formulary for sterility testing of<br />
pharmaceutical products. Used in the quantitative identification of yeast<br />
and mold.<br />
Order information Sabouraud Dextrose Media<br />
Product Description Qty/Pkg Order No<br />
Ampouled Media 2 ml 50 10 496 157<br />
Bottled broth 50 ml 8 10 496 702*<br />
100 ml 1 10 496 785*<br />
500 ml 1 10 496 786*<br />
Bottled agar 50 ml 8 10 496 781*<br />
100 ml 1 10 496 782*<br />
500 ml 1 10 496 783*<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
62
Products<br />
Media<br />
Sabouraud 4 % Dextrose <strong>Broth</strong><br />
Sabouraud 4 % Dextrose <strong>Broth</strong><br />
Used in the quantitative identification<br />
of yeast, mold and acidophilic<br />
microorganisms.This culture medium<br />
complies with the recommendations<br />
of the United States Pharmacopeia<br />
and the European Pharmacopeia.<br />
Description:<br />
Peptone in the media is used as a<br />
nitrogen source for the development of<br />
fungi. Dextrose acts as an energy<br />
source for the growth of microorganisms.<br />
The low pH is favorable for the<br />
development of fungi, especially dermatophytes,<br />
but at the same time<br />
inhibits the development of contaminating<br />
bacteria from clinical specimens.<br />
The use of Sabouraud media has been<br />
documented for establishing the microbial<br />
concentration in cosmetics and<br />
for the mycological evaluation of food.<br />
Interpretation:<br />
Growth is interpreted as yeast/mold.<br />
Sub-culturing and species/group<br />
specific identification is required.<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Candida albicans ATCC 10231,<br />
incubated 72 hours at 25–30º C.<br />
Negative control:<br />
Not undertaken.<br />
Sterility:<br />
7 days plated sterility test.<br />
Sabouraud 4 % Dextrose Media: Pure culture of<br />
Candida albicans ATCC 10231.<br />
Formulation:<br />
Per liter of water adjusted<br />
to pH 5.6 ± 0.2<br />
Peptone<br />
Dextrose<br />
10.0 g<br />
40.0 g<br />
Organisms<br />
Characteristics<br />
C. albicans ATCC 10231 Growth<br />
S. cerevisiae ATCC 9763 Growth<br />
Order information Sabouraud 4% Dextrose Media<br />
Product Description Qty/Pkg Order No<br />
Ampouled Media 2 ml 50 10 496 162<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
63
Media<br />
Standard Methods Agar<br />
Products<br />
Standard Methods Agar<br />
Used for obtaining microbiological<br />
plate counts from milk and dairy<br />
products, foods, water and other<br />
materials of sanitary importance.<br />
The formulation of this medium<br />
follows the demands of the Standard<br />
Methods for the Examination of<br />
Water and Wastewater, the American<br />
Public Health Association, the<br />
American Water Works Association<br />
and the Water Pollution Control<br />
Federation in 1992 and the Standard<br />
Methods for the Examination of Dairy<br />
Products of the American Public<br />
Health Association (1985).<br />
Description:<br />
All bacteria develop on Standard<br />
Methods and produce a range of<br />
different colored and sized colonies.<br />
Interpretation:<br />
It is not possible using Standard<br />
Methods Agar to presumptively identify<br />
any bacteria. Identification can only be<br />
undertaken using traditional microbiology<br />
techniques following initial colony<br />
development.<br />
Organisms<br />
Characteristics Coloring<br />
E. coli ATCC 25922 Growth Yellow to<br />
cream<br />
S. aureus ATCC 25923 Growth Yellow to<br />
cream<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Escherichia coli ATCC 25922,<br />
incubated 48 hours at 35º C.<br />
Negative control:<br />
Not undertaken.<br />
Sterility:<br />
7 days plated sterility test.<br />
Historical background:<br />
Standard Methods Agar is a<br />
modification of Bowers and<br />
Hucker Tryptone Glucose Skim<br />
Milk Agar. It was shown by Yale<br />
to be more effective in plate<br />
count procedures on milk and<br />
dairy products.<br />
The American Public Health<br />
Association in their ”Standard<br />
Methods for the Examination of<br />
Dairy Products“ recommends<br />
the use of Standard Methods<br />
Agar for performance of the<br />
”Standard Plate Count“ on<br />
dairy products.<br />
Standard Methods Agar is the<br />
same formulation as Plate<br />
Count Agar recommended by<br />
the APHA for use in the<br />
”Standard Plate Count“ on<br />
water.<br />
Standard Methods Agar with membrane filter and<br />
incubated with a pure culture of Escherichia coli<br />
ATCC 25922.<br />
Formulation:<br />
Per liter of water adjusted<br />
to pH 7.0 ± 0.2<br />
Pancreatic Digest of Casein<br />
Yeast extract<br />
Dextrose<br />
Agar<br />
5.0 g<br />
2.5 g<br />
1.0 g<br />
15.0 g<br />
Same agar as above but incubated with a mix<br />
culture of Escherichia coli ATCC 25922 and<br />
Staphylococcus aureus ATCC 25923. Both form<br />
yellow to creamy colonies.<br />
Order information Standard Methods Medium<br />
Product Description Qty/Pkg Order No<br />
Bottled agar 100 ml 1 10 496 706<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
64
Products<br />
Media<br />
Total Count Media with TTC<br />
Total Count Media with TTC<br />
Total Count Media with Indicator is<br />
used for the non-selective development<br />
and enumeration of all aerobic<br />
bacteria by the membrane filtration<br />
method.<br />
This media complies with the<br />
specifications given by the APHA for<br />
the examination of water (1992) and<br />
for food (1992).<br />
Description:<br />
All bacteria develop on Total Count<br />
Media with indicator and produce a red<br />
color as a result of the precipitation of<br />
formazan following the reduction of<br />
2,3,5-triphenyltetrazolium chloride<br />
(TTC) by bacteria. The table below<br />
shows typical organisms which can be<br />
enumerated with this media:<br />
Interpretation:<br />
It is not possible using Total Count<br />
Media with Indicator to presumptively<br />
identify any bacteria. Identification can<br />
only be undertaken using traditional<br />
microbiology techniques following<br />
initial colony development.<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Escherichia coli ATCC 25922<br />
incubated 24–48 hours at 35° C.<br />
Negative control:<br />
Not undertaken.<br />
Sterility<br />
7 days plated sterility test.<br />
Total Count Media with Indicator: Escherichia coli<br />
ATCC 25922 and Staphylococcus aureus ATCC<br />
25923 can be easily detected according to their<br />
red to pink colonies.<br />
Formulation:<br />
Per liter of water adjusted<br />
to pH 7.0 ± 0.2<br />
Pancreatic Digest of Casein<br />
Yeast extract<br />
Dextrose<br />
TTC, 1% solution<br />
10.0 g<br />
5.0 g<br />
2.0 g<br />
10.0 ml<br />
Organisms<br />
Characteristics Coloring<br />
E. coli ATCC 25922 Growth Pink to red<br />
S. aureus ATCC 25923 Growth Pink to red<br />
P. aeruginosa ATCC 10145 Growth Pink to red<br />
E. faecalis ATCC 29212 Growth Pink to red<br />
Order information Total Count Media with Indicator<br />
Product Description Qty/Pkg Order No<br />
Ampouled Media 2 ml 50 10 496 113<br />
Bottled broth 50 ml 8 10 496 857*<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
65
Media<br />
Trypticase Soy <strong>Broth</strong> (TSB)<br />
Products<br />
Trypticase Soy <strong>Broth</strong> (TSB)<br />
Trypticase Soy <strong>Broth</strong> (TSB) is a<br />
general-purpose medium used for<br />
the cultivation of most bacteria.<br />
Description:<br />
TSB broth is a medium that will support<br />
the growth of a wide variety of microorganisms<br />
including aerobic, facultative<br />
and anaerobic bacteria and fungi.<br />
Interpretation:<br />
When used as a broth adsorbed onto<br />
pads TSB will support the development<br />
of mixed cultures of bacteria and fungi.<br />
When used in sterility test applications<br />
turbidity indicates the presence of<br />
organisms when compared to an<br />
uninoculated control.<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive controls:<br />
Escherichia coli ATCC 25922,<br />
incubation for 24–48 hours at 35º C.<br />
Negative control:<br />
Not undertaken.<br />
Sterility:<br />
7 days plated sterility test.<br />
Ready to use Tryptic Soy <strong>Broth</strong> (USP). Not<br />
inoculated.<br />
Formulation:<br />
Per liter of water adjusted<br />
to pH 7.3 ± 0.2<br />
Pancreatic digest of casein<br />
Papaic digest of soybean meal<br />
Sodium chloride<br />
Dipotassium phosphate<br />
Dextrose<br />
17.0 g<br />
3.0 g<br />
5.0 g<br />
2.5 g<br />
2.5 g<br />
Organisms<br />
Characteristics<br />
B. subtilis ATCC 6633 Growth<br />
C. albicans ATCC 10231 Growth<br />
E. coli ATCC 25922 Growth<br />
S. aureus ATCC 25923 Growth<br />
Order information Trypticase Soy Media (TSB)<br />
Product Description Qty/Pkg Order No<br />
Ampoules 2 ml 50 10 496 160<br />
Bottled media 50 ml 8 10 496 791*<br />
100 ml 1 10 496 792*<br />
100 ml, Septa Cap 1 10 496 750*<br />
100 ml 1 10 496 707*<br />
Bottled agar 23 ml 15 10 496 862*<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
66
Products<br />
Media<br />
Trypticase Soy <strong>Broth</strong> – Single Strength<br />
Trypticase Soy <strong>Broth</strong> – Single Strength<br />
Description:<br />
General-purpose medium used in<br />
qualitative procedures for the cultivation<br />
of fastidious and non-fastidious<br />
microorganisms. Trypticase Soy <strong>Broth</strong> –<br />
Single Strength complies with the demands<br />
of the DIN Norm 10167 for the<br />
detection of Escherichia coli serotype<br />
0157:H7 in foods and FDA-BAM for the<br />
isolation of enterohemorrhagic Escherichia<br />
coli (EHEC). In addition the media<br />
conforms to the formula of the U.S.<br />
Pharmacopoeia.<br />
Interpretation:<br />
When used as a broth adsorbed onto<br />
pads TSB will support the development<br />
of mixed cultures of bacteria and fungi.<br />
When used in sterility test applications<br />
turbidity indicates the presence of<br />
organisms when compared to an uninoculated<br />
control.<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive control:<br />
Escherichia coli ATCC 25922,<br />
incubated 24–48 hours at 35º C.<br />
Negative control:<br />
Not undertaken.<br />
Sterility:<br />
7 days plated sterility test.<br />
Trypticase Soy <strong>Broth</strong>-Single Strength. Not<br />
inoculated.<br />
Formulation:<br />
Per liter of water adjusted<br />
to pH 7.3 ± 0.2<br />
Pancreatic digest of casein<br />
Papaic digest of soybean meal<br />
Sodium chloride<br />
Dipotassium phosphate<br />
Dextrose<br />
17.0 g<br />
3.0 g<br />
5.0 g<br />
2.5 g<br />
2.5 g<br />
Organisms<br />
Characteristics<br />
B. subtilis ATCC 6633 Growth<br />
C. albicans ATCC 10231 Growth<br />
E. coli ATCC 25922 Growth<br />
S. aureus ATCC 25923 Growth<br />
Historical background:<br />
Trypticase Soy <strong>Broth</strong> was originally developed for detecting the effectiveness<br />
of sulfonamides against microorganisms such as pneumococci<br />
without the addition of serum or blood to the medium. Spink and Hamilton<br />
demonstrated the suitability of the medium for cultivation of aerobic and<br />
facultative microorganisms including Brucella. Garrision and Hedgecock<br />
both used the medium to promote the growth of pathogenic fungi. The<br />
addition of agar to enhance the growth of anaerobic organisms is<br />
described by Mashima and Ellison and Fredette, Auger, and Forget.<br />
Sherman and Stark demonstrated that a medium containing 6.5 % NaCl<br />
could be used for differentiation of group D enterococci from other<br />
streptococci.<br />
Order information Trypticase Soy <strong>Broth</strong>-Single Strength<br />
Product Description Qty/Pkg Order No<br />
Bottled broth 10 ml, Vials 20 10 496 742*<br />
100 ml 1 10 496 707<br />
100 ml, wide mouthed 1 10 496 741*<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
67
Media<br />
Trypticase Soy <strong>Broth</strong> – Double Strength<br />
Products<br />
Trypticase Soy <strong>Broth</strong> – Double Strength<br />
General-purpose medium used in<br />
qualitative procedures for the cultivation<br />
of fastidious and non-fastidious<br />
microorganisms. This media is also<br />
recommended for sterility test<br />
according to U.S. Pharmacopeia.<br />
Description:<br />
TSB broth is a medium that will support<br />
the growth of a wide variety of<br />
microorganisms including aerobic,<br />
facultative and anaerobic bacteria and<br />
fungi.<br />
Interpretation:<br />
When used as a broth adsorbed onto<br />
pads TSB will support the development<br />
of mixed cultures of bacteria and fungi.<br />
When used in sterility test applications<br />
turbidity indicates the presence of<br />
organisms when compared to an<br />
uninoculated control.<br />
Quality Control and recommended<br />
incubation conditions:<br />
Positive controls:<br />
Escherichia coli ATCC 25922,<br />
incubated 24–48 hours at 35º C.<br />
Negative control:<br />
Not undertaken.<br />
Sterility:<br />
7 days plated sterility test.<br />
Trypticase Soy <strong>Broth</strong> (Double Strength). Not<br />
inoculated.<br />
Formulation:<br />
Per liter of water adjusted<br />
to pH 7.3 ± 0.2<br />
Pancreatic digest of casein<br />
Papaic digest of soybean meal<br />
Sodium chloride<br />
Dipotassium phosphate<br />
Dextrose<br />
34.0 g<br />
6.0 g<br />
10.0 g<br />
5.0 g<br />
5.0 g<br />
Organisms<br />
Characteristics<br />
B. subtilis ATCC 6633 Growth<br />
C. albicans ATCC 10231 Growth<br />
E. coli ATCC 25922 Growth<br />
S. aureus ATCC 25923 Growth<br />
Trypticase Soy <strong>Broth</strong> (Double Strength): left vial:<br />
control, right vial inoculated with Escherichia coli<br />
ATCC 25922.<br />
Order information Trypticase Soy <strong>Broth</strong> (Double Strength)<br />
Product Description Qty/Pkg Order No<br />
Bottled broth 50 ml, W/M bottle 1 10 496 725<br />
100 ml 1 10 496 708<br />
68
Products<br />
Media<br />
Wallerstein Nutrient <strong>Broth</strong> (WL) and WL Differential <strong>Broth</strong> (WLD)<br />
Wallerstein Nutrient <strong>Broth</strong> (WL) and<br />
WLDifferential <strong>Broth</strong> (WLD)<br />
WL Nutrient broth is used for the<br />
determination of microbiological flora<br />
in brewing and fermentation processes.<br />
WL differential media suppresses<br />
the growth of yeast and mold<br />
and cultivated bacteria encountered<br />
in brewing and industrial fermentation.<br />
The medium has the same<br />
formulation as WL nutrient media but<br />
has 4 mg of Cyclohexamide per liter<br />
added.<br />
Description:<br />
Use of the medium at pH 5.5 and<br />
incubation at 25° C will give reliable<br />
counts for brewer’s yeast. Adjustment<br />
of the pH to 6.5 and incubation at 30° C<br />
allows for the selective growth of<br />
bakers and distillers yeast.<br />
Interpretation WL Nutrient <strong>Broth</strong>:<br />
Media will develop all yeast that<br />
incubate at the selected temperature<br />
and pH range.<br />
WL Nutrient <strong>Broth</strong><br />
Organisms<br />
Characteristics<br />
Escherichia coli ATCC 25922Growth<br />
L. fermentum ATCC 9338 Growth<br />
S. cerevisiae ATCC 9763 Growth<br />
Interpretation WL Differential <strong>Broth</strong>:<br />
When incubated at pH 5.5 estimates of<br />
beer cocci and lactic rods can be made<br />
under anaerobic conditions. Estimates<br />
of acetic acid rods and thermobacteria<br />
are made under aerobic conditions.<br />
WL Differential <strong>Broth</strong><br />
Organisms<br />
Characteristics<br />
Escherichia coli ATCC 25922Growth<br />
L. fermentum ATCC 9338 Growth<br />
S. cerevisiae ATCC 9763 Partial to marked inhibition<br />
Quality Control and recommended<br />
incubation conditions WL Nutrient:<br />
Positive control:<br />
Sacchromyces cerivisiae ATCC 4098,<br />
incubated 48 hours at 25–30° C.<br />
Escherichia coli ATCC 25922,<br />
incubated 24–48 hours at 35° C.<br />
Negative control:<br />
Not applicable.<br />
Sterility:<br />
7 days plated sterility test.<br />
Quality Control and recommended<br />
incubation conditions WL Differential:<br />
Positive control:<br />
Lactobacillus plantarum ATCC 8014,<br />
incubated 48 hours at 35° C.<br />
Negative control:<br />
Sacchromyces cerivisiae ATCC 4098,<br />
incubated 48 hours at 25–30° C.<br />
Sterility:<br />
7 days plated sterility test.<br />
Culture of Sacchromyces cerivisiae ATCC 4098 on<br />
Wallerstein Nutrient <strong>Broth</strong> (WL).<br />
Formulation:<br />
Per liter of water adjusted<br />
to pH 5.5 ± 0.2<br />
Casein<br />
Yeast Extract<br />
Dextrose<br />
Monopotassium phospate<br />
Potassium chloride<br />
Calcium chloride<br />
Magnesium sulfate<br />
Ferric chloride<br />
Manganese sulfate<br />
Bromocresol green<br />
Additional Component<br />
for WL Differential <strong>Broth</strong>:<br />
Cyclohexamide<br />
5.0 g<br />
4.0 g<br />
50.0 g<br />
550 mg<br />
425 mg<br />
125 mg<br />
125 mg<br />
2.5 mg<br />
2.5 mg<br />
22 mg<br />
4 mg<br />
Additional information:<br />
WL Nutrient <strong>Broth</strong> has a pH of 5.5 which is optimal for the enumeration of<br />
brewers’ yeast. In the case of cultivating microorganisms from an alcoholic<br />
mash, tomato juice should be added to the medium. WL Differential Media<br />
contains additionally 4 mg of Cyclohexamide per liter. This component<br />
inhibits the developments of yeast without interfering with the development<br />
of bacteria generally encountered in beers. Cyclohexamide in WL Differential<br />
<strong>Broth</strong> is added to suppress yeast and any mold which may be<br />
present.<br />
69
Media<br />
Wallerstein Nutrient <strong>Broth</strong> (WL) and WL Differential <strong>Broth</strong> (WLD)<br />
Products<br />
Historical background:<br />
WL Nutrient Agar and WL Differential Agar were developed by GRAY and<br />
GREEN (1950), to study the microbial population of worts, beers, liquids,<br />
yeast, and other materials used in fermentation control procedures of the<br />
brewing industry and other fermentation industries.<br />
The WL Nutrient Agar is used for the cultivation and enumeration of yeast.<br />
At pH 5.5 brewers’ yeast will grow unrestricted at 25º C within 24–48 hours.<br />
For growth of bakers’ and distillers’ yeast, the pH of the medium should be<br />
adjusted to 6.5 with incubation at 30º C for 2–7 days. Where yeast cells are<br />
in low number, certain bacteria can be detected.<br />
The WL Differential <strong>Broth</strong> is used to determine bacterial counts. GRAY and<br />
GREEN (1950) recommended using the differential medium to culture<br />
bacteria normally encountered in brewing; aerobic conditions for acetic<br />
acid and thermophilic bacteria, anaerobic conditions for lactic bacteria and<br />
beer cocci.<br />
Wallerstein Differential <strong>Broth</strong> (WLD): Cultur of<br />
Lactobacillus plantarum ATCC 8014.<br />
Order information WL Nutrient Media<br />
Product Description Qty/Pkg Order No<br />
Ampoules 2 ml 50 10 496 108<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
Order information WL Differential Media<br />
Product Description Qty/Pkg Order No<br />
Ampoules 2 ml 50 10 496 109<br />
Bottled Media 50 ml 8 10 496 717*<br />
Petri dishes with sterile pads 47 mm 100 10 498 544<br />
Petri dishes with sterile pads 50 mm 50 10 445 905<br />
* available on request<br />
70