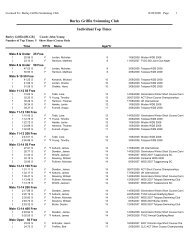
Year 7 Science Half Yearly Examination Revision Sheet Section 2 ...
Year 7 Science Half Yearly Examination Revision Sheet Section 2 ...
Year 7 Science Half Yearly Examination Revision Sheet Section 2 ...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
<strong>Year</strong> 7 <strong>Science</strong><br />
<strong>Half</strong> <strong>Year</strong>ly <strong>Examination</strong> <strong>Revision</strong> <strong>Sheet</strong> <strong>Section</strong> 2: Solids, Liquids and Gases and Mixtures<br />
and Their Separation<br />
You need to revise the following objectives in preparation for this section of the examination<br />
Solids, Liquids and Gases<br />
1. Describe the behaviour of matter in terms of particles that are continuously moving and<br />
interacting.<br />
2. Identify the three main states of matter.<br />
3. Describe how the particle model explains the different states of matter.<br />
4. List the properties of solids, liquids and gases.<br />
5. Relate the properties of solids, liquids and gases to the particle model of matter.<br />
6. Define the term "change in state".<br />
7. Describe the physical changes that take place during evaporation, condensation, boiling,<br />
melting, freezing and sublimation.<br />
8. Relate changes of state to the motion of particles as energy is removed or added.<br />
9. Relate energy transfers in evaporation, condensation, boiling, melting, freezing and<br />
sublimation to the particle model.<br />
10. Define the term "density" and explain density in terms of the particle model.<br />
11. Calculate the density of solids and liquids.<br />
Mixtures and their Separation<br />
1. Describe how a solution is formed.<br />
2. Define the terms "solute" and "solvent".<br />
3. Identify solutes and solvents in common solutions.<br />
4. Describe the difference between a "soluble" and an "insoluble" substance.<br />
5. Define the terms "concentrated", "dilute" and "saturated" as they apply to solutions.<br />
6. Describe how a suspension is formed and name some common suspensions.<br />
7. Describe the differences in appearance between solutions, suspensions and colloids.<br />
8. Describe the differences between solutions, suspensions and colloids in terms of particles.<br />
9. Identify, describe and outline everyday uses for the different methods of separating<br />
insoluble substances:<br />
· sedimentation (decanting);<br />
· centrifuging;<br />
· sieving;<br />
· magnetic separation;<br />
· filtering;<br />
· electrostatic separation;<br />
· gravity separation;<br />
· froth flotation.