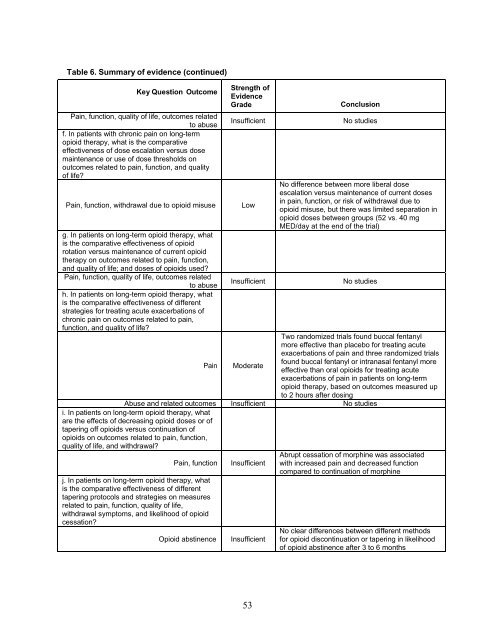
Table 6. Summary of evidence (continued)Key Question OutcomePain, function, quality of life, outcomes relatedto abusef. In patients with <strong>chronic</strong> <strong>pain</strong> on long-term<strong>opioid</strong> therapy, what is the comparativeeffectiveness of dose escalation versus dosemaintenance or use of dose thresholds onoutcomes related to <strong>pain</strong>, function, and qualityof life?Pain, function, withdrawal due to <strong>opioid</strong> misuseg. In patients on long-term <strong>opioid</strong> therapy, whatis the comparative effectiveness of <strong>opioid</strong>rotation versus maintenance of current <strong>opioid</strong>therapy on outcomes related to <strong>pain</strong>, function,and quality of life; and doses of <strong>opioid</strong>s used?Pain, function, quality of life, outcomes relatedto abuseh. In patients on long-term <strong>opioid</strong> therapy, whatis the comparative effectiveness of differentstrategies for treating acute exacerbations of<strong>chronic</strong> <strong>pain</strong> on outcomes related to <strong>pain</strong>,function, and quality of life?PainStrength ofEvidenceGradeInsufficientLowInsufficientModerateConclusionNo studiesNo difference between more liberal doseescalation versus maintenance of current dosesin <strong>pain</strong>, function, or risk of withdrawal due to<strong>opioid</strong> misuse, but there was limited separation in<strong>opioid</strong> doses between groups (52 vs. 40 mgMED/day at the end of the trial)No studiesTwo randomized trials found buccal fentanylmore effective than placebo for treating acuteexacerbations of <strong>pain</strong> and three randomized trialsfound buccal fentanyl or intranasal fentanyl moreeffective than oral <strong>opioid</strong>s for treating acuteexacerbations of <strong>pain</strong> in patients on long-term<strong>opioid</strong> therapy, based on outcomes measured upto 2 hours after dosingAbuse and related outcomes Insufficient No studiesi. In patients on long-term <strong>opioid</strong> therapy, whatare the effects of decreasing <strong>opioid</strong> doses or oftapering off <strong>opioid</strong>s versus continuation of<strong>opioid</strong>s on outcomes related to <strong>pain</strong>, function,quality of life, and withdrawal?Pain, functionj. In patients on long-term <strong>opioid</strong> therapy, whatis the comparative effectiveness of differenttapering protocols and strategies on measuresrelated to <strong>pain</strong>, function, quality of life,withdrawal symptoms, and likelihood of <strong>opioid</strong>cessation?Opioid abstinenceInsufficientInsufficientAbrupt cessation of morphine was associatedwith increased <strong>pain</strong> and decreased functioncompared to continuation of morphineNo clear differences between different methodsfor <strong>opioid</strong> discontinuation or tapering in likelihoodof <strong>opioid</strong> abstinence after 3 to 6 months53
Table 6. Summary of evidence (continued)Key Question Outcome4. Risk assessment and risk mitigationstrategiesa. In patients with <strong>chronic</strong> <strong>pain</strong> beingconsidered for long-term <strong>opioid</strong> therapy, what isthe accuracy of instruments for predicting risk of<strong>opioid</strong> overdose, addiction, abuse, or misuse?Diagnostic accuracy: Opioid Risk ToolDiagnostic accuracy: Screening and OpioidAssessment for Patients with Pain (SOAPP)version 1Strength ofEvidenceGradeInsufficientLowConclusionBased on a cutoff of >4, three studies (one poorquality,two poor-quality) <strong>report</strong>ed veryinconsistent estimates of diagnostic accuracy,precluding reliable conclusionsBased on a cutoff score of >8, sensitivity was0.68 and specificity of 0.38 in 1 study, for a PLRof 1.11 and NLR of 0.83. Based on a cutoff scoreof >6, sensitivity was 0.73 in 1 study.b. In patients with <strong>chronic</strong> <strong>pain</strong>, what is theeffectiveness of use of risk predictioninstruments on outcomes related to overdose,addiction, abuse, or misuse?Outcomes related to abuseInsufficientNo study evaluated the effectiveness of riskprediction instruments for reducing outcomesrelated to overdose, addiction, abuse, or misusec. In patients with <strong>chronic</strong> <strong>pain</strong> prescribed longterm<strong>opioid</strong> therapy, what is the effectiveness ofrisk mitigation strategies, including 1) <strong>opioid</strong>management plans, 2) patient education, 3)urine drug screening, 4) use of prescription drugmonitoring program data, 5) use ofmonitoring instruments, 6) more frequentmonitoring intervals, 7) pill counts, and 8) use ofabuse-deterrent formulations on outcomesrelated to overdose, addiction, abuse, ormisuse?Outcomes related to abuse Insufficient No studiesd. What is the comparative effectiveness of<strong>treatment</strong> strategies for managing patients withaddiction to prescription <strong>opioid</strong>s on outcomesrelated to overdose, abuse, misuse, <strong>pain</strong>,function, and quality of life?Outcomes related to abuse Insufficient No studiesAbbreviations: CI=confidence interval, HR=hazard ratio, MED= morphine equivalent dose, NLR=negative likelihood ratio,OR=odds ratio,PLR=positive likelihood ratio, SOAPP= Screening and Opioid Assessment for Patients with Pain.Findings in Relationship to What is Already KnownOur findings are generally consistent with prior systematic reviews of <strong>opioid</strong> therapy for<strong>chronic</strong> <strong>pain</strong> that also found no long-term, placebo-controlled randomized trials. 8, 44 Onesystematic review of outcomes associated with long-term <strong>opioid</strong> therapy concluded that manypatients discontinue <strong>treatment</strong> due to adverse events or insufficient <strong>pain</strong> relief, though patientswho continue <strong>opioid</strong> therapy experience clinically significant <strong>pain</strong> relief. 8 However, results ofthe studies included in this review are difficult to interpret because the studies had nonon<strong>opioid</strong> therapy control group, <strong>report</strong>ed substantial between-study heterogeneity, and weresusceptible to potential attrition and selection bias. Our findings are also consistent with asystematic review on comparative benefits and harms of various long-acting <strong>opioid</strong>s and54
- Page 1 and 2:
Evidence Report/Technology Assessme
- Page 4 and 5:
PrefaceThe Agency for Healthcare Re
- Page 6 and 7:
Charles Inturrisi, Ph.D.Professor o
- Page 8 and 9:
Robert Jamison, Ph.D.Professor of A
- Page 10 and 11:
Conclusions. Evidence on long-term
- Page 12 and 13:
no opioid on: (1) opioid abuse, add
- Page 14 and 15:
References ........................
- Page 16 and 17:
The purpose of this report is to sy
- Page 18 and 19:
patient education, (3) urine drug s
- Page 20 and 21:
Each abstract was independently rev
- Page 22 and 23:
We rated the quality of each cohort
- Page 24 and 25:
ResultsOverviewThe search and selec
- Page 26 and 27:
Key Question 4. Risk Assessment and
- Page 28 and 29:
Table A. Summary of evidence (conti
- Page 30 and 31:
Table A. Summary of evidence (conti
- Page 32 and 33:
Table A. Summary of evidence (conti
- Page 34 and 35:
Evidence on the effectiveness of di
- Page 36 and 37:
ApplicabilityA number of issues cou
- Page 38 and 39:
the exception of buccal or intranas
- Page 40:
References1. International Associat
- Page 43 and 44:
outcome study of long-term opioid d
- Page 45 and 46:
patients? Pain Physician. 2006;9(1)
- Page 47 and 48: American Academy of Pain Medicine,
- Page 49 and 50: d. What is the comparative effectiv
- Page 51 and 52: inclusion to English-language artic
- Page 53 and 54: adequately reported and acceptable;
- Page 55 and 56: eview comments will be made availab
- Page 57 and 58: Key Question 1aIn patients with chr
- Page 59 and 60: Key Question 2aIn patients with chr
- Page 61 and 62: and F2). 49-58 All were rated fair-
- Page 63 and 64: Table 1. Uncontrolled studies of lo
- Page 65 and 66: Table 1. Uncontrolled studies of lo
- Page 67 and 68: The annual overdose rate was 256 pe
- Page 69 and 70: The SOE for the association between
- Page 71 and 72: One fair-quality cross-sectional st
- Page 73 and 74: 5300 controls who did not visit the
- Page 75 and 76: Key Question 3cIn patients with chr
- Page 77 and 78: Table 2. Head-to-head trials and ob
- Page 79 and 80: Table 2. Head-to-head trials and ob
- Page 81 and 82: withdrawal due to opioid misuse, bu
- Page 83 and 84: Table 3. Trials of different strate
- Page 85 and 86: Table 3. Trials of different strate
- Page 87 and 88: • Two studies evaluated the Scree
- Page 89 and 90: Table 4. Studies of risk assessment
- Page 91 and 92: Detailed SynthesisThe APS review id
- Page 93 and 94: Evidence on the effectiveness of di
- Page 95 and 96: Table 6. Summary of evidence (conti
- Page 97: Table 6. Summary of evidence (conti
- Page 101 and 102: The setting in which studies were c
- Page 103 and 104: switching to other prescription opi
- Page 105 and 106: States: Fact Sheet. Atlanta, GA: Ce
- Page 107 and 108: overdose: a cohort study. Ann Inter
- Page 109 and 110: placebo-controlled study. Clin Ther
- Page 111 and 112: pain. Ann Intern Med. 2014;160(1):3
- Page 113 and 114: Appendix A. Search StrategiesDataba
- Page 115 and 116: 19. 18 and (random$ or control$ or
- Page 117 and 118: 11. (urine adj7 (screen$ or test$ o
- Page 119 and 120: 3. (alfentanil or alphaprodine or b
- Page 121 and 122: 25. Risk Reduction Behavior/ or Ris
- Page 123 and 124: KQ 4a-4c: Risk Prediction and Mitig
- Page 125 and 126: "meperidine" OR "meptazinol" OR (MH
- Page 127 and 128: PICOT Include ExcludeOutcomes • F
- Page 129 and 130: Moore TM, Jones T, Browder JH, et a
- Page 131 and 132: Banning A, Sjogren P, Kaiser F. Rea
- Page 133 and 134: study. Pain. 2004;108(1-2):17-27. P
- Page 135 and 136: Edlund MJ, Sullivan M, Steffick D,
- Page 137 and 138: Gianutsos L, Safrenek S. Is there a
- Page 139 and 140: China. Addiction. 2011;106(10):1801
- Page 141 and 142: study. Compr Psychiatry. 1979;20(1)
- Page 143 and 144: 2009;10(4):531-43. PMID: 19243306.
- Page 145 and 146: Mercadante S, Casuccio A, Tirelli W
- Page 147 and 148: 2003;107(3):486-92. PMID: 14506751.
- Page 149 and 150:
Quigley C. Hydromorphone for acute
- Page 151 and 152:
osteoarthritis-related pain: placeb
- Page 153 and 154:
2013;7(2):96-101. PMID: 23385449. E
- Page 155 and 156:
effect of maternal, postnatal, adol
- Page 157 and 158:
Vestergaard P, Rejnmark L, Mosekild
- Page 159 and 160:
Yulug B, Ozan E. Buprenorphine: a s
- Page 161 and 162:
Author, YearType of StudySettingDur
- Page 163 and 164:
Author, YearType of StudySettingDur
- Page 165 and 166:
Author, YearType of StudySettingDur
- Page 167 and 168:
Author,yearDunn,2010ScreenedEligibl
- Page 169 and 170:
Author,yearGomes,2011ScreenedEligib
- Page 171 and 172:
Author,yearLi, 2013Method For Asses
- Page 173 and 174:
Author,yearMethod For AssessingOutc
- Page 175 and 176:
Appendix Table E4. Observational St
- Page 177 and 178:
Author, YearKQLi, 2013 KQ2a Case-Co
- Page 179 and 180:
Appendix Table E5. Observational St
- Page 181 and 182:
0Appendix Table E6. Observational S
- Page 183 and 184:
Appendix Table E7. Trials of Differ
- Page 185 and 186:
Appendix Table E8a. Head-to-Head Tr
- Page 187 and 188:
AuthorYearStudydesignDurationWild 2
- Page 189 and 190:
Author,YearHartung,2007Type of Stud
- Page 191 and 192:
Author,YearKrebs,2011Type of Study,
- Page 193 and 194:
AuthorYearStudyDesignDurationRCT12
- Page 195 and 196:
Author yearAshburn, 2011Study Desig
- Page 197 and 198:
Author yearPortenoy, 2007Study Desi
- Page 199 and 200:
Author yearSimpson, 2007Study Desig
- Page 201 and 202:
Author yearStudy designDurationCowa
- Page 203 and 204:
Author YearRalphs, 1994StudyDesignD
- Page 205 and 206:
Author,Year Study DesignAkbik 2006
- Page 207 and 208:
Author,YearWebster2005Study DesignP
- Page 209 and 210:
Appendix F. Quality Assessment Tabl
- Page 211 and 212:
Author,YearGomes,2011Gomes,2013KQKQ
- Page 213 and 214:
Evaluatespopulationother than theon
- Page 215 and 216:
Key QuestionOutcomeStudy DesignNumb
- Page 217 and 218:
Key QuestionOutcomeStudy DesignNumb
- Page 219:
Key Question Outcome4. Risk assessm