Scientific Notation
Scientific Notation
Scientific Notation
- No tags were found...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
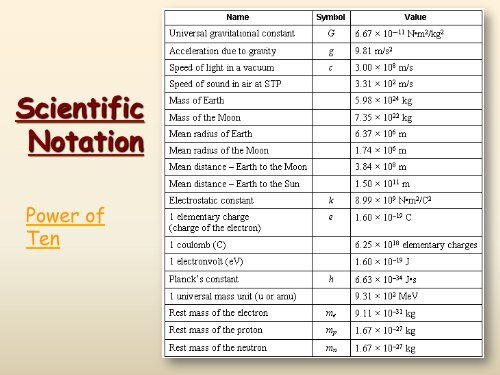
<strong>Scientific</strong><strong>Notation</strong>Power ofTen
<strong>Scientific</strong> <strong>Notation</strong>In science, we deal with some veryLARGE numbers:1 mole = 602000000000000000000000In science, we deal with some verySMALL numbers:Mass of an electron =0.000000000000000000000000000000091 kg
Imagine the difficulty of calculatingthe mass of 1 mole of electrons!0.000000000000000000000000000000091 kgx 602000000000000000000000???????????????????????????????????
<strong>Scientific</strong> <strong>Notation</strong>:A method of representing very large orvery small numbers in the form:M x 10 nØ M is a number between 1 - 9Ø n is an integer
2 500 000 000987.Step #1: Insert an understood decimal pointStep #2: Decide where the decimal must endup so that one number is to its leftStep #3: Count how many places you bouncethe decimal pointStep #4: Re-write in the form M x 10 n654321
2.5 x 10 9The exponent is thenumber of places wemoved the decimal.
0.00005791 2 3 4 5Step #2: Decide where the decimal must endup so that one number is to its leftStep #3: Count how many places you bouncethe decimal pointStep #4: Re-write in the form M x 10 n
5.79 x 10 -5The exponent is negativebecause the number westarted with was lessthan 1.
PERFORMINGCALCULATIONSIN SCIENTIFICNOTATIONADDITION AND SUBTRACTION
Review:<strong>Scientific</strong> notation expresses anumber in the form:1 ≤ M < 10M x 10 nn is aninteger
4 x 10 6 IF the exponents are+ 3 x 10 6 the same, we simply7 x 10 6add or subtract thenumbers in front andbring the exponentdown unchanged.
4 x 10 6- 3 x 10 6 The same holds truefor subtraction inscientific notation.1 x 10 6
4 x 10 6 If the exponents are+ 3 x 10 5 NOT the same, wemust move a decimalto make them thesame.
4.00 x 10 64.00 x 10 6+ 3.00 x 10 5 + .30 x 10 6Move the4.30 x 10 6decimal onthe smallernumber!
A Problem for you…2.37 x 10 -6+ 3.48 x 10 -4
Solution…002.37 x 10 -6+ 3.48 x 10 -4
Solution…0.0237 x 10 -4 cm+ 3.48 x 10 -4 cm3.5037 x 10 -4Adjust for sig figs (3)3.50 x 10 -4 cm
PERFORMINGCALCULATIONSIN SCIENTIFICNOTATIONMULTIPLICATION ANDDIVISION
In multiplication the exponents areADDED.4.35 x 10 6 cmx 3.1 x 10 3 cm(4.35 x 3.1) (10 6+3 )13.485 x 10 9 cm 21.3 x 10 10 cm 2
Remember: WHENPERFORMING CALCULATIONSIN SCIENTIFIC NOTATION:Always adjust the finalanswer to properscientific notation formatand adjust for sig figs!
Try This one:(5.23 x 10 6 µm) x (7.1 x 10 -2 µm)(5.23 x 7.1) (10 6+(-2) )37.133 x 10 4 µm 23.7 x 10 5 µm 2
In division the exponents aresubtracted.4.35 x 10 6 cm 23.1 x 10 3 cm4.353.1 x (106-3 )(cm 2 -cm) =1.403225806 x 10 3 cm1.4 x 10 3 cm
5.44 x 10 7 g8.1 x 10 4 mol)Try This one:5.448.1 x (107-4 )(g/mol)0.6716049383 x 10 3 g/mol6.7 x 10 2 g/mol