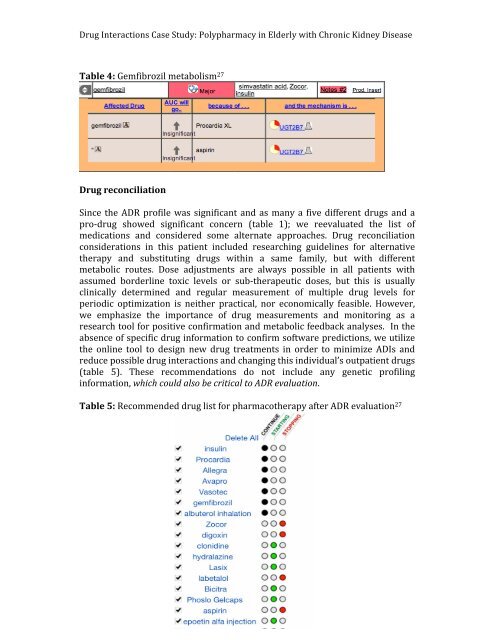
6 <strong>Drug</strong> Interactions Case Study: Polypharmacy <strong>in</strong> Elderly <strong>with</strong> Chronic Kidney Disease Table 3: Labetalol metabolism 27 Contribut<strong>in</strong>g to this <strong>patient</strong>’s s<strong>in</strong>us bradycardia, a meta-‐<strong>an</strong>alysis concluded that beta-‐blockers used for the treatment of hypertension are associated <strong>with</strong> <strong>in</strong>creased risk for new-‐onset diabetes mellitus, hypertriglyceridemia, bronchial asthma exacerbations, higher basel<strong>in</strong>e blood sugar <strong>an</strong>d masks hypoglycemic state 3, 23 . The <strong>patient</strong>’s history of MI <strong>an</strong>d uncontrolled hypertension currently requires multiple comb<strong>in</strong>ed <strong>an</strong>ti-‐hypertensive drugs for long-‐term m<strong>an</strong>agement. Moreover, some recent studies have argued aga<strong>in</strong>st rout<strong>in</strong>e beta-‐blocker use <strong>in</strong> <strong>patient</strong>s <strong>with</strong> hypertension despite current guidel<strong>in</strong>es, especially when comorbid contra<strong>in</strong>dications are present 15 . The most recent reports of the Jo<strong>in</strong>t National Committee (JNC VI) <strong>an</strong>d the World Health Org<strong>an</strong>ization recommend beta-‐blockers <strong>an</strong>d diuretics as first-‐l<strong>in</strong>e therapy for uncomplicated essential hypertension 11, 25 . Similar recommendations have been issued over the past few years by m<strong>an</strong>y authoritative sources <strong>an</strong>d research published <strong>in</strong> <strong>in</strong>fluential journals. These recommendations were supposedly based on multiple prospective r<strong>an</strong>domized trials attest<strong>in</strong>g that only beta-‐blockers <strong>an</strong>d diuretics, both <strong>in</strong> monotherapy <strong>an</strong>d comb<strong>in</strong>ed, reduced morbidity <strong>an</strong>d mortality <strong>in</strong> hypertension. Ever s<strong>in</strong>ce the Veter<strong>an</strong>s Adm<strong>in</strong>istration study <strong>in</strong> the 1970s 18 , multiple <strong>an</strong>d prospective r<strong>an</strong>domized trials have documented that diuretic-‐based therapy reduces the risk of stroke <strong>an</strong>d, to a lesser extent, of heart attacks <strong>an</strong>d cardiovascular morbidity <strong>an</strong>d mortality. However, the data are much less conv<strong>in</strong>c<strong>in</strong>g for beta-‐blockers 16 . In fact, no trial has shown that lower<strong>in</strong>g blood pressure <strong>with</strong> a beta-‐blocker reduces the risk of a heart attack or cardiovascular event <strong>in</strong> <strong>patient</strong>s <strong>with</strong> essential hypertension compared <strong>with</strong> placebo. In contrast, several prospective studies are now available show<strong>in</strong>g that blood pressure reduction <strong>with</strong> calcium ch<strong>an</strong>nel blockers dim<strong>in</strong>ishes cardiovascular morbidity <strong>an</strong>d mortality <strong>an</strong>d, at least <strong>in</strong> meta-‐<strong>an</strong>alysis, all-‐cause mortality. Therefore, this <strong>patient</strong> should benefit from stroke prevention <strong>an</strong>d <strong>an</strong>ti-‐hypertensive effects of diuretics (i.e. Lasix) <strong>an</strong>d optimize <strong>an</strong>tihypertensive therapy <strong>with</strong> current medications, but avoid<strong>in</strong>g the use of beta-‐blockers. We also f<strong>in</strong>d 100% <strong>in</strong>hibition of gemfibrozil metabolism when concurrently adm<strong>in</strong>istered <strong>with</strong> both: Procardia XL <strong>an</strong>d aspir<strong>in</strong> s<strong>in</strong>ce gemfibrozil is also metabolized through glucuronidation (table 4). Fibrates are a ma<strong>in</strong>stay <strong>in</strong> treat<strong>in</strong>g <strong>patient</strong>s <strong>with</strong> a subst<strong>an</strong>tial hypertriglyceridemic lipid abnormality. Seems logical to reconsider alternative therapy or adjust doses <strong>an</strong>d follow up <strong>with</strong> drug serum levels <strong>an</strong>d lipid p<strong>an</strong>els.
7 <strong>Drug</strong> Interactions Case Study: Polypharmacy <strong>in</strong> Elderly <strong>with</strong> Chronic Kidney Disease Table 4: Gemfibrozil metabolism 27<strong>Drug</strong> reconciliation S<strong>in</strong>ce the ADR profile was signific<strong>an</strong>t <strong>an</strong>d as m<strong>an</strong>y a five different drugs <strong>an</strong>d a pro-‐drug showed signific<strong>an</strong>t concern (table 1); we reevaluated the list of medications <strong>an</strong>d considered some alternate approaches. <strong>Drug</strong> reconciliation considerations <strong>in</strong> this <strong>patient</strong> <strong>in</strong>cluded research<strong>in</strong>g guidel<strong>in</strong>es for alternative therapy <strong>an</strong>d substitut<strong>in</strong>g drugs <strong>with</strong><strong>in</strong> a same family, but <strong>with</strong> different metabolic routes. Dose adjustments are always possible <strong>in</strong> all <strong>patient</strong>s <strong>with</strong> assumed borderl<strong>in</strong>e toxic levels or sub-‐therapeutic doses, but this is usually cl<strong>in</strong>ically determ<strong>in</strong>ed <strong>an</strong>d regular measurement of multiple drug levels for periodic optimization is neither practical, nor economically feasible. However, we emphasize the import<strong>an</strong>ce of drug measurements <strong>an</strong>d monitor<strong>in</strong>g as a research tool for positive confirmation <strong>an</strong>d metabolic feedback <strong>an</strong>alyses. In the absence of specific drug <strong>in</strong>formation to confirm software predictions, we utilize the onl<strong>in</strong>e tool to design new drug treatments <strong>in</strong> order to m<strong>in</strong>imize ADIs <strong>an</strong>d reduce possible drug <strong><strong>in</strong>teractions</strong> <strong>an</strong>d ch<strong>an</strong>g<strong>in</strong>g this <strong>in</strong>dividual’s out<strong>patient</strong> drugs (table 5). These recommendations do not <strong>in</strong>clude <strong>an</strong>y genetic profil<strong>in</strong>g <strong>in</strong>formation, which could also be critical to ADR evaluation. Table 5: Recommended drug list for pharmacotherapy after ADR evaluation 27