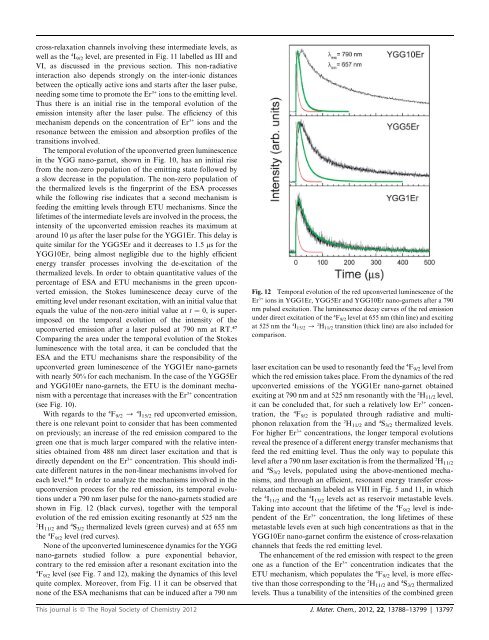
cross-relaxation channels involving these intermediate levels, aswell as the 4 I 9/2 level, are presented in Fig. 11 labelled as III <strong>and</strong>VI, as discussed in the previous section. This non-radiativeinteraction also depends strongly on the inter-ionic distancesbetween the optically active ions <strong>and</strong> starts after the laser pulse,needing some time to promote the Er 3+ ions to the emitting level.Thus there is an initial rise in the temporal evolution <strong>of</strong> theemission intensity after the laser pulse. The efficiency <strong>of</strong> thismechanism depends on the concentration <strong>of</strong> Er 3+ ions <strong>and</strong> theresonance between the emission <strong>and</strong> absorption pr<strong>of</strong>iles <strong>of</strong> thetransitions involved.The temporal evolution <strong>of</strong> the upconverted green <strong>luminescence</strong>in the YGG nano-garnet, shown in Fig. 10, has an initial risefrom the non-zero population <strong>of</strong> the emitting state followed bya slow decrease in the population. The non-zero population <strong>of</strong>the thermalized levels is the fingerprint <strong>of</strong> the ESA processeswhile the following rise indicates that a second mechanism isfeeding the emitting levels through ETU mechanisms. Since thelifetimes <strong>of</strong> the intermediate levels are involved in the process, theintensity <strong>of</strong> the upconverted emission reaches its maximum ataround 10 ms after the laser pulse for the YGG1Er. This delay isquite similar for the YGG5Er <strong>and</strong> it decreases to 1.5 ms for theYGG10Er, being almost negligible due to the highly efficientenergy transfer processes involving the de-excitation <strong>of</strong> thethermalized levels. In order to obtain quantitative values <strong>of</strong> thepercentage <strong>of</strong> ESA <strong>and</strong> ETU mechanisms in the green upconvertedemission, the Stokes <strong>luminescence</strong> decay curve <strong>of</strong> theemitting level under resonant excitation, with an initial value thatequals the value <strong>of</strong> the non-zero initial value at t ¼ 0, is superimposedon the temporal evolution <strong>of</strong> the intensity <strong>of</strong> theupconverted emission after a laser pulsed at 790 nm at RT. 47Comparing the area under the temporal evolution <strong>of</strong> the Stokes<strong>luminescence</strong> with the total area, it can be concluded that theESA <strong>and</strong> the ETU mechanisms share the responsibility <strong>of</strong> theupconverted green <strong>luminescence</strong> <strong>of</strong> the YGG1Er nano-garnetswith nearly 50% for each mechanism. In the case <strong>of</strong> the YGG5Er<strong>and</strong> YGG10Er nano-garnets, the ETU is the dominant mechanismwith a percentage that increases with the Er 3+ concentration(see Fig. 10).With regards to the 4 F 9/2 / 4 I 15/2 red upconverted emission,there is one relevant point to consider that has been commentedon previously; an increase <strong>of</strong> the red emission compared to thegreen one that is much larger compared with the relative intensitiesobtained from 488 nm direct laser excitation <strong>and</strong> that isdirectly dependent on the Er 3+ concentration. This should indicatedifferent natures in the non-linear mechanisms involved foreach level. 41 In order to analyze the mechanisms involved in theupconversion process for the red emission, its temporal evolutionsunder a 790 nm laser pulse for the nano-garnets studied areshown in Fig. 12 (black curves), together with the temporalevolution <strong>of</strong> the red emission exciting resonantly at 525 nm the2H 11/2 <strong>and</strong> 4 S 3/2 thermalized levels (green curves) <strong>and</strong> at 655 nmthe 4 F 9/2 level (red curves).None <strong>of</strong> the upconverted <strong>luminescence</strong> dynamics for the YGGnano-garnets studied follow a pure exponential behavior,contrary to the red emission after a resonant excitation into the4F 9/2 level (see Fig. 7 <strong>and</strong> 12), making the dynamics <strong>of</strong> this levelquite complex. Moreover, from Fig. 11 it can be observed thatnone <strong>of</strong> the ESA mechanisms that can be induced after a 790 nmFig. 12 Temporal evolution <strong>of</strong> the red upconverted <strong>luminescence</strong> <strong>of</strong> theEr 3+ ions in YGG1Er, YGG5Er <strong>and</strong> YGG10Er nano-garnets after a 790nm pulsed excitation. The <strong>luminescence</strong> decay curves <strong>of</strong> the red emissionunder direct excitation <strong>of</strong> the 4 F 9/2 level at 655 nm (thin line) <strong>and</strong> excitingat 525 nm the 4 I 15/2 / 2 H 11/2 transition (thick line) are also included forcomparison.laser excitation can be used to resonantly feed the 4 F 9/2 level fromwhich the red emission takes place. From the dynamics <strong>of</strong> the redupconverted emissions <strong>of</strong> the YGG1Er nano-garnet obtainedexciting at 790 nm <strong>and</strong> at 525 nm resonantly with the 2 H 11/2 level,it can be concluded that, for such a relatively low Er 3+ concentration,the 4 F 9/2 is populated through radiative <strong>and</strong> multiphononrelaxation from the 2 H 11/2 <strong>and</strong> 4 S 3/2 thermalized levels.For higher Er 3+ concentrations, the longer temporal evolutionsreveal the presence <strong>of</strong> a different energy transfer mechanisms thatfeed the red emitting level. Thus the only way to populate thislevel after a 790 nm laser excitation is from the thermalized 2 H 11/2<strong>and</strong> 4 S 3/2 levels, populated using the above-mentioned mechanisms,<strong>and</strong> through an efficient, resonant energy transfer crossrelaxationmechanism labeled as VIII in Fig. 5 <strong>and</strong> 11, in whichthe 4 I 11/2 <strong>and</strong> the 4 I 13/2 levels act as reservoir metastable levels.Taking into account that the lifetime <strong>of</strong> the 4 F 9/2 level is independent<strong>of</strong> the Er 3+ concentration, the long lifetimes <strong>of</strong> thesemetastable levels even at such high concentrations as that in theYGG10Er nano-garnet confirm the existence <strong>of</strong> cross-relaxationchannels that feeds the red emitting level.The enhancement <strong>of</strong> the red emission with respect to the greenone as a function <strong>of</strong> the Er 3+ concentration indicates that theETU mechanism, which populates the 4 F 9/2 level, is more effectivethan those corresponding to the 2 H 11/2 <strong>and</strong> 4 S 3/2 thermalizedlevels. Thus a tunability <strong>of</strong> the intensities <strong>of</strong> the combined greenThis journal is ª The Royal Society <strong>of</strong> Chemistry 2012 J. Mater. Chem., 2012, 22, 13788–13799 | 13797
<strong>and</strong> red upconverted emissions is possible in the Er 3+ -<strong>doped</strong>YGG nano-garnets.DConclusionsThe synthesis <strong>of</strong> Er 3+ -<strong>doped</strong> Y 3 Ga 5 O 12 (YGG) nanocrystals hasbeen successfully carried out via a citrate sol–gel synthesis. X-raydiffraction measurements confirm that the nanocrystals areformed in a single phase <strong>of</strong> the garnet <strong>structure</strong> with an averagegrain size <strong>of</strong> 60 nm, whereas HRTEM pictures show that thenanoparticles are agglomerated in interconnected spherical <strong>and</strong>elliptical shapes. The vibrational properties <strong>of</strong> the YGG nanogarnetsdetermined by Raman <strong>and</strong> FTIR spectroscopy alsoconfirm the good crystalline nature <strong>of</strong> the single phase nanoparticles<strong>and</strong> show that several impurities are attached to thenanocrystal surface. The peaks observed in the absorption <strong>and</strong><strong>luminescence</strong> spectra are due to the incorporation <strong>of</strong> the Er 3+ions into the low-phonon energy dodecahedral site, replacing theY 3+ position, since the interaction <strong>of</strong> the Er 3+ ions with theoxygen lig<strong>and</strong>s in a D 2 local point symmetry completely breaksthe degeneracy <strong>of</strong> the free-Er 3+ multiplets. When exciting the Er 3+ions in the blue range, intense green, red <strong>and</strong> infrared emissionsare observed, starting from four emitting levels, whose intensitiesstrongly depend on the Er 3+ concentration. Different <strong>luminescence</strong>dynamics observed for the different emitting levels for thehigher <strong>doped</strong> nano-garnets indicate the presence <strong>of</strong> differentcross-relaxation mechanisms between Er 3+ ions <strong>and</strong> the transfer<strong>of</strong> energy to quenching traps. The red-to-green intensity ratiosincrease due to cross-relaxation channels that efficiently quenchthe radiative de-excitation <strong>of</strong> the 2 H 11/2 , 4 S 3/2 green emittinglevels, whereas that <strong>of</strong> the 4 I 9/2 red emitting level is not affected.Under cw 790 nm laser excitation the Er 3+ -<strong>doped</strong> YGG nanogarnetsshow bright <strong>and</strong> intense green <strong>and</strong> red upconverted<strong>luminescence</strong>. Different dynamics are observed for the green <strong>and</strong>red upconverted emissions. Both ESA <strong>and</strong> ETU mechanisms areinvolved in the upconverted green <strong>luminescence</strong> <strong>of</strong> the YGG1Ernano-garnets with nearly 50% for each mechanism, whereasETU is dominant in the upconverted green <strong>luminescence</strong> <strong>of</strong> theYGG5Er <strong>and</strong> YGG10Er nano-garnets. The enhancement <strong>of</strong> thered emission with respect to the green one for the high-<strong>doped</strong>nano-garnets is ascribed to the existence <strong>of</strong> cross-relaxationchannels activated by the infrared laser. This ETU mechanismpopulates the 4 F 9/2 level more efficiently than those correspondingto the 2 H 11/2 <strong>and</strong> 4 S 3/2 thermalized levels. These resultsindicate that a tunability <strong>of</strong> the intensities <strong>of</strong> the combined green<strong>and</strong> red upconverted emissions is possible in the Er 3+ -<strong>doped</strong>YGG nano-garnets, making the YGG nano-garnet a potential<strong>and</strong> interesting material in different optical devices such as intwo-dimensional displays or upconversion lasers.AcknowledgementsThis work has been partially supported by Ministerio de Cienciae Innovacion <strong>of</strong> Spain (MICCIN) under The National Program<strong>of</strong> Materials (MAT2010-21270-C04-02; -03; -04), The Consolider-Ingenio2010 Program (MALTA CSD2007-0045), <strong>and</strong>The National Infra<strong>structure</strong> Program, by Ministerio de Economıay Competitividad <strong>of</strong> Spain (MINECO) within The Indo-Spanish Joint Programme <strong>of</strong> Cooperation in Science <strong>and</strong>Technology (PRI-PIBIN-2011-1153/DST-INT-Spain-P-38-11),<strong>and</strong> by the EU-FEDER funds (UCAN08-4E-008). S.F. Leon-Luis <strong>and</strong> V. Monteseguro wish to thank MICINN for the FPIgrants (BES-2008-003353 <strong>and</strong> BES-2011-044596). Dr V. Venkatramuis grateful to DAE-BRNS, Government <strong>of</strong> India for theaward <strong>of</strong> DAE Research Award for Young Scientists (no. 2010/20/34/5/BRNS/2223).Notes <strong>and</strong> references1 A. Speghini, F. Piccinelli <strong>and</strong> M. Bettinelli, Opt. Mater., 2011, 33, 247.2 F. Wang <strong>and</strong> X. Liu, Chem. Soc. Rev., 2009, 38, 976.3 V. Venkatramu, D. Falcomer, A. Speghini, M. Bettinelli <strong>and</strong>C. K. Jayasankar, J. Lumin., 2008, 128, 811.4 V. Venkatramu, M. Giarola, G. Mariotto, S. Enzo, S. Polizzi,C. K. Jayasankar, F. Piccinelli, M. Bettinelli <strong>and</strong> A. Speghini,Nanotechnology, 2010, 21, 175703.5 F. Vetrone, J. C. Boyer, J. A. Capobianco, A. Speghini <strong>and</strong>M. Bettinelli, J. Phys. Chem. B, 2003, 107, 1107.6 D. Hreniak, W. Strek, P. Gluchowski, M. Bettinelli <strong>and</strong> A. Speghini,Appl. Phys. B: Lasers Opt., 2008, 91, 89.7 E. Caponetti, D. C. Martino, M. L. Saladino <strong>and</strong> C. Leonelli,Langmuir, 2007, 23, 3947.8 R. Krsmanovic, V. A. Morozov, O. I. Lebedev, S. Polizzi,A. Speghini, M. Bettinelli <strong>and</strong> G. Van Tendeloo, Nanotechnology,2007, 18, 325604.9 R. Naccache, F. Vetrone, A. Speghini, M. Bettinelli <strong>and</strong>J. A. Capobianco, J. Phys. Chem. C, 2008, 112, 7750.10 F. Auzel, Chem. Rev., 2004, 104, 139.11 G. L. Vossler, C. L. Brooks <strong>and</strong> K. A. Winik, Electron. Lett., 1995, 31,1162.12 T. H. Whitley, C. A. Millar, R. Wyatt, M. C. Brierley <strong>and</strong>D. Szebesta, Electron. Lett., 1991, 27, 1785.13 J. E. Roman, P. Camy, M. Hempstead, W. S. Brocklesby, S. Nouth,A. Beguin, C. Lerminiaux <strong>and</strong> J. S. Wilkinson, Electron. Lett., 1995,31, 1345.14 A. Pollack <strong>and</strong> D. B. Chang, J. Appl. Phys., 1988, 64, 2885.15 L. H. Spangler, B. Farris, E. D. Filer <strong>and</strong> N. P. Bames, J. Appl. Phys.,1996, 79, 573.16 V. Mahalingam, F. Mangiarini, F. Vetrone, V. Venkatramu,M. Bettinelli, A. Speghini <strong>and</strong> J. A. Capobianco, J. Phys. Chem. C,2008, 112, 17745.17 G. H. Dieke <strong>and</strong> H. M. Crosswhite, Appl. Opt., 1963, 2, 675.18 S. S<strong>and</strong>ers, R. G. Waarts, D. G. Mehuys <strong>and</strong> D. F. Welch, Appl. Phys.Lett., 1995, 67, 1815.19 F. Hanson, Appl. Phys. Lett., 1995, 66, 3549.20 P. E.-A. M€obert, E. Heumann, G. Huber <strong>and</strong> B. H. T. Chai, Appl.Phys. Lett., 1998, 73, 139.21 F. Yu, D. Yuan, X. Cheng, X. Duan, X. Wang, L. Kong, L. Wang<strong>and</strong> Z. Li, Mater. Lett., 2007, 61, 2322.22 Y. X. Pan, M. Wu <strong>and</strong> Q. Su, Mater. Sci. Eng., B, 2004, 106, 251.23 A. Katelnikovas <strong>and</strong> A. Kareiva, Mater. Lett., 2008, 62, 1655.24 H. M. Rietveld, J. Appl. Crystallogr., 1969, 2, 65.25 J. Rodrıguez-Carvajal, Phys. Rev. B: Condens. Matter Mater. Phys.,1993, 192, 55.26 N. Matsushita, N. Tsuchiya, K. Kanatsuka <strong>and</strong> T. Yanagitani, J. Am.Ceram. Soc., 1999, 82, 1977.27 F. Euler <strong>and</strong> J. A. Bruce, Acta Crystallogr., 1965, 19, 971.28 K. Binnemans <strong>and</strong> C. J. G€orller-Walr<strong>and</strong>, Phys. Rev. B: Condens.Matter Mater. Phys., 1997, 9, 1637.29 K. Papagelis, J. Arvanitidis, E. Vinga, D. Christ<strong>of</strong>ilos,G. A. Kourouklis, H. Kimura <strong>and</strong> S. Ves, J. Appl. Phys., 2010, 107,113504.30 A. Leleckaite <strong>and</strong> A. Kareiva, Opt. Mater., 2004, 26, 123.31 J. P. Hurrell, S. P. S. Porto, I. F. Chang, S. S. Mitra <strong>and</strong>R. P. Bauman, Phys. Rev., 1958, 173, 851.32 R. L. Wadsack, J. L. Lewis, B. E. Argyle <strong>and</strong> B. K. Chang, Phys. Rev.B: Solid State, 1971, 3, 4342.33 J. J. Song, P. B. Klein, R. L. Wadsack, M. Selders, S. Mroczkowski<strong>and</strong> R. K. Chang, J. Opt. Soc. Am., 1973, 63, 1135.34 M. C. Saine, E. Husson <strong>and</strong> H. Brusset, Spectrochim. Acta, Part A,1982, 38, 25.13798 | J. Mater. Chem., 2012, 22, 13788–13799 This journal is ª The Royal Society <strong>of</strong> Chemistry 2012