Geol. 655 Isotope Geochemistry<strong>Lecture</strong> <strong>10</strong> Spring 2003much older than the eruption age. <strong>Th</strong>issuggests the crystals in the magma may havecrystallized as much as 20,000 years or morebefore eruption. <strong>Th</strong>e age 226 Ra– 230 <strong>Th</strong> is lessprecise because the slope is only slightly lessthan 1, and all samples seem to contain excess226 Ra. Nevertheless, the slope of the data isclearly greater than that of a 2 ka isochron,but significantly less than 1, so these data alsosuggest the crystallization significantlypredated eruption. According to Volpe andHammond (1991) the difference in the230 <strong>Th</strong>– 238 U and 226 Ra– 230 <strong>Th</strong> ages may reflectchanges in melt composition, perhaps due toassimilation of country rock or addition of aRa-rich gas or fluid phase to the magma.230 <strong>Th</strong>– 238 U and 226 Ra– 230 <strong>Th</strong> ages of the morerecent eruptive products of Mt. St. Helens,including the 1980-1986 eruptive phase, werecloser to the eruption ages. For example,Volpe and Hammond (1991) obtained a230 <strong>Th</strong>– 238 U age of 6±4 ka on the 1982 dacite.One possible interpretation of this is thatwhen the most recent eruptive stage of Mt. St.Helens began about 4000 years ago, there wassignificant volume of older magma stillpresent in the magma chamber, and that thisolder magma has been progressively replacedby fresh magma over the past 4000 years.2<strong>10</strong> Pb DatingVolpe and Hammond (1991).2<strong>10</strong> Pb has a much shorter half-life (22.3 yrs) than the nuclides we have discussed thus far, so it isuseful for much shorter time intervals. It has been applied to <strong>dating</strong> rapidly accumulating lakesediments and glacial ice. Like <strong>Th</strong>, Pb is only weakly soluble and very particle active and is scavengedby clay and other particles settling through lake water. Lake sediments thus often have significantunsupported 2<strong>10</strong> Pb in their upper intervals.We can write an equation analogous to the one equation we wrote for unsupported 230 <strong>Th</strong> (<strong>10</strong>.14):Again letting t = z/s, we have:2<strong>10</strong>Pb2<strong>10</strong>Pbu =u =2<strong>10</strong>Pb2<strong>10</strong>PbFigure <strong>10</strong>.5. 230 <strong>Th</strong>– 238 U and 226 Ra– 230 <strong>Th</strong> mineral isochronsfor the Castle Creek andesite of Mt. St.Helens. 14 C and tree ring chronology indicates thislava was erupted 1700-2200 years ago.0u e–l 2<strong>10</strong>t0u e–l 2<strong>10</strong>z/sAfter<strong>10</strong>.21<strong>10</strong>.22<strong>Th</strong>us we would expect the unsupported 2<strong>10</strong> Pb to <strong>decay</strong> exponentially with depth.It is generally not useful to ratio 2<strong>10</strong> Pb to one of the stable isotopes of Pb for several reasons. First, a<strong>decay</strong>ing 238 U will pass through a stage as 222 Rn, a gas, before <strong>decay</strong>ing to 2<strong>10</strong> Pb. Some of this gas willescape into the atmosphere. However, its half-life is only 4 days and the nuclides between 222 Rn and2<strong>10</strong> Pb are all very short-lived, so this Rn will quickly <strong>decay</strong> to Pb. <strong>Th</strong>e Pb will be adsorbed on particlesand washed out of the atmosphere by rain. <strong>Th</strong>us the pathway of some 2<strong>10</strong> Pb to sediment will bedifferent that that of normal Pb. Second, there has been a very substantial input of anthropogenic(stable) Pb to lakes and streams from leaded gasoline over the 20 th century. <strong>Th</strong>us for this reason aswell, the geochemistry of stable Pb in modern sediments is different from that of 2<strong>10</strong> Pb. For both these67 February 12, 2003
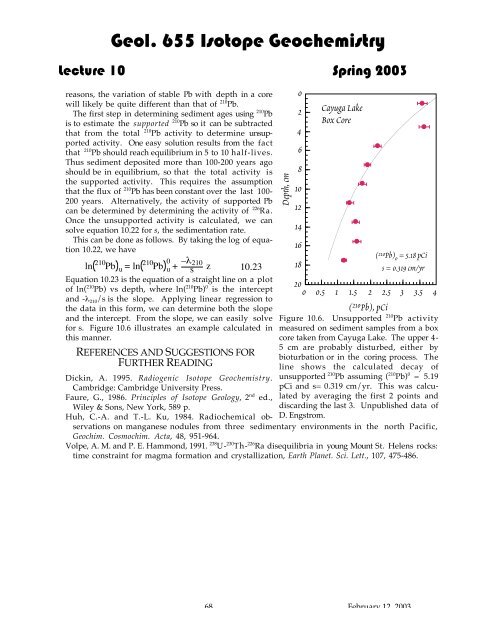
.Geol. 655 Isotope Geochemistry<strong>Lecture</strong> <strong>10</strong> Spring 2003reasons, the variation of stable Pb with depth in a corewill likely be quite different than that of 2<strong>10</strong> Pb.<strong>Th</strong>e first step in determining sediment ages using 2<strong>10</strong> Pbis to estimate the supported 2<strong>10</strong> Pb so it can be subtractedthat from the total 2<strong>10</strong> Pb activity to determine unsupportedactivity. One easy solution results from the factthat 2<strong>10</strong> Pb should reach equilibrium in 5 to <strong>10</strong> half-lives.<strong>Th</strong>us sediment deposited more than <strong>10</strong>0-200 years agoshould be in equilibrium, so that the total activity isthe supported activity. <strong>Th</strong>is requires the assumptionthat the flux of 2<strong>10</strong> Pb has been constant over the last <strong>10</strong>0-200 years. Alternatively, the activity of supported Pbcan be determined by determining the activity of 226 Ra.Once the unsupported activity is calculated, we cansolve equation <strong>10</strong>.22 for s, the sedimentation rate.<strong>Th</strong>is can be done as follows. By taking the log of equation<strong>10</strong>.22, we have2<strong>10</strong>ln Pb u = ln 2<strong>10</strong>Pb 0 –lu + 2<strong>10</strong>s z <strong>10</strong>.23Equation <strong>10</strong>.23 is the equation of a straight line on a plotof ln( 2<strong>10</strong> Pb) vs depth, where ln( 2<strong>10</strong> Pb) 0 is the interceptand -l 2<strong>10</strong> /s is the slope. Applying linear regression tothe data in this form, we can determine both the slopeand the intercept. From the slope, we can easily solvefor s. Figure <strong>10</strong>.6 illustrates an example calculated inthis manner.REFERENCES AND SUGGESTIONS FORFURTHER READINGDickin, A. 1995. Radiogenic Isotope Geochemistry.Cambridge: Cambridge University Press.Faure, G., 1986. Principles of Isotope Geology, 2 nd ed.,Wiley & Sons, New York, 589 p.Huh, C.-A. and T.-L. Ku, 1984. Radiochemical observationson manganese nodules from three sedimentary environments in the north Pacific,Geochim. Cosmochim. Acta, 48, 951-964.Volpe, A. M. and P. E. Hammond, 1991. 238 U- 230 <strong>Th</strong>- 226 Ra disequilibria in young Mount St. Helens rocks:time constraint for magma formation and crystallization, Earth Planet. Sci. Lett., <strong>10</strong>7, 475-486.Depth, cm02468<strong>10</strong>12141618Cayuga LakeBox CoreJJJJJJ200 0.5 1 1.5 2 2.5 3 3.5 4( 2<strong>10</strong> Pb), pCiJJJ( 2<strong>10</strong> Pb) 0 = 5.18 pCis = 0.319 cm/yrFigure <strong>10</strong>.6. Unsupported 2<strong>10</strong> Pb activitymeasured on sediment samples from a boxcore taken from Cayuga Lake. <strong>Th</strong>e upper 4-5 cm are probably disturbed, either bybioturbation or in the coring process. <strong>Th</strong>eline shows the calculated <strong>decay</strong> ofunsupported 2<strong>10</strong> Pb assuming ( 2<strong>10</strong> Pb) 0 = 5.19pCi and s= 0.319 cm/yr. <strong>Th</strong>is was calculatedby averaging the first 2 points anddiscarding the last 3. Unpublished data ofD. Engstrom.68 February 12, 2003