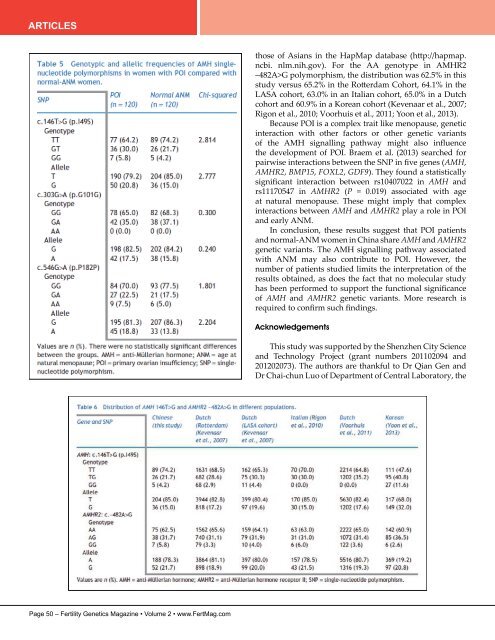
ARTICLES those of Asians in the HapMap database (http://hapmap. ncbi. nlm.nih.gov). For the AA genotype in AMHR2 –482A>G polymorphism, the distribution was 62.5% in this study versus 65.2% in the Rotterdam Cohort, 64.1% in the LASA cohort, 63.0% in an Italian cohort, 65.0% in a Dutch cohort and 60.9% in a Korean cohort (Kevenaar et al., 2007; Rigon et al., 2010; Voorhuis et al., 2011; Yoon et al., 2013). Because POI is a complex trait like menopause, genetic interaction with other factors or other genetic variants of the AMH signalling pathway might also influence the development of POI. Braem et al. (2013) searched for pairwise interactions between the SNP in five genes (AMH, AMHR2, BMP15, FOXL2, GDF9). They found a statistically significant interaction between rs10407022 in AMH and rs11170547 in AMHR2 (P = 0.019) associated with age at natural menopause. These might imply that complex interactions between AMH and AMHR2 play a role in POI and early ANM. In conclusion, these results suggest that POI patients and normal-ANM women in China share AMH and AMHR2 genetic variants. The AMH signalling pathway associated with ANM may also contribute to POI. However, the number of patients studied limits the interpretation of the results obtained, as does the fact that no molecular study has been performed to support the functional significance of AMH and AMHR2 genetic variants. More research is required to confirm such findings. Acknowledgements This study was supported by the Shenzhen City Science and Technology Project (grant numbers 201102094 and 201202073). The authors are thankful to Dr Qian Gen and Dr Chai-chun Luo of Department of Central Laboratory, the Page 50 – Fertility Genetics Magazine • Volume 2 • www.FertMag.com
ARTICLES Affiliated Shenzhen City Maternity and Child Healthcare Hospital of Southern Medical University, Shenzhen, PR China for technical help and advice. References Braem, M.G., Voorhuis, M., van der Schouw, Y.T., Peeters, P.H., Schouten, L.J., Eijkemans, M.J., Broekmans, F.J., Onland- Moret, N.C., 2013. Interactions between genetic variants in AMH and AMHR2 may modify age at natural menopause. PLoS ONE 8, e59819. de Vet, A., Laven, J.S., de Jong, F.H., Themmen, A.P., Fauser, B.C., 2002. Antimüllerian hormone serum levels: a putative marker for ovarian aging. Fertil. Steril. 77, 357–362. di Clemente, N., Josso, N., Gouédard, L., Belville, C., 2003. Components of the anti-Müllerian hormone signaling pathway in gonads. Mol. Cell. Endocrinol. 15, 9–14. Durlinger, A.L., Kramer, P., Karels, B., de Jong, F.H., Uilenbroek, J.T., Grootegoed, J.A., Themmen, A.P., 1999. Control of primordial follicle recruitment by anti-Müllerian hormone in the mouse ovary. Endocrinology 140, 5789–5796. Durlinger, A.L., Gruijters, M.J., Kramer, P., Karels, B., Kumar, T.R., Matzuk, M.M., Rose, U.M., de Jong, F.H., Uilenbroek, J.T., Grootegoed, J.A., Themmen, A.P., 2001. Anti-Müllerian hormone attenuates the effects of FSH on follicle development in the mouse ovary. Endocrinology 142, 4891–4899. Goswami, D., Conway, G.S., 2005. Premature ovarian failure. Hum. Reprod. Update 11, 391–410. Gruijters, M.J., Visser, J.A., Durlinger, A.L., Themmen, A.P., 2003. Anti-Müllerian hormone and its role in ovarian function. Mol. Cell. Endocrinol. 15, 85–90. Kevenaar, M.E., Themmen, A.P., Laven, J.S., Sonntag, B., Fong, S.L., Uitterlinden, A.G., de Jong, F.H., Pols, H.A., Simoni, M., Visser, J.A., 2007. Anti-Müllerian hormone and anti-Müllerian hormone type II receptor polymorphisms are associated with follicular phase estradiol levels in normo-ovulatory women. Hum. Reprod. 22, 1547–1554. Kevenaar, M.E., Laven, J.S., Fong, S.L., Uitterlinden, A.G., de Jong, F.H., Themmen, A.P., Visser, J.A., 2008. A functional anti-Müllerian hormone gene polymorphism is associated with follicle number and androgen levels in polycystic ovary syndrome patients. J. Clin. Endocrinol. Metab. 93, 1310–1316. Li, H.W., Anderson, R.A., Yeung, W.S., Ho, P.C., Ng, E.H., 2011. Evaluation of serum antimullerian hormone and inhibin B concentrations in the differential diagnosis of secondary oligoamenorrhea. Fertil. Steril. 96, 774–779. Messika-Zeitoun, L., Gouédard, L., Belville, C., Dutertre, M., Lins, L., Imbeaud, S., Hughes, I.A., Picard, J.Y., Josso, N., di Clemente, N., 2001. Autosomal recessive segregation of a truncating mutation of anti-Müllerian type II receptor in a family affected by the persistent Müllerian duct syndrome contrasts with its dominant negative activity in vitro. J. Clin. Endocrinol. Metab. 86, 4390–4397. Nie, G.N., Wang, X.Y., Yang, H.Y., Aihua, O.U., 2011. The investigation and analysis of the factors related with the menopausal age of urban women in China. CMCHC (CHIN) 8, 1191–1193. Qin, C.R., Chen, S.L., Yao, J.L., Wu, W.Q., Xie, J.S., 2011. Identification of novel missense mutations of the TGFBR3 gene in Chinese women with premature ovarian failure. Reprod. Biomed. Online 23, 697–703. Rigon, C., Andrisani, A., Forzan, M., D’Antona, D., Bruson, A., Cosmi, E., Ambrosini, G., Tiboni, G.M., Clementi, M., 2010. Association study of AMH and AMHRII polymorphisms with unexplained infertility. Fertil. Steril. 94, 1244–1248. Santoro, N., 2003. Mechanisms of premature ovarian failure. Ann. Endocrinol. 64, 87–92. Shelling, A.N., Burton, K.A., Chand, A.L., van Ee, C.C., France, J.T., Farquhar, C.M., Milsom, S.R., Love, D.R., Gersak, K., Aittomäki, K., Winship, I.M., 2000. Inhibin: a candidate gene for premature ovarian failure. Hum. Reprod. 15, 2644–2649. Tibiletti, M.G., Testa, G., Vegetti, W., Alagna, F., Taborelli, M., Dalprà, L., Bolis, P.F., Crosignani, P.G., 1999. The idiopathic forms of premature menopause and early menopause show the same genetic pattern. Hum. Reprod. 14, 2731–2734. Timmreck, L.S., Reindollar, R.H., 2003. Contemporary issues in primary amenorrhea. Obstet. Gynecol. Clin. North Am. 30, 287–302. van Rooij, I.A., Broekmans, F.J., te Velde, E.R., Fauser, B.C., Bancsi, L.F., de Jong, F.H., Themmen, A.P., 2002. Serum anti-Müllerian hormone levels: a novel measure of ovarian reserve. Hum. Reprod. 17, 3065–3071. Voorhuis, M., Onland-Moret, N.C., van der Schouw, Y.T., Fauser, B.C., Broekmans, F.J., 2010. Human studies on genetics of the age at natural menopause. Hum. Reprod. Update 16, 364–377. Voorhuis, M., Broekmans, F.J., Fauser, B.C., Onland-Moret, N.C., van der Schouw, Y.T., 2011. Genes involved in initial follicle recruitment may be associated with age at menopause. J. Clin. Endocrinol. Metab. 96, E473–E479. Weenen, C., Laven, J.S., Von Bergh, A.R., Cranfield, M., Groome, N.P., Visser, J.A., Kramer, P., Fauser, B.C., Themmen, A.P., 2004. Anti-Müllerian hormone expression pattern in the human ovary: potential implications for initial and cyclic follicle recruitment. Mol. Hum. Reprod. 10, 77–83. Yoon, S.H., Choi, Y.M., Hong, M.A., Kim, J.J., Lee, G.H., Hwang, K.R., Moon, S.Y., 2013. Association study of anti- Müllerian hormone and anti-Müllerian hormone type II receptor polymorphisms with idiopathic primary ovarian insufficiency. Hum. Reprod. 28, 3301–3305. Fertility Genetics Magazine • Volume 2 • www.FertMag.com – Page 51