• Patchless Transdermal Drug Delivery ... - Ecn5.com ecn5
• Patchless Transdermal Drug Delivery ... - Ecn5.com ecn5
• Patchless Transdermal Drug Delivery ... - Ecn5.com ecn5
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
VOLUME 3 NUMBER 2 Exclusively for the delivery of pharmaceuticals through the skin<br />
MARCH 2011<br />
<strong>•</strong> <strong>Patchless</strong> <strong>Transdermal</strong> <strong>Drug</strong> <strong>Delivery</strong><br />
<strong>•</strong> <strong>Transdermal</strong> Formulation of Peptide and<br />
Protein <strong>Drug</strong>s
www.transdermalmag.com March 2011 Volume 3 Number 2<br />
FoRMulATioN TEchNiquES<br />
DEPARTMENTS<br />
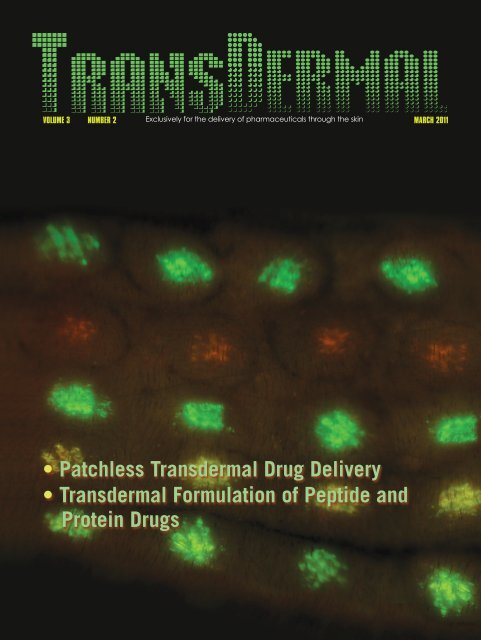
On the cover: Dermal electroporation using a<br />
minimally invasive device (MID-II) results in robust<br />
green fluorescent protein (GFP) and red fluorescent<br />
protein (RFP) expression following intradermal delivery<br />
of GFP and RFP reporter plasmids. This device offers a<br />
safe, tolerable, and efficient method to administer<br />
DNA vaccinations in a prophylactic setting and<br />
potentially represents an important new option for<br />
improved DNA-vaccine delivery in vivo. Courtesy of<br />
Inovio Pharmaceuticals, San Diego, California, USA.<br />
www.inovio.com<br />
<strong>Patchless</strong> <strong>Transdermal</strong> <strong>Drug</strong> <strong>Delivery</strong> (TDD) 5<br />
Bozena B Michniak-Kohn, Rutgers-The State University of New Jersey<br />
Kishore R Shah, Polytherapeutics, Inc<br />
On the <strong>Transdermal</strong> Formulation of Peptide and Protein <strong>Drug</strong>s 12<br />
Xiaorong Shen and David Jones<br />
Primera Analytical Solutions<br />
Industry news 2 Calendar 16<br />
Research news 3 People news C3<br />
Issue focus 5 Supplier news C3<br />
<strong>Transdermal</strong> (ISSN 1948-5824 online ISSN 1948-3600 print) is published six times a year in January, March, May, July,<br />
September, and November by CSC Publishing, Inc., 1155 Northland Drive, Saint Paul, MN 55120.
EdiTORiAl<br />
Peggy Wright<br />
Editor<br />
pwright@cscpub.com<br />
PROducTiOn And ART<br />
Maria novak<br />
Production Manager<br />
mnovak@cscpub.com<br />
harry Myers<br />
Assistant Production Manager<br />
hmyers@cscpub.com<br />
linda Rost<br />
Production Coordinator<br />
lrost@cscpub.com<br />
christopher Myers<br />
Prepress Manager<br />
cmyers@cscpub.com<br />
ciRculATiOn<br />
Aileen hough<br />
Circulation Manager<br />
ahough@cscpub.com<br />
sarah highum<br />
Assistant Circulation Manager<br />
shighum@cscpub.com<br />
Judie hadley<br />
Circulation Assistant<br />
jhadley@cscpub.com<br />
BusinEss<br />
Richard R. cress<br />
Publisher<br />
rcress@cscpub.com<br />
heather fitch<br />
Administrative Manager<br />
hfitch@cscpub.com<br />
Melinda cress<br />
Acctg/Oper Manager<br />
lcress@cscpub.com<br />
PhilAdElPhiA OfficE<br />
800 West State St., Suite 103<br />
Doylestown, PA 18901<br />
+1 215 340-6988<br />
FAX: +1 215 340-6989<br />
e-mail: segenolf@cscpub.com<br />
w w w . t r a n s d e r m a l m a g . c o m<br />
PuBlishing OfficE<br />
1155 Northland Drive<br />
St. Paul, MN 55120<br />
+1 651 287-5600<br />
FAX: +1 651 287-5650<br />
e-mail: transdermal@cscpub.com<br />
cindy fischer<br />
Acctg/Oper Administrator<br />
cfischer@cscpub.com<br />
Michelle R. Robinson<br />
Acctg/Mktg Coordinator<br />
mrobinson@cscpub.com<br />
liz dorry<br />
Web site Coordinator<br />
ldorry@cscpub.com<br />
Kelly Weyandt<br />
Web and Online Development<br />
kweyandt@cscpub.com<br />
AdvERTising<br />
Marybeth stewart<br />
West Coast & Mid-Atlantic Sales<br />
Manager<br />
mstewart@cscpub.com<br />
+1 541 318-4657<br />
Maggie Johnson<br />
Midwest & Southeastern Sales<br />
Manager<br />
mjohnson@cscpub.com<br />
+1 651 287-5600<br />
steve Egenolf<br />
Eastern Sales Manager<br />
segenolf@cscpub.com<br />
+1 215 340-6988<br />
Jan lee<br />
Midwest Sales Manager<br />
jlee@cscpub.com<br />
+1 513 583-9768<br />
Kevin clohesey<br />
Upstate New York, Canada,<br />
& Western Sales Manager<br />
kclohesey@cscpub.com<br />
+1 651 287-5600<br />
Bonnie Kaye<br />
National Accounts Manager<br />
bkaye@cscpub.com<br />
+1 651 213-1400<br />
cincinnATi OfficE<br />
887 Murle Lane<br />
Loveland, OH 45140<br />
+1 513 583-9768<br />
FAX: +1 513 583-9736<br />
e-mail: jlee@cscpub.com<br />
<strong>Transdermal</strong> <strong>•</strong> MARCH 2011<br />
Editorial<br />
Advisory Board<br />
Ajay K. Banga,<br />
Mercer University<br />
James Birchall,<br />
Cardiff University<br />
Joke Bouwstra,<br />
Leiden University<br />
Somesh Choudhury,<br />
Noven Pharmaceuticals, Inc.<br />
Lawrence A. Hill,<br />
Watson Pharmaceuticals<br />
Bozena Michniak-Kohn,<br />
Rutgers University and New<br />
Jersey Center for Biomaterials<br />
Reinhard Neubert,<br />
Martin-Luther-University (MLU)<br />
Halle-Wittenberg<br />
Danyi Quan,<br />
Xel Pharmaceuticals<br />
Michael S. Roberts,<br />
University of Queensland and<br />
University of South Australia<br />
Audra Stinchcomb,<br />
University of Kentucky<br />
Phillip Wertz,<br />
University of Iowa<br />
Thean Yeoh,<br />
Pfizer Inc.<br />
Technical<br />
Advisory Board<br />
Kishore R. Shah,<br />
Polytherapeutics, Inc.<br />
©2011 by CSC Publishing, Inc. All rights reserved.<br />
Published six times per year. No part of<br />
this publication may be reproduced or transmitted<br />
in any form or by any means without<br />
written permission from the publisher.<br />
<strong>Transdermal</strong> assumes no responsibility for statements<br />
or opinions of editorial contributors or<br />
advertisers.<br />
Go to www.transdermalmag.com to apply for<br />
your free subscription.<br />
1
2<br />
MARCH 2011 <strong>•</strong> TransDermal<br />
Industry news<br />
Eisai finalizes<br />
Japanese license<br />
agreement<br />
Tokyo, Japan — Eisai finalized an<br />
exclusive license agreement with<br />
Teikoku Seiyaku, which will re -<br />
search, develop, and market a<br />
new transdermal formulation of<br />
Aricept for treatment of Alz -<br />
heimer’s disease in Japan. Eisai<br />
will supply the active pharmaceutical<br />
ingredients for clinical and<br />
nonclinical use.<br />
Prosolus<br />
Pharmaceuticals plans<br />
to relocate<br />
Philadelphia, Pennsylvania,<br />
USA — According to the South<br />
Florida Business Journal, Prosolus<br />
Pharmaceuticals plans to relocate<br />
to Doral, Florida in Miami-Dade<br />
County. The company, only a year<br />
old, has licensed a transdermal<br />
system and wants to develop a<br />
5,000-square-foot manufacturing<br />
and product-development center<br />
there.<br />
Commercial launch of<br />
Prelude is on track<br />
Franklin, Massachusetts, USA —<br />
Echo Therapeutics’ partner, Ferndale<br />
Pharma Group, received<br />
comments from the US FDA on<br />
the 510(k) submission of the Prelude<br />
SkinPrep System that both<br />
companies deemed minor and<br />
consistent with expectations. Echo<br />
anticipates that the planned date<br />
for commercial launch of the product<br />
will remain on track, pending<br />
FDA clearance.<br />
Alcohol Monitoring<br />
Systems files patent suit<br />
Littleton, Colorado, USA — Alco-<br />
hol Monitoring Systems (AMS)<br />
has filed a lawsuit claiming that<br />
BI, Incorporated has infringed<br />
multiple patents with their TAD<br />
(<strong>Transdermal</strong> Alcohol Detection)<br />
system. AMS manufactures a<br />
trans dermal alcohol monitoring<br />
system, SCRAMx (Secure Continuous<br />
Remote Alcohol Monitor),<br />
which samples a subject’s perspiration<br />
for the purposes of monitoring<br />
alcohol consumption.<br />
Rutgers University<br />
launches center for<br />
dermal research<br />
Piscataway, New Jersey, USA —<br />
On March 3, 2011, the Center for<br />
Dermal Research at Rutgers University<br />
held its inaugural meeting.<br />
Dr Bozena Michniak-Kohn, PhD,<br />
leads the center. The university<br />
created the center to provide professional<br />
networking opportunities<br />
for its paid industrial members<br />
as well as opportunities for<br />
research collaborations with the<br />
Laboratory for <strong>Drug</strong> <strong>Delivery</strong>,<br />
which Michniak-Kohn heads.<br />
Skinvisible signs<br />
feasibility agreement<br />
Las Vegas, Nevada, USA — Skinvisible<br />
Pharmaceuticals signed an<br />
agreement with Novartis Pharma<br />
AG to assess the technical feasibility<br />
of a topical formulation of an<br />
undisclosed com pound using its<br />
Invisicare polymer delivery system.<br />
The agreement includes an<br />
option for the two companies to<br />
enter into a future, exclusive licen -<br />
sing agreement that would in -<br />
clude a licensing fee and royalties<br />
based on sales.<br />
Transdel<br />
Pharmaceuticals<br />
explores strategic<br />
alternatives<br />
La Jolla, California, USA —<br />
Transdel Pharmaceuticals is ex -<br />
ploring strategic alternatives<br />
with two investment banks,<br />
Amer ican Med Tech Advisors<br />
and ESC Advisors, a division of<br />
KEMA Partners. These alternatives<br />
could include, but are not<br />
limited to, partnering or other<br />
collaborative agreements, a<br />
merger, equity or debt financing,<br />
or sale of the company.<br />
Actavis voluntarily<br />
recalls transdermal<br />
Fentanyl<br />
Hafnarfirdi, Iceland — Actavis<br />
issued a voluntary recall of 18 lots<br />
of its 25 mcg Fentanyl patch after<br />
finding one defective lot that<br />
caused the medicine to release<br />
too quickly into the bloodstream.<br />
Apogee Technology<br />
resumes OTC trading<br />
Norwood, Massachusetts, USA<br />
— Apogee Technology resumed<br />
trading on the Over-the-Counter<br />
(OTC) board under the symbol<br />
ATCS.OB.<br />
NICE guidance<br />
recommends Exelon<br />
patch<br />
Basel, Switzerland — The Na -<br />
tional Institute for Health and<br />
Clinical Excellence (NICE) guidance<br />
TA1111 recommended<br />
Novar tis Pharmaceuticals UK’s<br />
Exelon transdermal patch as a<br />
cost-effective option for treatment<br />
of mild-to-moderate Alz -<br />
heimer’s disease.<br />
TransPharma changes<br />
name of transdermal<br />
system<br />
Lod, Israel — TransPharma Medical<br />
changed the name of its drug
delivery system from ViaDerm to<br />
ViaDor. The change occurred in<br />
part to break the association of<br />
TransPharma’s drug delivery system<br />
with other products carrying<br />
the name ViaDerm.<br />
BA Research acquires<br />
Hill Top Research<br />
Miamiville, Ohio, USA — BA<br />
Research, part of the Cliantha<br />
Group, acquired Hill Top Re -<br />
search. All current clinical sites in<br />
North America and India will continue<br />
in full operation going forward.<br />
Hill Top will continue as Hill<br />
Top Re search in North America.<br />
Echo Therapeutics<br />
receives $5.5M<br />
Franklin, Massachusetts, USA<br />
— Echo received $5.5 million in<br />
new financing in a deal that its<br />
majority shareholder, Platinum<br />
Montaur Life Sciences LLC, led.<br />
The company will use the funds<br />
to develop new products, move<br />
clinical studies forward, and draw<br />
upon for working capital and<br />
general corporate purposes. The<br />
agreement includes private stock<br />
placements, netting the company<br />
approximately $2.5 million, and a<br />
binding commitment for another<br />
$3 million.<br />
Research news<br />
FDA approves<br />
Fentanyl patch<br />
St. Louis, Missouri, USA — The<br />
US FDA approved Covidien’s<br />
ANDA for its Fentanyl <strong>Transdermal</strong><br />
System (FTS) patch. Its patch<br />
is a generic alternative to Johnson<br />
& Johnson’s branded Duragesic<br />
patch. FTS is an opioid analgesic<br />
indicated in opioid-tolerant pa -<br />
tients for management of persistent,<br />
moderate-to-severe chronic<br />
<strong>Transdermal</strong><br />
Contract Manufacturing<br />
cGMP<br />
FDA - <strong>Drug</strong>, Device,<br />
Food, Cosmetics<br />
EU Compliant<br />
Japanese Certified<br />
ISO 9001:2008<br />
ISO 13485:2003<br />
DEA Registered<br />
Schedule III – V<br />
Manufacturer of<br />
Controlled Substances<br />
SoluStrip soluble film converting<br />
for buccal transdermal delivery<br />
TransDermal <strong>•</strong> MARCH 2011<br />
Active and Passive transdermal<br />
converting<br />
�Finished patch or component<br />
�Reservoir and matrix<br />
Experience with ATDD technologies<br />
�Iontophoresis<br />
�Microneedles<br />
�Heat-activated<br />
�Dermabrasion<br />
Snapplicator unit dose<br />
packaging for transdermal<br />
gels & ointments<br />
Visit us at INTERPHEX Booth 1271<br />
Tapemark<br />
1-800-535-1998<br />
www.tapemark.com<br />
Write an article for<br />
<strong>Transdermal</strong><br />
Technical professionals write our<br />
articles for technical professionals.<br />
See technical article guidelines at<br />
www.transdermalmag.com for<br />
information on the types of articles<br />
we are looking for.<br />
Contact:<br />
Peggy Wright, Editor<br />
Tel. +1 651 698-2760<br />
E-mail: pwright@cscpub.com<br />
3
4<br />
MARCH 2011 <strong>•</strong> TransDermal<br />
pain. Covidien expects to launch<br />
the patch in the USA in 25mcg/<br />
hr, 50mcg/hr, 75mcg/hr, and<br />
100mcg/hr strengths.<br />
PLEASE–FSH patch provides<br />
first successful<br />
IVF pregnancy<br />
Ruggell, Liechtenstein — A<br />
woman conceived successfully<br />
after being implanted with a fertilized<br />
oocyte (egg cell) and re -<br />
ceiving treatment with Pantec<br />
Biosolutions’ FSH (follicle stimulating<br />
hormone) patch subsequent<br />
to skin microporation with<br />
the company’s proprietary<br />
P.L.E.A.S.E. (Painless Laser Epidermal<br />
System) device. The pregnancy<br />
is the first to occur using<br />
the technology.<br />
Inovio and University<br />
of Southampton begin<br />
vaccine study<br />
Blue Bell, Pennsylvania, USA<br />
and Southampton, UK — Inovio<br />
Pharmaceuticals obtained regulatory<br />
approval for a Phase 2 clinical<br />
trial that the University of<br />
Southampton will run. The study<br />
will use Inovio’s automated,<br />
ELGEN 1000 vaccine-delivery<br />
device and will evaluate a DNA<br />
vaccine that the university developed<br />
to treat chronic myeloid<br />
leukemia and acute myeloid<br />
leukemia. Leukaemia and Lymphoma<br />
Research (LLR) and the<br />
Efficacy and Mechanisms Evaluation<br />
(EME) program, which the<br />
UK Medical Research Council<br />
funds and the UK National Institute<br />
for Health Research manages,<br />
will provide financial support.<br />
Alvogen launches<br />
transdermal delivery<br />
system<br />
Parsippany, New Jersey, USA —<br />
Alvogen introduced an AB-1rated<br />
nitroglycerin transdermal<br />
delivery system. The hypoallergenic,<br />
transparent patches are<br />
available in 30-count cartons and<br />
four dosage strengths, including<br />
0.1, 0.2, 0.4, 0.6 mg/hr.<br />
BioSante receives<br />
orphan-drug status for<br />
vaccine<br />
Lincolnshire, Illinois, USA —<br />
The US FDA granted orphandrug<br />
status for BioSante Pharmaceuticals’<br />
potential skin-cancer<br />
vaccine. The company also has<br />
received orphan-drug status for<br />
other vaccines for pancreatic cancer,<br />
acute myeloid leukemia, and<br />
chronic myeloid leukemia.<br />
Purdue Pharma<br />
releases Butrans<br />
transdermal system<br />
Stamford, Connecticut, USA —<br />
Purdue Pharma’s Butrans (bupre -<br />
nor phine) <strong>Transdermal</strong> System<br />
CIII is now available by prescription.<br />
The drug is indicated for the<br />
management of moderate-tosevere<br />
chronic pain in patients<br />
requiring an around-the-clock<br />
opioid analgesic for an extended<br />
period of time. Butrans is the first<br />
opioid analgesic that continuously<br />
releases buprenorphine for<br />
seven days.<br />
Alliqua completes<br />
study for pain patch<br />
New York, New York, USA —<br />
Alliqua, formerly HepaLife Technologies,<br />
successfully completed<br />
an initial study for its transdermal<br />
pain patch. In the comparative<br />
dissolution study, its profile<br />
was favorable when compared to<br />
the current leading product for<br />
treating postherpetic neuralgia<br />
(PHN) pain. Alliqua now will<br />
start the next developmental<br />
stage, including in-vitro testing.<br />
Foamix starts Phase 2<br />
clinical trial<br />
Rehovot, Israel — Foamix en -<br />
rolled the first patient in a Phase<br />
2 trial of its Topical Minocycline<br />
Foam. The study aims to demonstrate<br />
efficacy and safety in<br />
patients suffering from Rosacea.<br />
Skin-penetration studies demonstrated<br />
a high occurrence of the<br />
drug in the viable layers of the<br />
skin and also revealed that the<br />
drug is not delivered transdermally,<br />
indicating that its topical<br />
application will not involve any<br />
systemic adverse effects.<br />
FDA accepts Zelrix<br />
NDA for filing<br />
Conshohocken, Pennsylvania,<br />
USA — NuPathe received notice<br />
that the US FDA accepted the<br />
company‘s Zelrix NDA, submitted<br />
on October 29, 2010, for filing.<br />
The company expects the FDA to<br />
complete its review of the NDA<br />
by August 29, 2011. Zelrix is the<br />
first-ever submission to the FDA<br />
of a transdermal patch for the<br />
treatment of migraine.<br />
FDA approves Fortesta<br />
gel<br />
Newark, Delaware, USA — The<br />
US FDA approved Endo Pharmaceuticals’<br />
Fortesta gel for treatment<br />
of low testosterone, also<br />
known as hypogonadism. The<br />
gel comes in a metered-dose<br />
pump that delivers the correct<br />
dose per complete depression.<br />
Endo ex pects to introduce the gel<br />
in the USA in early 2011.
TransDermal <strong>•</strong> MARCH 2011<br />
<strong>Patchless</strong> <strong>Transdermal</strong> <strong>Drug</strong> <strong>Delivery</strong> (TDD)<br />
Bozena B Michniak-Kohn, Rutgers-The State University of New Jersey<br />
Kishore R Shah, Polytherapeutics, Inc<br />
We can trace the advent of transdermal<br />
drug delivery to the FDA’s approval and<br />
commercialization of scopolamine (1979)<br />
and nitroglycerin (1981) patches. Since then, companies<br />
have marketed many transdermal products<br />
worldwide, and transdermals have become a multibillion-dollar<br />
industry. Some of the advantages of<br />
transdermal drug delivery include:<br />
1. A reproducible and prolonged drug-delivery rate<br />
2. Elimination of hepatic first-pass metabolism<br />
3. Minimization of undesirable side effects<br />
4. Increased convenience for and compliance by<br />
patients<br />
5. Rapid termination of drug therapy when desired<br />
Despite these advantages, the barrier function of the<br />
skin, and its irritation and sensitization by many<br />
drugs and certain patch components, provide the<br />
principal impediments to a wider use of transdermal<br />
drug delivery. To some extent, patchless TDD in<br />
the form of topically applied gels, creams, lotions, or<br />
solutions can overcome the limitations of the skin<br />
barrier by application of the drug formulation to a<br />
much larger area of skin than is possible with plastic<br />
patches.<br />
Formulators can use patchless TDD to elicit a therapeutic<br />
effect within tissues near the site of product<br />
application and/or to distribute the drug throughout<br />
the body using the circulatory system. <strong>Patchless</strong><br />
delivery may be suitable for treatment of various<br />
indications, such as pain, hormone replacement therapy,<br />
contraception, overactive bladder, CNS disorders,<br />
male or female sexual dysfunction, and smoking<br />
cessation.<br />
Key advantages of patchless transdermal products<br />
include:<br />
<strong>•</strong> The potential for expanding the scope of trans-<br />
dermal delivery to drugs requiring significantly<br />
higher dosages than possible with patches<br />
<strong>•</strong> Flexibility of dosing<br />
<strong>•</strong> Reduction in skin irritation due to lack of<br />
occlusion<br />
<strong>•</strong> Lack of irritation from patch components such as<br />
adhesives<br />
<strong>•</strong> Ease of manufacture<br />
<strong>•</strong> Cost effectiveness<br />
<strong>•</strong> Cosmetic elegance<br />
The deficiencies of the patchless TDD in contrast to<br />
patches may include:<br />
<strong>•</strong> Some variability in precision of dosing<br />
<strong>•</strong> Lack of suitability for drugs having a high skinpermeation<br />
rate, such as fentanyl and clonidine<br />
<strong>•</strong> A narrow therapeutic window<br />
<strong>•</strong> Lack of retention of the drug and enhancer at the<br />
site of application<br />
Formulation Considerations<br />
Dosage forms for patchless TDD can be clear solutions,<br />
gels, or emulsions. The formulator selects the<br />
form with an emphasis on its spreadability on skin,<br />
drying time, cosmetic feel, and acceptability to users.<br />
A mixture of water and alcohol is often a good<br />
medium for the preparation of a solution or gel. A<br />
lower alkyl alcohol, such as ethanol or isopropanol,<br />
helps to solubilize the drug. Polymeric thickeners<br />
can adjust the viscosity of the solution for optimum<br />
handling and spreadability. An emulsion (oil-inwater<br />
or water-in-oil) is a useful medium for drugs<br />
that are hydrophobic and poorly soluble in hydroalcoholic<br />
medium. In an emulsion, the drug is solubilized<br />
in the oil phase. Key ingredients for the preparation<br />
of such emulsions include oils, waxes,<br />
emulsifiers, and emollients.<br />
5
6<br />
MARCH 2011 <strong>•</strong> TransDermal<br />
Excipients for TDD formulations should be nontoxic,<br />
nonirritating, and nonsensitizing to skin, even upon<br />
repeated application to the same site. The transdermal<br />
composition may use different excipients<br />
depending on the dosage form. Excipients may<br />
include solvents for the drug, thickeners, preservatives,<br />
anti-oxidants, chelating agents, fragrances, etc.<br />
A key ingredient for a patchless TDD formulation is<br />
the skin-permeation enhancer, which facilitates<br />
delivery of a therapeutically required dosage of the<br />
drug. Some of the common enhancers include oleic<br />
acid, dimethyl isosorbide, lauryl lactate, dimethyl<br />
sulfoxide, decyl methyl sulfoxide, ethoxydiglycol,<br />
N-methyl 2-pyrrolidone, and isopropyl myristate. A<br />
review by Danyi Quan, et al provides a detailed discussion<br />
of enhancers for TDD [1, 2]. Many enhancers<br />
cause skin irritation when used at higher concentrations.<br />
Therefore, a balance between drug-permeation<br />
enhancement and skin safety is important in optimizing<br />
the formulation.<br />
One of the key issues with patchless TDDs is retention<br />
of the drug and enhancer at the site of application.<br />
Lack of retention for the desired duration of<br />
time is a common problem. Nature designed the<br />
skin to shed materials adhering to it. Loss of an<br />
applied dose also can occur by transfer to other<br />
objects, such as articles of clothing, or other people.<br />
The loss results in an inadequate therapeutic effect<br />
and a shorter duration of pharmacological activity<br />
for the drug. In addition, transfer of a drug, such as<br />
testosterone, to others by touch is a particularly sensitive<br />
issue pertaining to safety of the product.<br />
An appropriate film-forming polymeric delivery system,<br />
which can serve as a matrix for the drug and<br />
enhancers, can address some of these issues; however,<br />
the use of a polymer delivery system for TDD has its<br />
own challenges. Skin is a living substrate; that is, it<br />
breathes, perspires, stretches, and contracts. The<br />
formed film must accommodate the various functions<br />
of the skin during different activities and environmental<br />
conditions, such as temperature and humidity.<br />
It must be bioadherent and possess viscoelastic properties,<br />
breathability, and moisture-vapor permeability<br />
similar to skin. In the absence of such characteristics,<br />
the film tends to feel uncomfortable or tight and peels<br />
and flakes during use. The net result is inadequate<br />
wear time and loss of efficacy.<br />
Another consideration is possible crystallization of<br />
the drug on the skin when the applied formulation<br />
dries. Other critical issues that formulators need to<br />
address in patchless TDD development include the<br />
hydrolytic and oxidative stability of the drug in<br />
aqueous or hydroalcoholic medium, pH effects, and<br />
cosmetic acceptability.<br />
Commercial Approaches to <strong>Patchless</strong><br />
TDD Products<br />
Acrux, Vivus, Ther-Rx<br />
Acrux (www.acrux.com.au) initially acquired its<br />
technology — discovered by B Finnin, B Reid, and<br />
T Morgan — from Monash University in Australia.<br />
Sunscreens have used its ACROSS ® enhancer octisalate<br />
for a long time at higher doses. It is a known,<br />
safe, nonirritating formulation and allows the passage<br />
of drugs across the barrier layer of the skin.<br />
Acrux has placed the ACROSS enhancer in a<br />
Metered Dose <strong>Transdermal</strong> System ® (MDTS), allowing<br />
application of a pre-set dose of a drug. The system<br />
also creates a patchless reservoir in the skin that<br />
can deliver drugs for various indications for human<br />
and veterinary use. Vivus (www.vivus.com) is the<br />
licensee in the USA for Acrux’s testosterone spray<br />
for women, trademarked as Luramist ® . Another<br />
ACROSS product, Evamist ® , is a 1.7% estradiol topical<br />
spray for systemic transdermal delivery of the<br />
hormone (Ther-Rx, www.ther-rx.com).<br />
Biosante, Antares Pharma, Azur Pharma<br />
Biosante Pharmaceuticals (www.biosantepharma<br />
.com) is a company specializing in products for<br />
female sexual-health and oncology. The lead is<br />
LibiGel ® , a transdermal testosterone gel for treatment<br />
of female sexual dysfunction. Antares Pharma<br />
(www.antarespharma.com), Biosante’s partner, is<br />
developing it. It is in Phase 3 clinical development<br />
under an FDA Special Protocol Assessment. Biosante<br />
intends to submit the LibiGel NDA in 2011 for potential<br />
approval by the FDA in 2012.<br />
Azur Pharma (www.azurpharma.com) in Ireland is<br />
a private pharmaceutical company focused on the<br />
Central Nervous System (CNS) and the health of<br />
women. Azur is marketing Elestrin ® , which they<br />
acquired from Biosante, in the USA. It is an estradiol<br />
gel for the treatment of menopausal vasomotor<br />
symptoms.
Novavax, Abbot, Auxilium<br />
Researchers initially developed micellar nanoparticle<br />
technology in the mid-1990s. Novavax (www.nova<br />
vax.com ) further developed, patented, and marketed<br />
the first product using this approach, Estrasorb ® , a<br />
transdermal lotion launched in 2003 [3]. This technique<br />
uses a multiphasic nanoemulsion consisting of<br />
one or two active ingredients, a solvent (often<br />
ethanol), a stabilizer (often a nonionic surfactant), oil,<br />
and an aqueous medium. These emulsions can incorporate<br />
both water-soluble and poorly water-soluble<br />
drugs and can accommodate both crystalline and<br />
amorphous drugs. Formulators have achieved drug<br />
loading of up to 20% (w/w). These lotions behave like<br />
patchless patches and are highly stable formulations;<br />
for example, Estrasorb has a shelf life of three years<br />
at room temperature.<br />
Androsorb ® is a topical testosterone formulation<br />
based on micellar nanoparticle technology<br />
(Novavax, Inc.).<br />
Other topical testosterone products include Androgel<br />
® (Abbott, www.abbott.com), which is a regular<br />
or metered-dose formulation [4], and Testim ® , a<br />
hydroalcoholic gel (Auxilium Pharmaceuticals,<br />
www.auxilium.com). These products do not use<br />
micellar nanoparticle technology.<br />
Antares Pharma<br />
In early 2010, Antares Pharma and the Population<br />
Council announced very promising outcomes from<br />
a dose-finding, Phase 2 clinical trial for a contraceptive<br />
gel that contains the new, synthetic progestin<br />
nestorone and estradiol formulated in Antares<br />
Pharma’s Advanced <strong>Transdermal</strong> <strong>Delivery</strong> (ATD)<br />
gel system [5, 6]. They conducted the open-label,<br />
crossover study on 18 women at three sites, evaluating<br />
the effect of the gel on ovulation suppression in<br />
normal women of fertile age. The ATD gel is a clear<br />
formulation that dries quickly on the skin and leaves<br />
no residue. The technology uses a combination of<br />
permeation enhancers to achieve sustained transdermal<br />
drug delivery.<br />
Antares Pharma also developed a transdermal, oxybutynin<br />
gel formulation, its Anturol ® gel, delivered<br />
using a metered dose pump [5]. The drug has<br />
antimuscarinic and antispasmodic effects on smooth<br />
muscle and treats incontinence in overactive bladders.<br />
Two dose strengths are under investigation, 56<br />
TransDermal <strong>•</strong> MARCH 2011<br />
and 84 mg, and the pump allows dose titration.<br />
Studies have indicated that approximately 16% of<br />
adults are affected by an overactive bladder, and it<br />
is particularly a problem in women and the elderly.<br />
Kennelly, who tested the ATD, reported that transdermal<br />
delivery of oxybutynin (gel) significantly<br />
improved cognitive tolerability when compared to<br />
an orally administered drug [7].<br />
BioChemics, Vaso Active Pharmaceuticals<br />
BioChemics’ technologies (www.biochemics.com)<br />
include VALE ® (Vaso-Active Lipid Encapsulated)<br />
and PENtoCORE ® (intradermal delivery), which<br />
Vaso Active Pharmaceuticals (www.vasoactive.us)<br />
commercialized [8, 9]. Patients use VALE as a patchless<br />
cream or gel with dose control for targeted and<br />
systemic delivery of small molecules. When applied<br />
to the surface of skin, the formulation vanishes in<br />
about one minute. An example is Osteon ® , which<br />
delivers menthol (analgesic) for the temporary relief<br />
of minor aches and pains of muscles and joints associated<br />
with arthritis, sprains, strains, and simple<br />
back pain [10]. The inactive ingredients of the gel are<br />
deionized water, isopropanol, olive oil, propylene<br />
glycol, eucalyptus oil, sodium alginate, cetyl alcohol,<br />
glyceryl stearate, kelp extract, marshmallow extract,<br />
allantoin, and xanthan gum. AR Extreme ® Pain<br />
Relieving Lotion provides temporary relief from<br />
pain from minor sports injuries, and TERMIN8 ®<br />
treats athlete’s foot and provides relief for burning,<br />
cracked, and itchy feet. Both of these formulations<br />
use the PENtoCORE technology.<br />
Mika Pharma<br />
Established in 1994 for development of products for<br />
the dermal and buccal markets, in 1995 MIKA<br />
Pharma (www.mika-pharma.de) was the first company<br />
worldwide to introduce a stable, topical liposomal<br />
product (liposomal MIKA heparin spray).<br />
Brunner et al reported on a MIKA spray gel formulation<br />
with diclofenac, comparing it to oral Voltaren<br />
in 12 healthy males [11]. The spray gel consisted of<br />
water, isopropyl alcohol, and propylene glycol as<br />
basic solvents and soy bean lecithin, ethanol, disodium<br />
phosphate dodecahydrate, sodium dihydrogen<br />
phosphate dehydrate, peppermint oil, and<br />
ascorbyl palmitate as excipients. Patients sprayed it<br />
on the skin, and the alcoholic components slowly<br />
evaporated, leaving behind a low viscous gel that<br />
stuck to the skin. Using microdialysis to evaluate<br />
7
8<br />
MARCH 2011 <strong>•</strong> TransDermal<br />
pharmacokinetic parameters, the authors concluded<br />
that the MIKA formulation exhibited low systemic<br />
availability and produced effective diclofenac concentrations<br />
equivalent to or better than the oral<br />
dosage form. Other researchers conducted studies<br />
using the MIKA topical liposomal technology: a) a<br />
heparin spray used to treat superficial venous<br />
thrombosis [12, 13] and b) topical delivery of plasmid<br />
DNA to skin [14].<br />
NexACT, MacroChem<br />
NexMed, a specialty CRO with a pipeline of products<br />
based on the NexACT ® technology, recently<br />
changed its name (September 2010) to Apricus Biosciences<br />
(www.apricusbio.com) and moved from<br />
New Jersey to California in the USA [15, 16]. Its technology<br />
is based on permeation enhancers that affect<br />
the fluidization of lipids in cellular membranes. An<br />
example of one of the compounds is dodecyl 2-(N,<br />
N-dimethylamino)-propionate. Formulators can use<br />
this technology topically, but as a recent preclinical<br />
study showed, also can deliver insulin and other<br />
drugs with larger molecular weights, such as Taxol,<br />
subcutaneously in a depot-like fashion (slow re -<br />
lease). <strong>Delivery</strong> occurs over 24 hours from a singledose<br />
formulation. Recent investigations by Michniak-Kohn’s<br />
lab at Rutgers University have shown<br />
the potential use of this technology in transbuccal<br />
delivery [17].<br />
MacroChem, which Access Pharmaceuticals<br />
acquired (www.accesspharma.com), developed<br />
SEPA (Soft Enhancer of Percutaneous Absorption)<br />
based on dioxolane chemistry, which they have<br />
shown improves drug permeation through the skin.<br />
Diani et al published an example of the enhancement<br />
of activity, showing that 2-n-nonyl-1, 3-dioxolane<br />
augments hair-growth effects of topically<br />
applied minoxidil in the balding stump-tail-macaque<br />
model [18].<br />
Watson Pharmaceuticals<br />
Watson Pharmaceuticals (www.watson.com) markets<br />
Gelnique ® (oxybutynin chloride) gel 10%, which<br />
is indicated for overactive bladder. It is formulated in<br />
an alcohol-based gel containing alcohol, glycerin,<br />
hydroxypropylcellulose, sodium hydroxide and<br />
purified water. The patient receives the formulation<br />
in a sachet that must be used immediately upon<br />
opening due to evaporation of the alcohol.<br />
BHR Pharma<br />
BHR Pharma (www.bhr-pharma.com) has developed<br />
a transdermal estradiol gel for the palliative treatment<br />
of vasomotor side effects in men undergoing androgen-deprivation<br />
therapy (ADT) for pros tate cancer<br />
and advanced androgen-sensitive carcinoma of the<br />
prostate. Located in Herndon, Virginia, USA, BHR<br />
Pharma is a wholly owned subsidiary of Besins<br />
Healthcare (www.besins-healthcare.com), a Belgian<br />
company, and an affiliate of Ascend Therapeutics<br />
(www.ascendtherapeutics.com), Besins’ pharmaceutical<br />
sales and marketing entity in the USA.<br />
BHR’s technology involves the Enhanced Hydroalcoholic<br />
Gel ® (EHG) that Besins pioneered and commercialized<br />
in 1975. Other products include Estrogel<br />
® (www.estrogel.com), which is a clear and<br />
colorless topical gel containing estradiol 0.06% for<br />
treatment of menopausal symptoms and which is<br />
available in a dosing pump or tube, and Progestogel<br />
® , a 1% progesterone hydroalcoholic gel used to<br />
treat breast pain and tenderness.<br />
Transdel Pharma<br />
Ketotransdel ® is a novel cream containing 10% ketoprofen,<br />
recommended for acute soft tissue injuries<br />
(sprains and strains). A 2010 Phase 3 study in 364<br />
patients suggested that Ketotransdel is effective over<br />
placebo and did not produce any severe side effects.<br />
Researchers detected only minimal levels of ketoprofen<br />
in the blood of the volunteers, supporting the<br />
fact that low concentrations were absorbed systemically<br />
[19]. Transdel (ww.transdelpharma.com)<br />
focuses primarily on topical drug delivery, and the<br />
Transdel technology facilitates the dissolution of the<br />
API and enhances the passage of the drug across the<br />
barrier of the skin.<br />
Novartis, Endo Pharmaceuticals<br />
Voltaren ® is a diclofenac sodium gel that contains<br />
10mg/g of drug with the following excipients:<br />
ammonia, carbomer homopolymer Type C, isopropyl<br />
alcohol, mineral oil, polyoxyl 20 cetostearyl<br />
ether, propylene glycol, and water [20]. Endo Pharmaceuticals<br />
(www.endo.com) markets Novartis<br />
Consumer Health’s product for relief of pain caused<br />
by osteoarthritis of joints, such as knees and hands<br />
(www.novartis.com). Patients apply the gel using a<br />
dosing card that is supplied with the medication to<br />
ensure the correct dose of diclofenac.
Nuvo Research, Covidien, King<br />
Pharmaceuticals<br />
Pennsaid ® contains diclofenac sodium 1.5 % w/w as<br />
a topical solution with glycerin, propylene glycol,<br />
ethanol, purified water, and a skin-penetration<br />
enhancer, dimethyl sulfoxide [21, 22]. Nuvo Research<br />
(www.nuvoresearch.com), formerly Dimethaid, manufactures<br />
the product, and Covidien (www.covidien.com)<br />
markets it in the USA under license from<br />
Nuvo. In November 2009, the FDA approved Pennsaid<br />
in the USA (originally launched in Canada),<br />
where it is one of three products for osteoarthritis. The<br />
other two are Voltaren ® gel (discussed previously) and<br />
the Flector ® patch, which contains 1.3% diclofenac<br />
sodium and is a product of King Pharmaceuticals<br />
(www.kingpharm.com).<br />
Esba Laboratories<br />
Lidocaine hydrochloride topical gels are available<br />
from various manufacturers. An example is Topicaine<br />
® from Esba Laboratories (www.esbalabs.com)<br />
[23]. This formulation contains 40 mg of lidocaine in<br />
a gel composed of water, ethanol (35% w/w), glycerin,<br />
jojoba oil, aloe vera oil, glyceryl monolaurate,<br />
benzyl alcohol, carbomer 940, and EDTA-ethylenediaminetetraacetic<br />
acid. Patients apply it topically<br />
for relief of pain, and it provides analgesia in the skin<br />
by reversibly blocking the flux of sodium ions<br />
through the neuronal membranes.<br />
AlphaRx<br />
A Canadian company, AlphaRx (www.alpharx.com),<br />
launched its Colloidal Lipid Dispersion (CLD) technology,<br />
which provides percutaneous enhancement<br />
of topically applied drugs. In addition, formulators<br />
can combine this product with polymers and adhesives<br />
for patch development and transdermal drug<br />
delivery [24]. The Flexogan BCD ® (Bioadhesive Colloidal<br />
Dispersion) series of products marketed in<br />
Canada uses this technology for arthritis and muscle<br />
and joint pain. The formulation consists of highly dispersed<br />
oil droplets encapsulating natural menthol<br />
and camphor. The droplet size is less than one micron,<br />
and the formulation is nongreasy and allows a much<br />
higher availability of the active ingredient.<br />
Embil Pharmaceuticals<br />
Embil Pharmaceuticals in Turkey (www.embil.com)<br />
has patented a novel formulation, the Sulidin ® nime-<br />
TransDermal <strong>•</strong> MARCH 2011<br />
sulide LPS Gel [25]. The drug is indicated for osteo -<br />
arthritis, rheumatoid arthritis, periarthritis, and posttraumatic<br />
or acute musculoskeletal disorders. The<br />
LPS (Lamellar Penetration System) is based on the<br />
idea of using components of the skin structure as the<br />
delivery system. The lamellar hydrophobic structure<br />
both protects and facilitates the delivery of the API<br />
into the skin.<br />
Polytherapeutics<br />
PharmaDur ® (www.pharmadur.com) is a graft<br />
copolymer delivery system designed to address<br />
issues with polymeric delivery systems discussed<br />
above under “Formulation Considerations.” It has<br />
been commercially available over the last few years<br />
under that trade name. Kishore Shah first reported<br />
on PharmaDur and related graft copolymers for<br />
mucosal and skin applications [26]. PharmaDur<br />
polymer combines hydrophilic and hydrophobic<br />
structural moieties (Figure 1).<br />
The copolymer exhibits a combination of bioadhesive<br />
and controlled-release properties because of its<br />
unique structural characteristics [27]. When a patient<br />
applies a dermatological vehicle, such as a cream,<br />
lotion, or gel formulated with the PharmaDur polymer,<br />
the vehicle forms an imperceptible and invisible<br />
hydrogel film, a virtual patch. The PharmaDur polymer<br />
film is nontacky and nongreasy, has a very<br />
smooth feel, does not exhibit flaking or skin stretchiness,<br />
and is breathable. It is characterized by excellent<br />
bioadhesion to skin and exhibits controlled<br />
release of both hydrophobic and hydrophilic molecules.<br />
Because PharmaDur polymer is very hydro -<br />
Figure 1<br />
PharmaDur Graft Copolymer<br />
R – alkyl radical<br />
n, m, x, y, & z – positive integers having unspecified different<br />
large values<br />
9
10<br />
MARCH 2011 <strong>•</strong> TransDermal<br />
philic, it also helps to moisturize the skin, an added<br />
benefit.<br />
PharmaDur film on the skin serves as a matrix for<br />
the drug and other nonvolatile excipients for the<br />
product formulation. The skin retains the polymer<br />
film and the drug contained therein for 24+ hours,<br />
and the film provides continuous release of the drug<br />
by a process of diffusion for transdermal delivery to<br />
the body (Figure 2). The formed polymer film is not<br />
removed by perspiration or articles of clothing or<br />
transferred by touch. Thus, it functions as a virtual<br />
patch without the limitations of plastic patches.<br />
Formation of an invisible hydrogel patch for time<br />
release of medications is an attractive concept for the<br />
Figure 2<br />
Schematic Diagram of PharmaDur Virtual<br />
Patch on Skin<br />
Figure 3<br />
Comparison of Flux of Caffeine with Oleic Acid<br />
Formulations (controls) and with Added 2%<br />
PharmaDur Polymer<br />
consumer. A cosmetically elegant and invisible virtual<br />
patch is a unique concept for transdermal delivery<br />
of drugs, such as nonsteroidal, anti-inflammatory<br />
drugs (NSAIDs) for systemic activity. The<br />
virtual patch provides the following key advantages:<br />
<strong>•</strong> Delivers drugs requiring a high dosage or those<br />
with low skin permeability effectively since<br />
patients can apply the product over a large area of<br />
skin (~ 300 to 500 cm2).<br />
<strong>•</strong> Provides all-day, sustained therapeutic efficacy<br />
with a once-a-day application due to its long-lasting<br />
retention on skin, which also improves convenience<br />
for and compliance by consumers.<br />
<strong>•</strong> Reduces undesirable side effects due to avoidance<br />
of peaks and valleys in systemic drug concentrations<br />
as are typically found with oral drug<br />
administration.<br />
<strong>•</strong> Delivers many drugs that are not suitable for<br />
transdermal administration using conventional<br />
plastic patches and semisolid dosage forms.<br />
Furthermore, our studies have shown that the Pharma<br />
Dur polymer acts synergistically with skin-penetration<br />
enhancers in maximizing the rate of transdermal<br />
drug delivery. For example, Michniak et al<br />
(28) previously reported the effect of PharmaDur<br />
polymer on transdermal permeation of caffeine in<br />
the presence of the enhancer oleic acid. The re -<br />
searchers studied the skin-permeation rate of caffeine<br />
with and without the PharmaDur polymer,<br />
using human cadaver skin and Franz diffusion cells.<br />
The presence of the PharmaDur polymer almost<br />
doubled the skin-permeation rate of caffeine compared<br />
to that of the control formulation consisting of<br />
caffeine and oleic acid (Figure 3).<br />
The researchers postulate that the PharmaDur polymer<br />
film provides controlled release of both caffeine<br />
and the enhancer, thereby maximizing the enhancement<br />
in the caffeine permeation rate.<br />
Conclusion<br />
<strong>Patchless</strong> TDD is emerging as a convenient and<br />
patient-friendly means for extending the scope of<br />
transdermal delivery of drugs. Several new products<br />
and technologies using this approach for eliciting<br />
both local and systemic therapeutic effects have<br />
emerged in the last decade.
References<br />
1. Quan D, Hui X, and Maibach, H. Chemical enhancement<br />
of transdermal drug delivery systems-Part I. <strong>Transdermal</strong>. March<br />
2010, 2(2):5-10.<br />
2. Quan D, Hui X, and Maibach H. Chemical enhancement<br />
of transdermal drug delivery systems-Part II. <strong>Transdermal</strong>. March<br />
2010, (2(3):15-20.<br />
3. Lee, RW, Shenoy DB, Sheel R. Micellar nanoparticles:<br />
Applications for topical and passive transdermal drug delivery.<br />
Chapter 2 in Handbook of Non-invasive <strong>Drug</strong> <strong>Delivery</strong>. Toronto,<br />
Canada: Elsevier, 2010, pp 37- 58.<br />
4. Androgel information at<br />
www.drugs.com/androgel.html, accessed 1/19/2011.<br />
5. Information on Anturol gel at<br />
antarespharma.com/pipeline/anturol-gel, accessed 1/19/2011.<br />
6. Contraceptive gel study, www.popcouncil.org/mediacenter/newsreleases/2010_antarespharma/,<br />
accessed 12/03/2010.<br />
7. Kennelly, MJ. A comparative review of oxybutynin chloride<br />
formulations: Pharmacokinetics and therapeutic efficacy in<br />
overactive bladder. Reviews in Urology. 2010 Winter, 12(1):12-19.<br />
8. Biochemics’ company information,<br />
www.biochemics.com, accessed 1/19/2011.<br />
9. Vasoactive product information, www. vasoactive.us,<br />
accessed 1/19/2011.<br />
10. Osteon lotion information, www.osteonlotion.com,<br />
accessed 1/19/2011.<br />
11. Brunner M, Dehghanyar P, Seigfried B, Martin W, Menke<br />
G, Muller M. Favourable dermal penetration of diclofenac after<br />
administration to the skin using a novel spray formulation. British J<br />
Clin Pharmacol. 2005 Nov, 60(5):573-577.<br />
12. Gorski G, Szopinski P, Michalak J et al. Liposomal<br />
heparin spray: A new formula in adjunctive treatment of superficial<br />
venous thrombosis. Angiology. 2005 Jan-Feb, 56, 9-17.<br />
13. Katzenschlager R, Ugurluoglu A, Minar E, Hirschi M.<br />
Liposomal heparin-spraygel in comparison with subcutaneous low<br />
molecular weight heparin in patients with superficial venous<br />
thrombosis: A randomized, controlled, open multicenter study. J<br />
Kardiol. 2003, 10(9): 375-378.<br />
14. Meykadeh N, Mirmohammadsadegh A, Wang Z, Basner-<br />
Tschakarjan E, Hengge UR. Topical application of plasmid DNA to<br />
mouse and human skin. J Mol Med, 2005 Nov, 83(11): 897-903.<br />
15. NexACT technology, www.newsmedical.net/news/20100318/NexACT-technology-demonstratesability-to-deliver-Taxol-subcutaneously,<br />
accessed 12/03/2010.<br />
16. NexMed’s company information,<br />
www.nexmed.com/nexact.html, accessed 12/03/2010.<br />
17. Hu L, Damaj BB, Martin R, Michniak-Kohn BB. Enhanced<br />
in vitro transbuccal delivery of ondansetron HCl. Int J Pharm. Epub,<br />
2010 November 5, doi:10.1016/j.ijpharm.2010.10.052.<br />
18. Diani AR, Shull KL, Zaya MJ, Brunden MN. The penetration<br />
enhancer SEPA augments stimulation of scalp hair growth by<br />
topical minoxidil in the balding stumptail macaque. Skin<br />
Pharmacol. 1995, 8(5):221-228.<br />
19. Ekman EF, Strepnik N, Lawson, Schupp JP. Efficacy and<br />
safety of ketoprofen 10% cream in the treatment of pain associated<br />
with acute soft tissue injuries (Phase 3 Study TDLP-110-001). 13th<br />
TransDermal <strong>•</strong> MARCH 2011<br />
World Congress on Pain, Montreal, Canada, Aug. 29-Sept. 2, 2010,<br />
Poster # PH 420.<br />
20. Voltaren gel information, www.drugs.com/pro/voltarengel.html,<br />
accessed 12/03/2010.<br />
21. Pennsaid information, www.progressive-medical.com,<br />
accessed 12/04/2010.<br />
22. Pennsaid information,<br />
www.medisave.ca/<strong>Drug</strong>MoreInfo2391.aspx, accessed 12/04/2010.<br />
23. Esba Laboratories’ information,<br />
www.esbalabs.com/medical_information.htm, accessed<br />
12/04/2010.<br />
24. Colloidal lipid dispersion system, www.alpharx.com,<br />
accessed 12/03/2010.<br />
25. Information on Sulidin nimesulide LPS non-steroidal topical<br />
anti-inflammatory gel at http://pharmalicensing.com/public/<br />
outlicensing/view/1160/+, accessed 01/19/2011.<br />
26. Shah K. Mucoadhesive compositions for administration<br />
of biologically active agents to animal tissue. U.S. Patent 5,942,243<br />
(24 August 1999).<br />
27. Shah K, PharmaDur Bioadhesive <strong>Delivery</strong> System. In<br />
Skin <strong>Delivery</strong> Systems; <strong>Transdermal</strong>s, Dermatologicals, and<br />
Cosmetic Actives. John J Wille, (ed). Ames, Iowa: Blackwell<br />
Publishing, 2006, pp 211-222.<br />
28. Michniak B, Thakur R, and Shah K. Evaluation of novel<br />
polymer for transdermal permeation of caffeine in presence of different<br />
concentrations of chemical enhancer. Poster # 323,<br />
Proceedings of Controlled Release Society 32nd Annual Meeting,<br />
Miami Beach, Florida, June 2005.<br />
Dr Bozena Michniak-Kohn is a professor in pharmaceutics at the Ernest<br />
Mario School of Pharmacy, Rutgers-The State University of New Jersey in<br />
Piscataway, New Jersey, USA and is the director of the Center for Dermal<br />
Research and Laboratory for <strong>Drug</strong> <strong>Delivery</strong> of the New Jersey Center for<br />
Biomaterials (NJCBM). Her research focuses on topical, buccal, and transdermal<br />
drug delivery. Michniak-Kohn has directed over 26 PhD students,<br />
6 Master’s students, and 200+ undergraduates, and the work resulted in<br />
over 350 abstracts, two books, and more than 100 papers and book chapters.<br />
She is a Fellow of AAPS and is a reviewer for over a dozen pharmaceutical<br />
and drug delivery journals.<br />
Dr Kishore R Shah is president of Polytherapeutics, a research and development<br />
company focused on topical mucosal and transdermal drug delivery.<br />
He received his doctoral degree in organic chemistry from the<br />
University of Maryland in College Park, Maryland, USA. He has over 25<br />
years of research experience in the medical device and pharmaceutical<br />
industry, including at Bristol-Myers Squibb and Johnson & Johnson. His<br />
technical expertise includes development of polymeric biomaterials, their<br />
applications, and controlled drug delivery technologies. Shah possesses<br />
experience in taking products from concept to commercialization. He is an<br />
author or co-author of more than 45 patents and technical publications,<br />
several of which are in the field of transdermal drug delivery.<br />
Contact Dr Shah at Polytherapeutics, Inc, 568 Cabot Hill Road,<br />
Bridgewater, New Jersey 08807, USA; Tel: +1 908 393 9938; E-mail:<br />
krshah@polytherapeutics.com.<br />
www.transdermalmag.com<br />
11
12<br />
MARCH 2011 <strong>•</strong> TransDermal<br />
On the <strong>Transdermal</strong> Formulation of Peptide<br />
and Protein <strong>Drug</strong>s<br />
Xiaorong Shen and David Jones, Primera Analytical Solutions<br />
This short review covers some of the recent<br />
approaches to transdermal formulations for<br />
delivering proteins and peptides. These methods<br />
may overcome some of the difficulties<br />
associated with the delivery of those molecules.<br />
Protein and peptide drugs offer powerful therapeutic<br />
benefits to patients. While companies<br />
have marketed them since the introduction<br />
of insulin in the early twentieth century, the methods<br />
for dosing patients usually have been limited<br />
to administration by injection. This method not only<br />
is inconvenient for patients but also causes other<br />
difficulties, such as allergic reactions at the injection<br />
site. For drugs with relatively short half lives, injection<br />
methods — without the use of IV solutions —<br />
do not afford maintenance of appropriate levels of<br />
the drug without repeated dosing. The most convenient<br />
type of dosing, oral, is generally not available<br />
for peptide and protein drugs because proteolytic<br />
enzymes in the alimentary canal hydrolyze the<br />
peptides.<br />
Peptides are widely used in skin-care products,<br />
mostly in connection with the cosmetics industry,<br />
although medical providers routinely apply antibiotic<br />
peptides, such as polymyxin B, topically for<br />
wound care and other dermatological infections.<br />
While such topical use is important, the ability to<br />
transport peptides and proteins into physiological<br />
matrices in other organs is of vital importance.<br />
Proteins incorporate four levels of structure:<br />
<strong>•</strong> A primary structure that derives from the aminoacid<br />
sequence<br />
<strong>•</strong> A secondary structure that arrangements in<br />
helices and sheets define<br />
<strong>•</strong> A tertiary structure that refers to the way that the<br />
secondary structures fit together as a whole<br />
<strong>•</strong> A quaternary structure that is a representation of<br />
protein-to-protein interactions; for instance, whether<br />
the proteins are monomers, dimers, or oligomers of<br />
single or multiple protein units.<br />
Effects of Structure<br />
All of these structures affect not only the transport of<br />
these molecules but also their activity. To a lesser<br />
extent, the problems with hydrolysis, absorption,<br />
and stability also apply to peptides that, while<br />
smaller, exhibit at least a primary and a secondary<br />
structure. Both proteins and peptides also often<br />
depend on intramolecular bonds that can rearrange<br />
under some circumstances. Most often disulfide<br />
linkages connect these bonds, but some peptides<br />
are cyclic in nature. Again polymixin B is an example.<br />
Any chemical exposure can affect molecular<br />
rearrangement, racemization, and rearrangement of<br />
disulfide bonds, further complicating attempts to<br />
formulate and deliver them.<br />
Nevertheless many approaches to transdermal<br />
formulation are now under active investigation,<br />
and some have reached the stage of clinical<br />
development.<br />
Approaches to <strong>Transdermal</strong><br />
Formulation<br />
The dermis is the vascularized portion of the skin<br />
where hair follicles originate, where lymph vessels<br />
and sweat glands are found, and where drugs can
find their way into circulation in other tissues. To<br />
reach the dermis, molecules — in this case large,<br />
sensitive molecules — must penetrate the epidermis,<br />
which generally is about a tenth of a millimeter<br />
in thickness but nonetheless seals off the dermis [1].<br />
Formulators have attempted to transport peptides<br />
across membranes other than those represented by<br />
the skin, such as recent efforts to formulate insulin<br />
as an inhalable product. These efforts have proved<br />
promising and have resulted in the investment of<br />
much effort and money.<br />
Iontophoresis<br />
One transdermal approach that holds significant<br />
promise for accomplishing the task is iontophoresis,<br />
in which a small electrical potential is applied<br />
across skin layers. Proteins and peptides often<br />
exhibit well-defined polarities, deriving from the<br />
location of acidic residues, such as glutamic acid<br />
and aspartic acid, and of basic residues, such as histidine,<br />
lysine, or arginine. Depending on the pH,<br />
these residues can be charged and will migrate<br />
across many matrices. Many laboratory procedures,<br />
such as SDS PAGE, use electrical potentials to separate<br />
and transport peptides and proteins, and iontophoresis<br />
is not all that different except that the<br />
matrix is the skin.<br />
A typical system might consist of two electrodes,<br />
placed in nearby areas of the skin, with a small<br />
potential applied to each. The charged ions are<br />
drawn into the skin<br />
The use of a silver electrode and a silver chloride<br />
electrode is one method that has generated much<br />
interest for the delivery of peptides and proteins<br />
transdermally. A peptide that has a suitable charge,<br />
such as one having a number of arginines, moves in<br />
the direction of the current flow and into the dermis.<br />
After absorption into the tissues, the current maintains<br />
the flow of other ions, such as peptide counterions<br />
or physiological ions such as chloride.<br />
Leutinizing Hormone Releasing Hormone (LHRH)<br />
is a peptide that has been administered successfully<br />
in various settings in this way. This peptide contains<br />
three basic residues — histidine, arginine, and<br />
lysine — and thus is charged [2]. Often researchers<br />
must control the rate of migration carefully; for<br />
TransDermal <strong>•</strong> MARCH 2011<br />
example, by varying the voltage or the wave form<br />
of the voltage. They can adjust the currents to render<br />
the pro cess essentially painless. Other peptides<br />
delivered transdermally using iontophoresis in -<br />
clude salmon calcintonin [3] and human parathyroid<br />
hormone [4].<br />
Iontophoresis is just one approach and is clearly<br />
only workable with peptides and proteins that are<br />
highly charged and contain either basic or acidic<br />
residues.<br />
Chaperon Molecules<br />
Another approach involves the use of chaperon<br />
molecules to transport proteins across skin layers.<br />
Peptides do affect skin pores, and under the right<br />
conditions, can themselves be used as chaperones<br />
that help transport other peptides across the stratum<br />
corneum. For instance, researchers originally investigated<br />
the peptide Magainin, which is isolated from<br />
frog skin, in clinical trials as a potential anti-infective.<br />
Although regulators ultimately did not<br />
approve the drug for this application, Magainin is<br />
considered a safe compound and is known to<br />
increase pore size and porosity in skin. It works by<br />
disrupting the bilayer structure of the stratum<br />
corneum lipids, increasing porosity [5].<br />
Researchers are investigating insulin delivery in this<br />
way, and many ways to optimize chaperone-type<br />
delivery exist. Samir Mitragotri and his coworkers<br />
in the Chemical Engineering Department at the University<br />
of California Santa Barbara used a combinatorial,<br />
high-throughput-screening process to explore<br />
the transdermal applications of delivering peptides.<br />
Again, the peptide was LHRH [6]. The screening<br />
involved the use of 34 penetration-enhancing<br />
agents, with variable concentrations, allowing for<br />
the screening of thousands of formulations.<br />
Mitragotri was able to show a 100-fold increase in<br />
LHRH delivered to hairless rats and an 80-fold<br />
increase in delivery of macrosugar inulin through<br />
use of the best chaperone formulations that they<br />
developed.<br />
Another approach to maximizing the effectiveness<br />
of small peptides as transdermal chaperone molecules<br />
involves the discovery, via phage display, of<br />
peptides that can penetrate the skin. Wen and his<br />
13
14<br />
MARCH 2011 <strong>•</strong> TransDermal<br />
coworkers applied a phage library to rat skin and<br />
then collected phages that were isolated from the<br />
animal’s blood stream [7]. They were able to isolate a<br />
peptide with the sequence CSSSPSKHC. The re -<br />
searchers synthesized a cyclized analogue of this<br />
peptide and ultimately were able to show improved<br />
insulin delivery.<br />
Conclusion<br />
The methods described in this article have shown that<br />
it is possible to deliver peptides in vivo via transdermal<br />
routes. This article, however, is not a comprehensive<br />
discussion of such formulations. Many other<br />
transdermal approaches are known, including techniques<br />
such as painless microneedles [8].<br />
References<br />
1. Michniak, et al. European Journal of Pharmaceutics and<br />
Biopharmaceutics. 2005, 60:179–191.<br />
2. Raiman J, Koljonen M, Huikko K, Kostiainen R, Hirvonen<br />
J. <strong>Delivery</strong> and stability of LHRH and Nafarelin in human skin: The<br />
effect of constant/pulsed iontophoresis. Eur. J. Pharm. Sci. 2004,<br />
21:371–377.<br />
3. Chang SL, Hofmann GA, Zhang L, Deftos LJ, Banga AK.<br />
Stability of a transdermal salmon calcitonin formulation. <strong>Drug</strong><br />
Deliv. 2003, 10:41–45.<br />
4. Medi BM and Singh J. Electronically facilitated transdermal<br />
delivery of human parathyroid hormone (1-34). Int. J. Pharm.<br />
2003, 263:25–33.<br />
5. Prausnitz, MR and Langer R. Nature Biotechnology. 2008,<br />
26(11):1261-1268.<br />
6. Karande P, Jain A, Mitragotri S. Nature Biotechnology<br />
2004, 22(2):192-197.<br />
7. Chen Y, Shen Y, Guo X, Zhang C, Yang W, Ma M, Liu S,<br />
Zhang M, Wen L. <strong>Transdermal</strong> protein delivery by a co-administered<br />
peptide identified via phage display. Nature Biotechnology.<br />
2006, 24(4):455-460.<br />
8. Daniel P. Wermeling et al. Proc Natl Acad Sci. USA 102,<br />
4688–4693 (2005).<br />
Xiaorong Shen received her bachelor’s and master’s degrees in biology at<br />
Nanjing University in China and a PhD in genetics at Purdue<br />
University in the USA. Before receiving her PhD, she worked for more<br />
than eight years at the Chinese Jiangsu Institute on preformulations,<br />
formulations, manufacturing processes, analytical methods, and development.<br />
In 2007, she joined Primera Analytical Solutions where she<br />
now heads its formulations group. She works primarily on advanced<br />
material formulations, including bioresorbable polymers, large-molecule<br />
formulations, and stability and compatibility issues with novel, cuttingedge<br />
formulations.<br />
David Jones joined Primera Analytical Solutions in 2007 and currently<br />
is vice president of sales and marketing. With 25 years in the pharmaceutical<br />
industry, he previously was director of pharmaceutical sales for<br />
Isochem and vice president of development at Hopewell Laboratories. In<br />
the past, he worked on the development of a novel class of peptide<br />
reagents, urethane protected N-carboxyanhydrides (UNCAs), for<br />
Professor Murray Goodman’s laboratories and managed a laboratory at<br />
Multiple Peptide Systems that produced the first commercial peptide<br />
libraries. He received his bachelor’s and master’s degrees in chemistry at<br />
the State University of New York at Stony Brook<br />
Contact David Jones at Primera Analytical Solutions, 259 Wall Street,<br />
Princeton NJ 08534; Tel: +1 908 295 9011; E-mail: david.jones<br />
@primera-corp.com. www.primera-corp.com<br />
www.transdermalmag.com
ISSUE<br />
FOCUS<br />
pharmadur ®<br />
Polytherapeutics has developed<br />
patented transdermal technology<br />
based on its proprietary PharmaDur<br />
polymer. <strong>Drug</strong> formulation<br />
in the PharmaDur vehicle, as<br />
a gel or emulsion, forms an invisible<br />
and imperceptible hydrogel<br />
film that provides sustained drug<br />
delivery through the skin for 24<br />
hours.<br />
The company’s lead product is<br />
Pharmagesic <strong>Transdermal</strong> System,<br />
comprising the NSAID diclo -<br />
fenac in PharmaDur gel. Studies<br />
have demonstrated the product to<br />
be capable of producing effective<br />
diclofenac concentrations in the<br />
blood for eliciting systemic pharmacological<br />
activity with once-aday<br />
dosing. The key benefits of<br />
Pharmagesic include sustained<br />
therapeutic activity, minimization<br />
of GI-tract irritation, and potentially<br />
lower systemic-toxicity risks<br />
ContraCt researCh<br />
and development<br />
Located in Princeton, New Jersey,<br />
Primera Analytical Solutions is a<br />
contract research organization<br />
(CRO) founded in 2002. As a CRO,<br />
Primera supports the research and<br />
development of new drug substances.<br />
Primera is FDA-registered<br />
and audited, with no 483s issued.<br />
The company provides a full range<br />
of GLP bioanalytical LC/MS/MS<br />
and immunochemistry services as<br />
well as cGMP analytical services<br />
for drug development — discov-<br />
Formulation<br />
Techniques<br />
by avoiding high, peak diclofe -<br />
nac blood concentrations and<br />
hepatic first-pass metabolism.<br />
Phar magesic and PharmaDur are<br />
available as licensing or partnering<br />
opportunities for use with<br />
other drugs.<br />
ery and preclinical and clinical<br />
programs — and pharmaceutical<br />
product and process development<br />
for small and large molecules. Its<br />
core cap abilities include method<br />
development, validation, sample<br />
analysis, formulation, stability and<br />
release testing, and pharmaceutical<br />
testing services.<br />
Primera Analytical Solutions<br />
Corp<br />
Princeton, New Jersey, USA<br />
Tel: +1 609 921 7715<br />
TransDermal <strong>•</strong> marCh 2011<br />
Polytherapeutics, Inc.<br />
Bridgewater, New Jersey, USA<br />
Tel: +1 908 393 9938<br />
E-mail:<br />
info@polytherapeutics.com<br />
www.polytherapeutics.com<br />
Fax: +1 609 921 7716<br />
E-mail:<br />
David.Jones@primera-corp.com<br />
www.primera-corp. com<br />
15
16<br />
MARCH 2011 <strong>•</strong> TransDermal<br />
Calendar<br />
March 2011<br />
12th Skin Forum Annual Meeting.<br />
March 28th and 29th at Campus<br />
Westend, Goethe Universität<br />
in Frankfurt, Germany. Organized<br />
by the International Association<br />
for Pharmaceutical Technology<br />
in partnership with Skin<br />
Forum. www.apv-mainz.de/<br />
index.php?id=277<br />
April 2011<br />
<strong>Drug</strong> <strong>Delivery</strong> and Formulation<br />
America 2011. April 27th and<br />
28th at the Hyatt Regency La<br />
Jolla in San Diego, California,<br />
USA. Organized by World Trade<br />
Group (WTG). www.ddfsum<br />
mit.com<br />
July 2011<br />
The 38th Annual Meeting and<br />
Exposition of the Controlled Re -<br />
lease Society. July 30 to August 3<br />
at the Gaylord National Resort<br />
and Convention Center, National<br />
Harbor, Maryland, USA; Tel: +1<br />
651 454 7250 (Minnesota, USA).<br />
www.controlledrelease.org/<br />
main/meetings/default.cfm<br />
August 2011<br />
Barrier Function of Mammalian<br />
Skin. August 7 to 12 at the Waterville<br />
Valley Resort, Waterville<br />
Valley, New Hampshire, USA; Email:<br />
gmenon@ispcorp.com<br />
(New Jersey, USA) or juergen.l<br />
ademann@charite.de (Germany).<br />
www.grc.org/programs.aspx?<br />
year =2011&program=barrier<br />
For less than 2 minutes of your<br />
time, you can receive 6 free issues<br />
of TRANSDERMAL in 2011<br />
anywhere in the world.<br />
www.transdermalmag.com<br />
October 2011<br />
AAPS Annual Meeting and<br />
Convention. October 23 to 27 at<br />
the Washington Convention Center,<br />
Washington, DC, USA; Tel: +1<br />
703 243 2800 (Virginia, USA).<br />
www.aapspharmaceutica.com/<br />
meetings/annualmeet/am11/<br />
exhibit.asp<br />
November 2011<br />
The 1st Annual Symposium of<br />
<strong>Drug</strong> <strong>Delivery</strong> Systems. November<br />
3 to 5 in Shenzhen, China.<br />
Organized by Dalian BIT Life Sciences,<br />
Inc, 26 Gaoneng Street,<br />
R401, Dalian Hightech Zone<br />
Dalian, LN 116025, China; Tel:<br />
+0086 411 84799609 821; e-mail:<br />
grace@bitlifesciences.com.<br />
www.bitlifesciences.com/sdds20<br />
11/default.asp<br />
Do you qualify? It’s easy!<br />
Just visit our web site at www.transdermalmag.com<br />
Fill out the form completely.<br />
Make sure all information is correct.<br />
Click on the submit button, and you’re done!<br />
www.transdermalmag.com
January 2012<br />
<strong>Drug</strong> <strong>Delivery</strong> Partnerships<br />
2012. January 25 to 27 at Caesers<br />
Palace in Las Vegas, Nevada,<br />
USA. Tel: 1 888 670 8200 (toll free);<br />
+1 941 951 7885 (International); Email:<br />
register@iirusa.com. www<br />
.iirusa.com/ddp/welcome.xml<br />
People news<br />
Corium International<br />
adds two board<br />
members<br />
Menlo Park, California, USA —<br />
Corium International appointed<br />
Dr Bhaskar Chaudhuri and<br />
David L Greenwood to the board<br />
of direc tors. Chaudhuri most<br />
recently served as president of<br />
Valeant Phar maceuticals International<br />
and previously was president<br />
and CEO of Dow Pharmaceutical<br />
Services. Greenwood is<br />
executive vice president and CEO<br />
of Geron Corporation.<br />
Aveva hires alliance<br />
management director<br />
and marketing<br />
manager<br />
Miramar, Florida, USA — Aveva<br />
<strong>Drug</strong> <strong>Delivery</strong> Systems hired Paul<br />
Davis as director, alliance management,<br />
and Jared Hahn as marketing<br />
manager, both reporting<br />
directly to Robert J Bloder, vice<br />
president. With over eight years of<br />
experience at Beckman Coulter,<br />
Davis spent the last four years in<br />
business development and<br />
alliance management. Jared Hahn<br />
most recently worked for over<br />
eight years at Technology Catalysts<br />
Inter national (TCI), managing the<br />
company’s pharmaceutical and<br />
drug-delivery services, including<br />
the marketing of TCI’s services to<br />
pharmaceutical companies.<br />
Avantor names new<br />
CEO<br />
Phillipsburg, New Jersey, USA<br />
— Avantor Performance Materials<br />
named Jean-Marc Gilson as<br />
chief executive officer. Gilson was<br />
previously executive vice president<br />
and general manager of<br />
Dow Corning’s specialty chemicals<br />
business.<br />
Alliqua appoints five<br />
directors to board<br />
New York, New York, USA —<br />
Alliqua, previously Hepalife<br />
Technologies, appointed five<br />
mem bers to its board of directors.<br />
Joseph M Leone is an expert in<br />
commercial lending to small and<br />
midsized businesses. Michael<br />
Goldberg, MD, currently is a<br />
managing partner at the investment<br />
firm Montaur Capital Partners.<br />
Nochum Stein currently<br />
serves as Chairman and CEO of<br />
American European Group<br />
(AEG), a New-York-based in surance<br />
holding company focusing<br />
on supporting small to mid-sized<br />
businesses. Kenneth Pearsen,<br />
MD currently is the CEO of Western<br />
New York Radiology Associates.<br />
Jeffrey Sklar is the managing<br />
partner of Sklar, Heyman,<br />
and Company, a re gional accounting<br />
firm.<br />
Alliqua retains<br />
strategic consultant<br />
New York, New York, USA —<br />
Alli qua, previously Hepalife<br />
Tech nologies, appointed Dr<br />
Ronald S Harland as a strategic<br />
consultant. Harland has worked<br />
in the field of product development<br />
and studied advanced drug<br />
delivery systems and applications<br />
for over 20 years. Previously,<br />
Harland was founder and<br />
CEO at PharmSolv.<br />
Supplier news<br />
Adhesives Research<br />
receives ISO 13485<br />
certification<br />
Glen Rock, Pennsylvania, USA<br />
— Adhesives Research received<br />
ISO 13485 certification for its North<br />
American manufactur ing facility<br />
in Glen Rock. This certification<br />
supplements the company’s existing<br />
ISO 9001 standards.<br />
NuSil introduces<br />
thermally conductive<br />
silicones<br />
Carpinteria, California, USA —<br />
NuSil Technology introduced two<br />
thermally conductive, health care<br />
silicone elastomers that can be<br />
used as a cure-in-place adhesive<br />
or potting compound between<br />
electrical components and heat<br />
sinks to keep medical devices<br />
operating at optimal levels. With<br />
a two-part configuration, MED-<br />
2980, a 1.6 W/(mK) silicone, is<br />
good for larger devices. With low<br />
volatility and low ionic con tent,<br />
MED-2955 serves more sensitive<br />
electronic devices or processes.<br />
IDEX acquires Microfluidics<br />
International<br />
Lake Forest, Illinois and Newton,<br />
Massachusetts, USA —<br />
IDEX will acquire Microfluidics<br />
International for $1.35 per share,<br />
net to the seller in cash and subject<br />
to adjustment as defined in<br />
the merger agreement. The companies<br />
expect the transaction to<br />
close in the first quarter of 2011.<br />
Advertiser index<br />
Aveva <strong>Drug</strong> <strong>Delivery</strong><br />
Systems <strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong> Cover 4<br />
TapeMark<strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong><strong>•</strong>3
T R A N S D E R M A L T R A N S C E N D E N C E<br />
A h i g h e r l e v e l o f p e r f o r m a n c e D e l i v e r s n e w p o s s i b i l i t i e s<br />
That’s the promise of Aveva <strong>Drug</strong> <strong>Delivery</strong> Systems,<br />
combining innovation and unparalleled industry<br />
experience to advance drug delivery and pioneer<br />
new frontiers in transdermal drug delivery for new<br />
chemical entities and life cycle management<br />
opportunities to enhance existing products.<br />
As one of the world’s largest manufacturers<br />
of transdermal patches offering a full range<br />
of research, development and manufacturing<br />
capabilities, Aveva transcends traditional limitations<br />
of patch technology and business partnerships to<br />
achieve new levels of product and corporate<br />
performance.<br />
© Copyright 2005. Aveva <strong>Drug</strong> <strong>Delivery</strong> Systems. All rights reserved.<br />
> Customizing solutions to the unique characteristics<br />
of each drug<br />
> Masterfully balancing the patch properties of<br />
adhesion reliability and gentleness that lead to<br />
an enhanced patient experience.<br />
> Managing a higher drug concentration in a<br />
smaller patch<br />
A flexible, customer-oriented business philosophy<br />
that adds value to projects and exceeds customer<br />
expectations.<br />
To license a product or to see how we can add value<br />
to your project, call Robert J. Bloder, Vice President<br />
Business Development, at 954.624.1374 or visit<br />
www.AvevaDDS.com