Targeted development of biosimilar pharmaceutical products
Targeted development of biosimilar pharmaceutical products
Targeted development of biosimilar pharmaceutical products
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
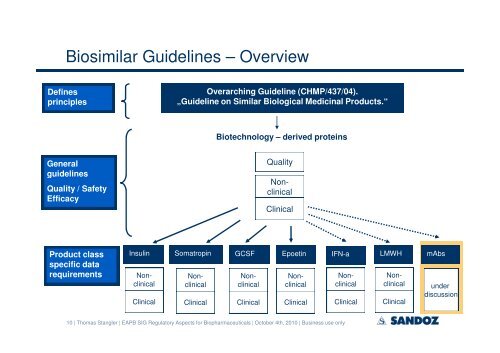
Biosimilar Guidelines – Overview<br />
Defines<br />
principles<br />
General<br />
guidelines<br />
Quality / Safety<br />
Efficacy<br />
Product class<br />
specific data<br />
requirements<br />
Overarching Guideline (CHMP/437/04).<br />
„Guideline on Similar Biological Medicinal Products.“<br />
Biotechnology – derived proteins<br />
Quality<br />
Nonclinical<br />
Clinical<br />
Insulin Somatropin GCSF Epoetin IFN-a LMWH<br />
Nonclinical<br />
Clinical<br />
Nonclinical<br />
Clinical<br />
Nonclinical<br />
Clinical<br />
Nonclinical<br />
Clinical<br />
10 | Thomas Stangler | EAPB SIG Regulatory Aspects for Bio<strong>pharmaceutical</strong>s | October 4th, 2010 | Business use only<br />
Nonclinical<br />
Clinical<br />
Nonclinical<br />
Clinical<br />
mAbs<br />
under<br />
discussion







![Roy Forster [Kompatibilitätsmodus]](https://img.yumpu.com/7737040/1/190x135/roy-forster-kompatibilitatsmodus.jpg?quality=85)



