Targeted development of biosimilar pharmaceutical products
Targeted development of biosimilar pharmaceutical products
Targeted development of biosimilar pharmaceutical products
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
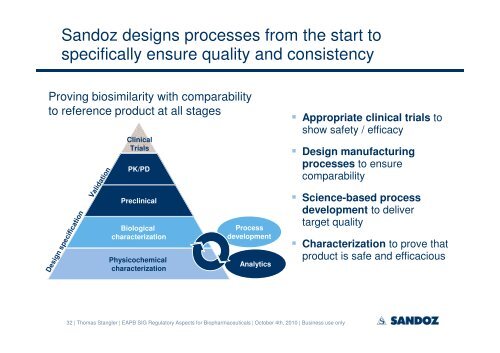
Sandoz designs processes from the start to<br />
specifically ensure quality and consistency<br />
Proving <strong>biosimilar</strong>ity with comparability<br />
to reference product at all stages<br />
Clinical<br />
Trials<br />
PK/PD<br />
Preclinical<br />
Biological<br />
characterization<br />
Physicochemical<br />
characterization<br />
Process<br />
<strong>development</strong><br />
Analytics<br />
32 | Thomas Stangler | EAPB SIG Regulatory Aspects for Bio<strong>pharmaceutical</strong>s | October 4th, 2010 | Business use only<br />
� Appropriate clinical trials to<br />
show safety / efficacy<br />
� Design manufacturing<br />
processes to ensure<br />
comparability<br />
� Science-based process<br />
<strong>development</strong> to deliver<br />
target quality<br />
� Characterization to prove that<br />
product is safe and efficacious







![Roy Forster [Kompatibilitätsmodus]](https://img.yumpu.com/7737040/1/190x135/roy-forster-kompatibilitatsmodus.jpg?quality=85)



