Clinical Research Nurses: Roles and Qualifications in South Korea
Clinical Research Nurses: Roles and Qualifications in South Korea
Clinical Research Nurses: Roles and Qualifications in South Korea
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
254 MEDICAL INFORMATION<br />
Jeong, Kang, Kim<br />
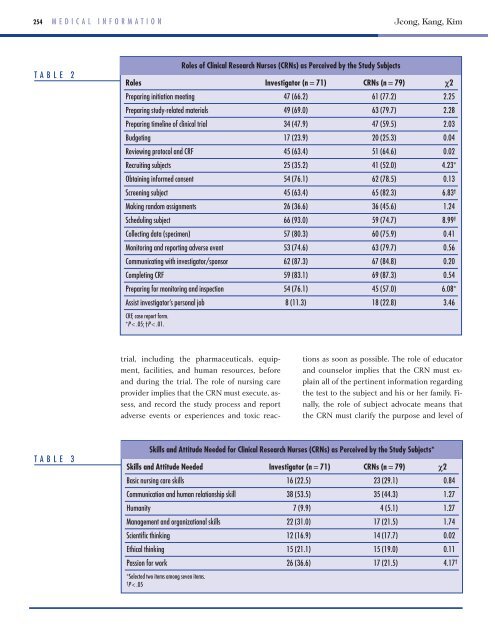
TABLE 2<br />
TABLE 3<br />
<strong>Roles</strong> Investigator (n = 71) CRNs (n = 79) χ2<br />
Prepar<strong>in</strong>g <strong>in</strong>itiation meet<strong>in</strong>g 47 (66.2) 61 (77.2) 2.25<br />
Prepar<strong>in</strong>g study-related materials 49 (69.0) 63 (79.7) 2.28<br />
Prepar<strong>in</strong>g timel<strong>in</strong>e of cl<strong>in</strong>ical trial 34 (47.9) 47 (59.5) 2.03<br />
Budget<strong>in</strong>g 17 (23.9) 20 (25.3) 0.04<br />
Review<strong>in</strong>g protocol <strong>and</strong> CRF 45 (63.4) 51 (64.6) 0.02<br />
Recruit<strong>in</strong>g subjects 25 (35.2) 41 (52.0) 4.23*<br />
Obta<strong>in</strong><strong>in</strong>g <strong>in</strong>formed consent 54 (76.1) 62 (78.5) 0.13<br />
Screen<strong>in</strong>g subject 45 (63.4) 65 (82.3) 6.83 †<br />
Mak<strong>in</strong>g r<strong>and</strong>om assignments 26 (36.6) 36 (45.6) 1.24<br />
Schedul<strong>in</strong>g subject 66 (93.0) 59 (74.7) 8.99 †<br />
Collect<strong>in</strong>g data (specimen) 57 (80.3) 60 (75.9) 0.41<br />
Monitor<strong>in</strong>g <strong>and</strong> report<strong>in</strong>g adverse event 53 (74.6) 63 (79.7) 0.56<br />
Communicat<strong>in</strong>g with <strong>in</strong>vestigator/sponsor 62 (87.3) 67 (84.8) 0.20<br />
Complet<strong>in</strong>g CRF 59 (83.1) 69 (87.3) 0.54<br />
Prepar<strong>in</strong>g for monitor<strong>in</strong>g <strong>and</strong> <strong>in</strong>spection 54 (76.1) 45 (57.0) 6.08*<br />
Assist <strong>in</strong>vestigator’s personal job 8 (11.3) 18 (22.8) 3.46<br />
CRF, case report form.<br />
*P < .05; †P < .01.<br />
<strong>Roles</strong> of <strong>Cl<strong>in</strong>ical</strong> <strong>Research</strong> <strong>Nurses</strong> (CRNs) as Perceived by the Study Subjects<br />
trial, <strong>in</strong>clud<strong>in</strong>g the pharmaceuticals, equipment,<br />
facilities, <strong>and</strong> human resources, before<br />
<strong>and</strong> dur<strong>in</strong>g the trial. The role of nurs<strong>in</strong>g care<br />
provider implies that the CRN must execute, assess,<br />
<strong>and</strong> record the study process <strong>and</strong> report<br />
adverse events or experiences <strong>and</strong> toxic reac-<br />
tions as soon as possible. The role of educator<br />
<strong>and</strong> counselor implies that the CRN must expla<strong>in</strong><br />
all of the pert<strong>in</strong>ent <strong>in</strong>formation regard<strong>in</strong>g<br />
the test to the subject <strong>and</strong> his or her family. F<strong>in</strong>ally,<br />
the role of subject advocate means that<br />
the CRN must clarify the purpose <strong>and</strong> level of<br />
Skills <strong>and</strong> Attitude Needed for <strong>Cl<strong>in</strong>ical</strong> <strong>Research</strong> <strong>Nurses</strong> (CRNs) as Perceived by the Study Subjects*<br />
Skills <strong>and</strong> Attitude Needed Investigator (n = 71) CRNs (n = 79) χ2<br />
Basic nurs<strong>in</strong>g care skills 16 (22.5) 23 (29.1) 0.84<br />
Communication <strong>and</strong> human relationship skill 38 (53.5) 35 (44.3) 1.27<br />
Humanity 7 (9.9) 4 (5.1) 1.27<br />
Management <strong>and</strong> organizational skills 22 (31.0) 17 (21.5) 1.74<br />
Scientific th<strong>in</strong>k<strong>in</strong>g 12 (16.9) 14 (17.7) 0.02<br />
Ethical th<strong>in</strong>k<strong>in</strong>g 15 (21.1) 15 (19.0) 0.11<br />
Passion for work 26 (36.6) 17 (21.5) 4.17 †<br />
*Selected two items among seven items.<br />
† P < .05