FAQ Brochure - Gore Medical
FAQ Brochure - Gore Medical
FAQ Brochure - Gore Medical
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
4<br />
How does end-point covalent bonding of heparin differ from<br />
conventional covalent bonding of heparin and ionic bonding of heparin?<br />
Conventional Covalent Bonding<br />
In conventional, multiple point, covalent bonding, the anticoagulant properties of heparin are lost<br />
due to the fact that the heparin active site is not available to antithrombin.<br />
Ionic Bonding<br />
In ionic bonding, the attractive force of negatively charged heparin and positively charged coating<br />
can be easily broken, thus allowing heparin to be released from the surface over time. Sustained<br />
thromboresistance cannot be achieved.<br />
End-point Covalent Bonding<br />
How long does the heparin last?<br />
The heparin molecule (yellow) is bonded (white) at<br />
multiple points to the device surface (orange).<br />
The heparin active site (red) is unavailable to<br />
antithrombin. Without bioactivity, the anticoagulant<br />
properties of heparin are lost.<br />
The negatively (-) charged heparin molecule (yellow) is<br />
attracted to a positively (+) charged coating (orange) on the<br />
device surface. This attractive force can be broken, allowing<br />
heparin to release from the surface over time. Sustained<br />
thromboresistance cannot be achieved.<br />
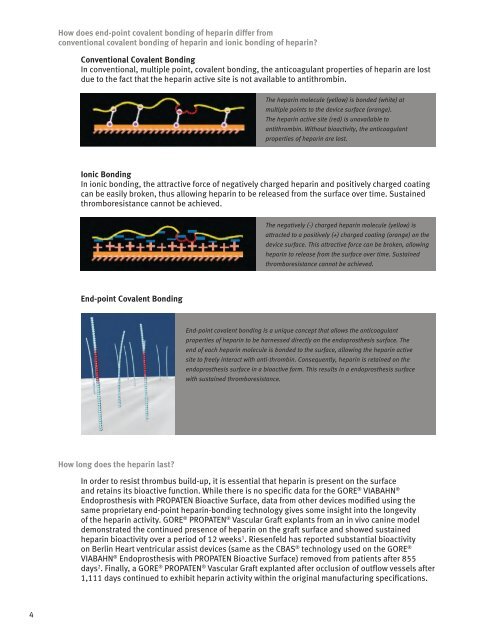
End-point covalent bonding is a unique concept that allows the anticoagulant<br />
properties of heparin to be harnessed directly on the endoprosthesis surface. The<br />
end of each heparin molecule is bonded to the surface, allowing the heparin active<br />
site to freely interact with anti-thrombin. Consequently, heparin is retained on the<br />
endoprosthesis surface in a bioactive form. This results in a endoprosthesis surface<br />
with sustained thromboresistance.<br />
In order to resist thrombus build-up, it is essential that heparin is present on the surface<br />
and retains its bioactive function. While there is no specific data for the GORE ® VIABAHN ®<br />
Endoprosthesis with PROPATEN Bioactive Surface, data from other devices modified using the<br />
same proprietary end-point heparin-bonding technology gives some insight into the longevity<br />
of the heparin activity. GORE ® PROPATEN ® Vascular Graft explants from an in vivo canine model<br />
demonstrated the continued presence of heparin on the graft surface and showed sustained<br />
heparin bioactivity over a period of 12 weeks 1 . Riesenfeld has reported substantial bioactivity<br />
on Berlin Heart ventricular assist devices (same as the CBAS ® technology used on the GORE ®<br />
VIABAHN ® Endoprosthesis with PROPATEN Bioactive Surface) removed from patients after 855<br />
days 2 . Finally, a GORE ® PROPATEN ® Vascular Graft explanted after occlusion of outflow vessels after<br />
1,111 days continued to exhibit heparin activity within the original manufacturing specifications.