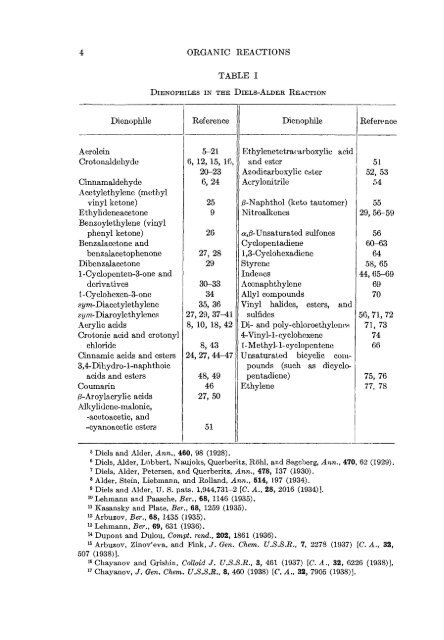
4 OBGANIC REACTIONS Dienophile Acrolein Crotonaldehyde Cinnamaldehyde Acetylethylene (methyl vinyl ketone) Ethylideneacetone Benzoylethylene (vinyl phenyl ketone) Benzalacetone and benzalacetophenone Dibenzalacetone l-Cyclopenten-3-one and derivatives l-Cyclohexen-3-one st/m-Diacctylethylene st/m-Diaroylethylenes Acrylic acids Crotonic acid and crotonyl chloride Cinnamic acids and esters 3,4-Dihy dro- 1-naphthoic acids and esters Coumarin /3-Aroylacrylic acids Aikylidene-malonic, -acetoacetic, and -cyanoacetic esters TABLE I DlENOPHILES IN THE DiELS-ALDEB. REACTION i Reference 5-21 6, 12, 15, 16, 20-23 6,24 25 9 26 27, 28 29 I 30-33 34 35, 36 27, 29, 37-41 8, 10, 18, 42 8,43 24, 27, 44r-47 48, 49 46 27, 50 51 Dienophile I Ethylenetetraearboxylic acid and ester 51 Azodicarboxylic ester 52, 53 Acrylonitrile I 54 /3-Naphthol (keto tautomer) Nitroalkenes ^^-Unsaturated sulfones Cyclopentadiene 1,3-Cyclohexadiene Styrene Indencs Acenaphthylene Allyl compounds Vinyl haHdes, esters, and sulfides Di- and poly-chloroethylenes 4-Vinyl-l-cyclohexene 1-Methyl- 1-cyclopentene Unsaturated bicyclic compounds (such as dicyclopentadiene) Ethylene Reference 55 29,56-59 56 60-63 64 58,65 44, 65-69 69 70 56,71,72 71,73 74 66 75,76 77, 78 5 Diols and Alder, Ann,, 460, 98 (1928), 6 Diols, Alder, Liibbert, Naujoks, Querberitz, Rohl, and Segeberg, Ann., 470, 62 (1929). 7 Diels, Alder, Petersen, and Querberitz, Ann., 478, 137 (1930). 8 Alder, Stein, Liebmann, and Holland, Ann., 514, 197 (1934). 9 Diels and Alder, U. S. pats. 1,944,731-2 [C. A., 28, 2016 (1934)]. 10 Lehmann and Paasche, Ber., 68, 1146 (1935). 11 Kasansky and Plate, Ber., 68, 1259 (1935). 12 Arbuzov, Ber., 68, 1435 (1935). 13 Lehmann, Ber., 69, 631 (1936). 14 Dupont and Dulou, Compt. rend., 202, 1861 (1936). 15 Arbuzov, Zinov'eva, and Fink, J. Gen. Chem. U.S.S.R., 7, 2278 (1937) [C. A., 32, 507 (1938)]. 16 Chayanov and Grishin, Colloid J. U.S.S.B., 3, 461 (1937) [C. A., 32, 6226 (1938)]. 17 Chayanov, /. Gen. Chem. U.S.S.B., 8, 460 (1938) [C. A., 32, 7905 (1938)].
DIENE SYNTHESIS I 5 ^Lehmann, Ber., 71, 1874 (1938). 19 Dupont, Dulou, Desreux, and Picoux, Bull. soc chim. France, [5] 5, 322 (1938). 20 Langenbeck, Godde, Weschky, and Schaller, Ber., 75, 232 (1942) [C. A., 37, 3746 (1943)]. 21 Fiesselmann, Ber., 75, 881 (1942) [C. A., 37, 3417 (1943)]. 22 Shorygin and Guseva, J. Gen. Chem. U.S.S.R., 6, 1569 (1936) [C. A., 31, 2184 (1937)]. 23 Clar, Ber., 72, 1817 (1939). 24 Fujise, Horiuti, and Takahashi, Ber., 69, 2102 (1936). 26 Petrov, /. Gen. Chem. U.S.S.R., 11, 309 (1941) [C. A., 35, 5873 (1941)]. 26 Allen, Bell, Bell, and Van Allan, /. Am. Chem. Soc, 62, 656 (1940). 27 Bergmann and Eschinazi, J. Am. Chem. Soc, 65, 1405 (1943). 28 Natsinskaya and Petrov, J. Gen. Chem. U.S.S.R., 11, 665 (1941) [C. A., 35, 6934 (1941)]. 29 Bergmann, Eschinazi, and Neeman, J. <strong>Org</strong>. Chem., 8, 179 (1943). 30 Dane, Schmitt, and Rautenstrauch, Ann., 532, 29 (1937). 31 Dane and Schmitt, Ann., 536, 196 (1938). 32 Dane and Schmitt, Ann., 537, 246 (1939). 33 Dane and Eder, Ann., 539, 207 (1939). 34 Bartlett and Woods, J. Am. Chem. Soc, 62, 2933 (1940). 35 Goldberg and Muller, HeIv. Chim. Acta, 21, 1699 (1938). 36 Goldberg and Muller, HeIv. Chim. Acta, 23, 831 (1940). 37 Adams and Geissman, /. Am. Chem. Soc, 61, 2083 (1939). 38 Adams and Gold, J. Am. Chem. Soc, 62, 56 (1940). 39 Adams and Wearn, /. Am. Chem. Soc, 62, 1233 (1940). 40 Adams and Gold, /. Am. Chem. Soc, 62, 2038 (1940). 41 Adams, U. S. pat. 2,341,850 [C. A., 38, 4270 (1944)]. 42 Komppa and Komppa, 'Ber., 69, 2606 (1936). 43 Komppa and Beckmann, Ann., 523, 68 (1936). 44 Weiss and Beller, Monatsh., 61, 143 (1932). 45 Weizmann, Bergmann, and Berlin, J. Am. Chem. Soc, 60, 1331 (1938), 46 Adams, McPhee, Carlin, and Wicks, J. Am. Chem. Soc, 65, 356 (1943). 47 Adams and Carlin, J. Am. Chem. Soc, 65, 360 (1943). 48 Fieser and Holmes, /. Am. Chem. Soc, 58, 2319 (1936). 49 Fieser and Holmes, J. Am. Chem. Soc, 60, 2548 (1938). 60 Fieser and Fieser, J". Am. Chem. Soc, 57, 1679 (1935). 6 1 Alder and Rickert, Ber., 72, 1983 (1939). 52 Diels, Blom, and KoIl, Ann., 443, 242 (1925). 53 Diels, Schmidt, and Witte, Ber., 71, 1186 (1938). 54 Wolfe, U. S. pat. 2,217,632 [C. A., 35, 1069 (1941)]. 55 SaKeId, Ber., 73, 376 (1940). 56 Alder, Rickert, and Windemuth, Ber., 71, 2451 (1938). 57 Allen and Bell, /. Am. Chem. Soc, 61, 521 (1939). 58 Allen, Bell, and Gates, J. <strong>Org</strong>. Chem., 8, 373 (1943). 59 Nightingale and Janes, J. Am. Chem. Soc, 66, 352 (1944). 60 Alder, Stein, and Finzenhagen, Ann., 485, 223 (1931). 61 Alder, Stein, Eckardt, Buddenbrock, and Schneider, Ann., 504, 216 (1933). 62 Alder and Stein, Angew. Chem., 47, 837 (1934). ^Grummitt, Klopper, and Blenkhorn, /. Am. Chem. Soc, 64, 604 (1942). 64 Alder and Stein, Ann., 496, 197 (1932). 65 Alder and Rickert, Ber., 71, 379 (1938). ^Bergmann and Weizmann, J. <strong>Org</strong>. Chem., 9, 352 (1944). 67 Swain and Todd, J. Chem. Soc, 1942, 626. 68 Mameli, Pancotto, and Crestani, Gazz. chim. UaL, 67, 669 (1937). 69 Dilthey, Henkels, and Schaefer, Ber., 71, 974 (1938). 70 Alder and Windemuth, Ber., 71, 1939 (1938). 71 Alder and Rickert. Ann., 543, 1 (1940).
- Page 1 and 2: Organic Reactions VOLUME IV EDITORI
- Page 3 and 4: PREFACE TO THE SERIES In the course
- Page 5 and 6: CONTENTS CHAPTER PAGE 1. THE DIELS-
- Page 7 and 8: CHAPTER 1 THE DIELS-ALDER REACTION
- Page 9: 2. C6H5CH=CHA. DIENE SYNTHESIS I S
- Page 13 and 14: DIENE SYNTHESIS I 7 and IV for thes
- Page 15 and 16: DIENE SYNTHESIS I 9 tion of the com
- Page 17 and 18: Maleic anhydride Fumaric anhydride
- Page 19 and 20: DIENE SYNTHESIS I 13 jugated system
- Page 21 and 22: DIENE SYNTHESIS I 15 Mesaconic (met
- Page 23 and 24: DIENE SYNTHESIS I 17 An interesting
- Page 25 and 26: DIENE SYNTHESIS I 19 CH2 CH3 O Il I
- Page 27 and 28: CH C2H5 I CH2 XXX DIENE SYNTHESIS I
- Page 29 and 30: DIENE SYNTHESIS I cyclic dienes fre
- Page 31 and 32: DIENE SYNTHESIS I 25 Although l,2,3
- Page 33 and 34: DIENE SYNTHESIS I 27 Certain types
- Page 35 and 36: DIENE SYNTHESIS I 29 Hydrocarbons c
- Page 37 and 38: DIENE SYNTHESIS I 31 Diels-Alder re
- Page 39 and 40: DIENE SYNTHESIS I 33 give LXXX. If
- Page 41 and 42: DIENE SYNTHESIS I 35 Numerous other
- Page 43 and 44: DIENE SYNTHESIS I 37 Maleic anhydri
- Page 45 and 46: DIENE SYNTHESIS I 39 Indole derivat
- Page 47 and 48: DIENE SYNTHESIS I 41 anhydride. In
- Page 49 and 50: DIENE SYNTHESIS I 43 tetrahydronaph
- Page 51 and 52: DIENE SYNTHESIS I 45 TABLE III—Co
- Page 53 and 54: DIENE SYNTHESIS I 47 TABLE IV ADDtr
- Page 55 and 56: Cycloheptatriene DIENE SYNTHESIS I
- Page 57 and 58: BIENE SYNTHESIS I 51 TABLE V—Cont
- Page 59 and 60: DIENE SYNTHESIS I 53 TABLE VI ADDTJ
- Page 61 and 62:
BIENE SYNTHESIS I 55 TABLE VII—Co
- Page 63 and 64:
DIENE SYNTHESIS I 57 REFERENCES TO
- Page 65 and 66:
DIENE SYNTHESIS I 59 422 Diels and
- Page 67 and 68:
DIENE SYNTHESIS II 61 PAGE TABLKs O
- Page 69 and 70:
DIENE SYNTHESIS II 63 The configura
- Page 71 and 72:
DIENE SYNTHESIS II 65 Aromatic comp
- Page 73 and 74:
DIENE SYNTHESIS II 67 55%). 13 Cycl
- Page 75 and 76:
H8Cr H8C JCO2H XSV iOCH, DIENE SYNT
- Page 77 and 78:
DIENE SYNTHESIS II 71 additions wit
- Page 79 and 80:
DIENE SYNTHESIS II , 73 been report
- Page 81 and 82:
DIENE SYNTHESIS II 75 drogenation u
- Page 83 and 84:
DIENE SYNTHESIS II 77 Allyl, Vinyl,
- Page 85 and 86:
DIENE SYNTHESIS II 79 (XLVII) that
- Page 87 and 88:
~r (PH.)* + CO2H CO5 :H CO2CH3 CO2C
- Page 89 and 90:
DIENE SYNTHESIS II 83 covered as th
- Page 91 and 92:
DIENE SYNTHESIS II 85 anhydride fro
- Page 93 and 94:
DIENE SYNTHESIS II 87 or even preve
- Page 95 and 96:
DIENE SYNTHESIS II 89 SELECTION OF
- Page 97 and 98:
DIENE SYNTHESIS II 91 liquid separa
- Page 99 and 100:
Acetylenic Dienophile Ethyl acetyle
- Page 101 and 102:
DIENE SYNTHESIS II 95 TABLE IV YIEL
- Page 103 and 104:
1-Diethylaminobutadiene 1,1-Dimeth
- Page 105 and 106:
1-Methylbutadiene (piperylene) l-Me
- Page 107 and 108:
1,5,5,6-Tetramethylcyclohexadiene (
- Page 109 and 110:
1-Methyl-l-phenylbtrfcadiene l-Met
- Page 111 and 112:
Benzalacetophenone Bicyclohexenyl r
- Page 113 and 114:
f$-Benzoylacrylic acid, 2, b-dimeth
- Page 115 and 116:
Bicyelohexenyl c r 2,3-Dimethylbuta
- Page 117 and 118:
Isoprene Cinnamic acid,2,6~ dimetho
- Page 119 and 120:
Isoprene Cinnamic acid,omeihoxy- (t
- Page 121 and 122:
Coumann Butadiene 2,3-Dimethylbutad
- Page 123 and 124:
1,3-Dimethylbutadiene 1,4-Dimethyl
- Page 125 and 126:
1,1,3-Trimethylbutadiene 1,1,4-Tri
- Page 127 and 128:
2,3-Dimethylbutadiene Croio7toladon
- Page 129 and 130:
2,3-Dimethylbutadiene2,3-Diphenylbu
- Page 131 and 132:
Cyclopentadiene 2,3-Dimethylbutadie
- Page 133 and 134:
Z}4rDihydro-5-bromo- 1\8-dimethoxy-
- Page 135 and 136:
Z^Dihydro-l-ftieihoxy-2-naphthoic a
- Page 137 and 138:
Ethyl ethytidenemalonate Butadiene
- Page 139 and 140:
1,4-Diphenylbutadiene2,3-Diphenylbu
- Page 141 and 142:
6 The product gradually decomposes
- Page 143 and 144:
Cyclohexadiene Cyclopentadiene 2,3-
- Page 145 and 146:
p-Nitrostyrenej 3,4-cfomethoxy- 2,3
- Page 147 and 148:
Dihydrothiophene-1 - dioxide Butadi
- Page 149 and 150:
Piperylcyclone Tetraphenyleyelopent
- Page 151 and 152:
AUyI iodide Cyclopentadiene AUyI is
- Page 153 and 154:
Tetraphenylcyclopentadienone Pheney
- Page 155 and 156:
Indene a,a , -Diphenyl-/3,j3'isoben
- Page 157 and 158:
1,5,5-Trimethyleyclopentadiene Tri
- Page 159 and 160:
Addends Acetylene Tetraphenylcyelop
- Page 161 and 162:
Trimethylcyclopentadiene? (Damsky)
- Page 163 and 164:
Cyclopentadiene 9,10-Dibromoaiithra
- Page 165 and 166:
Propargyl aldehyde 2,3-Dimethylbuta
- Page 167 and 168:
Addends Acetylenedicarboxylic acid
- Page 169 and 170:
I-* SO N-ce-Dimethylindole 3,5-Dim
- Page 171 and 172:
Oi N-Methylindole a-Methylpyrrole N
- Page 173 and 174:
OS Quinaldine Quinoline CX | JCH. j
- Page 175 and 176:
2,3,4-Trimethylpyrrole H3Cp CCO2CH3
- Page 177 and 178:
1,3-Dimethylbutadiene 1,1,3-Trimet
- Page 179 and 180:
DIENE SYNTHESIS II 110 Diels and Al
- Page 181 and 182:
PREPARATION OF AMINES BY REDUCTIVE
- Page 183 and 184:
PREPARATION OF AMINES BY REDUCTIVE
- Page 185 and 186:
PREPARATION OF AMINES BY REDUCTIVE
- Page 187 and 188:
PREPARATION OF AMINES BY REDUCTIVE
- Page 189 and 190:
PREPARATION OF AMINES BY REDUCTIVE
- Page 191 and 192:
PREPARATION OF AMINES BY REDUCTIVE
- Page 193 and 194:
PREPARATION OF AMINES BY REDUCTIVE
- Page 195 and 196:
PREPARATION OF AMINES BY REDUCTIVE
- Page 197 and 198:
PREPARATION OF AMINES BY REDUCTIVE
- Page 199 and 200:
PREPARATION OF AMINES BY REDUCTIVE
- Page 201 and 202:
PREPARATION OF AMINES BY REDUCTIVE
- Page 203 and 204:
PREPARATION OF AMINES BY REDUCTIVE
- Page 205 and 206:
PREPARATION OF AMINES BY REDUCTIVE
- Page 207 and 208:
PREPARATION OF AMINES BY REDUCTIVE
- Page 209 and 210:
PREPARATION OF AMINES BY REDUCTIVE
- Page 211 and 212:
PREPARATION OF AMINES BY REDUCTIVE
- Page 213 and 214:
PREPARATION OF AMINES BY REDUCTIVE
- Page 215 and 216:
CH3CH=CHCHO CH3(CHS)2CH=C(CH2CH3)CH
- Page 217 and 218:
(CHs)2C=CHCOCH3 (CH3)2C=CH(CH2)2COC
- Page 219 and 220:
CH3COCH3 CH3COCH3 CH3COCH2CH3 CH3CO
- Page 221 and 222:
H2N (CH2) 2NH2 H2N(CH2)2NH2 H2N{CH2
- Page 223 and 224:
P-HOC6H4CH2CH(CH3)NH2 CH2O P-CH3OC6
- Page 225 and 226:
CH3NH2 CH3NH2 CH3NH2 CH3NH2 CH3NH2
- Page 227 and 228:
HO(CH2)2NH2 HO(CHa)2NH2 HO(CH2)2NH2
- Page 229 and 230:
(GHg)2COHCH2NH2 (CHs)2COHCH2NH2 CH3
- Page 231 and 232:
Amine, Nitro, Nitroso, or Azo Compo
- Page 233 and 234:
P-HOC6H4NH2 P-HOC6H4NH2 - P-HOC6H4N
- Page 235 and 236:
CeHgNHs C6H5NH2 C6HgNH2 C6H5NH2 P-C
- Page 237 and 238:
HN=C(CH3)CO2H CH2 SehifPs Base / \
- Page 239 and 240:
CH3CH2N=CHC6H5J CH3(CH2)2N=CHCH3f C
- Page 241 and 242:
C6HuN=CH(CH2)2CH3t C6Hi1N=CH(CH2)2C
- Page 243 and 244:
p-C6H5(CH2)2N=CHC6H40H f p-C6H5(CH2
- Page 245 and 246:
C6H5N-=CHCH2CH(CH3)2f C6H5N=CH(CHOH
- Page 247 and 248:
2,4'-CH3C6H4N=CHC6H4CIf 2,4 / -CH3C
- Page 249 and 250:
0-CH3OC6H4N=CH(CHOH)4CH2OH m-CH3OC6
- Page 251 and 252:
Amine Used CH3CH2NH2 Amine Used CH3
- Page 253 and 254:
Nitro Compound Used ^HOC6H4NO2 P-H2
- Page 255 and 256:
C6H5CHOHCH2CH(CH3)- NHCH3 C6H5CHOHC
- Page 257 and 258:
Amine Used (CHs)2NH (CHs)2NH (CH3)2
- Page 259 and 260:
C6H5NH(CHs)2CH3 C6H5NH(CHs)3CH3 C6H
- Page 261 and 262:
PREPARATION OF AMINES BY REDUCTIVE
- Page 263 and 264:
THE ACYLOINS 257 euphony and to avo
- Page 265 and 266:
THE ACYLOINS 259 A mechanism simila
- Page 267 and 268:
THE ACYLOINS 261 There is a strikin
- Page 269 and 270:
TSE ACYLOINS 263 upon the rigid exc
- Page 271 and 272:
THE ACYLOINS 265 carbonate. This pr
- Page 273 and 274:
THE ACYLOINS 267 is thought that py
- Page 275 and 276:
CHAPTER 6 THE SYNTHESIS OF BENZOINS
- Page 277 and 278:
THE SYNTHESIS OF BENZOINS 271 are i
- Page 279 and 280:
THE SYNTHESIS OF BENZOINS 273 metri
- Page 281 and 282:
THE SYNTHESIS OF BENZOINS 275 Symme
- Page 283 and 284:
THE SYNTHESIS OF BENZOINS 277 cent
- Page 285 and 286:
THE SYNTHESIS OF BENZOINS 279 Since
- Page 287 and 288:
Benzoin Reaction 1 * 54 Benzoin Ben
- Page 289 and 290:
LESS STABLE V C6H5COCHOHC6H4OCH3^ V
- Page 291 and 292:
THE SYNTHESIS OF BENZOINS 285 66-86
- Page 293 and 294:
THE SYNTHESIS OF BENZOINS 287 Aliph
- Page 295 and 296:
THE SYNTHESIS OF BENZOINS 289 the i
- Page 297 and 298:
THE SYNTHESIS OF BENZOINS 291 The e
- Page 299 and 300:
THE SYNTHESIS OF BENZOINS 293 effec
- Page 301 and 302:
THE SYNTHESIS OF BENZOINS 295 The e
- Page 303 and 304:
THE SYNTHESIS OF BENZOINS 297 ethox
- Page 305 and 306:
THE SYNTHESIS OF BENZOINS 299 C6HBC
- Page 307 and 308:
Formula CI5HHO2 CI6HHO3 CI6HHO3 CI5
- Page 309 and 310:
Formula C19H22O2 C20H1GO2 C20H24O2
- Page 311 and 312:
CHAPTER 6 SYNTHESIS OF BENZOQUINONE
- Page 313 and 314:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 315 and 316:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 317 and 318:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 319 and 320:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 321 and 322:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 323 and 324:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 325 and 326:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 327 and 328:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 329 and 330:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 331 and 332:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 333 and 334:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 335 and 336:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 337 and 338:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 339 and 340:
SYNTHESIS OF BENZOQUINpNES BY OXIDA
- Page 341 and 342:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 343 and 344:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 345 and 346:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 347 and 348:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 349 and 350:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 351 and 352:
Quinone 5,6-Dimethoxy-2hydi oxyquin
- Page 353 and 354:
Quinone 3-Hydroxythymoqui- J none |
- Page 355 and 356:
Quinone 2-Methoxy-6-tridecylquinone
- Page 357 and 358:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 359 and 360:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 361 and 362:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 363 and 364:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 365 and 366:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 367 and 368:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 369 and 370:
ROSENMUND REDUCTION 363 For accompl
- Page 371 and 372:
ROSENMUND REDUCTION 365 acid in alm
- Page 373 and 374:
ROSENMUND REDUCTION 367 The Hydroge
- Page 375 and 376:
ROSENMUND REDUCTION 369 solution of
- Page 377 and 378:
ROSENMUND REDUCTION 371 TABLE I ALI
- Page 379 and 380:
Acid Chloride Benzoyl chloride p-Ca
- Page 381 and 382:
Acid Chloride 3-Furoic- 4~Carbometh
- Page 383 and 384:
ROSENMUND REDUCTION 377 106 Shoesmi
- Page 385 and 386:
THE WOLFF-KISHNER REDUCTION 379 INT
- Page 387 and 388:
THE W0LFF-KISHNER REDUCTION 381 mol
- Page 389 and 390:
THE WOLFF-KISHNER REDUCTION 383 dan
- Page 391 and 392:
THE WOLFF-KISHNER REDUCTION 385 Sod
- Page 393 and 394:
THE WOLFF-KISHNER REDUCTION 387 TAB
- Page 395 and 396:
THE WOLFF-KISHNER REDUCTION 389 Red
- Page 397 and 398:
THE WOLFF-KISHNER REDUCTION 391 Dir
- Page 399 and 400:
C7Hi2O C7H14O C7H0O2 C7H8O2 C7H6O4
- Page 401 and 402:
C9Hi0O C9Hi2O C9Hi4O Formula C9Hi6O
- Page 403 and 404:
Formula Ci0Hi6O CioHisO Ci0H6O2 Ci0
- Page 405 and 406:
Formula CnH16O CnHi8O CnHi6O2 CnHi8
- Page 407 and 408:
Formula Ci3H22O C13H10O3 Ci3Hi8O3 C
- Page 409 and 410:
Formula CI6HHO2 CI6HHO3 CI6H24O3 C1
- Page 411 and 412:
Formula Ci8H13ON Ci8H2IO3N Ci8Hi9O4
- Page 413 and 414:
Formula C2IH34O C21H30O2 C21H32O2 C
- Page 415 and 416:
Formula C23H32O7 C23Hi7ON C24H3603
- Page 417 and 418:
Formula C25H40O7 C26Hi6O C26H44O C2
- Page 419 and 420:
Formula C29H46O C29H46O2 C29H48O3 C
- Page 421 and 422:
Formula C32H52O3 C32H46O4 C32H50O4
- Page 423 and 424:
THE WOLFF-KISHNER REDUCTION 417 133
- Page 425 and 426:
THE WOLFF-KISHNER REDUCTION 419 235
- Page 427 and 428:
THE WOLFF-KISHNER REDUCTION 421 331
- Page 429 and 430:
DEX Numbers in bold-face type r :ef
- Page 431 and 432:
Dialkylaminoquinonedisulfonates, 35
- Page 433 and 434:
3-Methylpyrene, preparation by Wolf