Antiamoebic drugs for treating amoebic colitis - The Cochrane Library
Antiamoebic drugs for treating amoebic colitis - The Cochrane Library
Antiamoebic drugs for treating amoebic colitis - The Cochrane Library
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
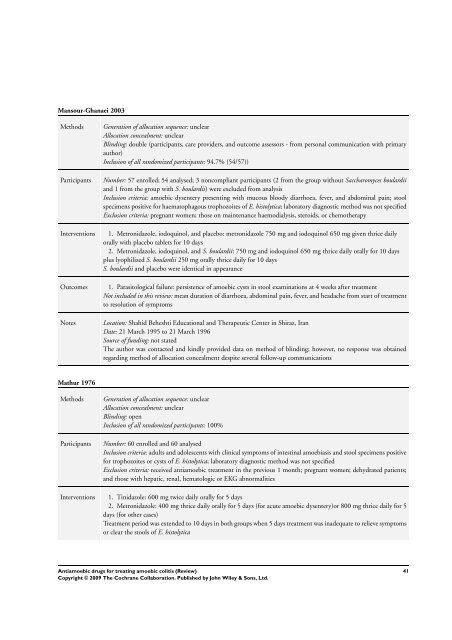
Mansour-Ghanaei 2003<br />
Methods Generation of allocation sequence: unclear<br />
Allocation concealment: unclear<br />
Blinding: double (participants, care providers, and outcome assessors - from personal communication with primary<br />
author)<br />
Inclusion of all randomized participants: 94.7% (54/57))<br />
Participants Number: 57 enrolled; 54 analysed; 3 noncompliant participants (2 from the group without Saccharomyces boulardii<br />
and 1 from the group with S. boulardii) were excluded from analysis<br />
Inclusion criteria: <strong>amoebic</strong> dysentery presenting with mucous bloody diarrhoea, fever, and abdominal pain; stool<br />
specimens positive <strong>for</strong> haematophagous trophozoites of E. histolytica; laboratory diagnostic method was not specified<br />
Exclusion criteria: pregnant women; those on maintenance haemodialysis, steroids, or chemotherapy<br />
Interventions 1. Metronidazole, iodoquinol, and placebo: metronidazole 750 mg and iodoquinol 650 mg given thrice daily<br />
orally with placebo tablets <strong>for</strong> 10 days<br />
2. Metronidazole, iodoquinol, and S. boulardii: 750 mg and iodoquinol 650 mg thrice daily orally <strong>for</strong> 10 days<br />
plus lyophilized S. boulardii 250 mg orally thrice daily <strong>for</strong> 10 days<br />
S. boulardii and placebo were identical in appearance<br />
Outcomes 1. Parasitological failure: persistence of <strong>amoebic</strong> cysts in stool examinations at 4 weeks after treatment<br />
Not included in this review: mean duration of diarrhoea, abdominal pain, fever, and headache from start of treatment<br />
to resolution of symptoms<br />
Notes Location: Shahid Beheshti Educational and <strong>The</strong>rapeutic Center in Shiraz, Iran<br />
Date: 21 March 1995 to 21 March 1996<br />
Source of funding: not stated<br />
<strong>The</strong> author was contacted and kindly provided data on method of blinding; however, no response was obtained<br />
regarding method of allocation concealment despite several follow-up communications<br />
Mathur 1976<br />
Methods Generation of allocation sequence: unclear<br />
Allocation concealment: unclear<br />
Blinding: open<br />
Inclusion of all randomized participants: 100%<br />
Participants Number: 60 enrolled and 60 analysed<br />
Inclusion criteria: adults and adolescents with clinical symptoms of intestinal amoebiasis and stool specimens positive<br />
<strong>for</strong> trophozoites or cysts of E. histolytica; laboratory diagnostic method was not specified<br />
Exclusion criteria: received anti<strong>amoebic</strong> treatment in the previous 1 month; pregnant women; dehydrated patients;<br />
and those with hepatic, renal, hematologic or EKG abnormalities<br />
Interventions 1. Tinidazole: 600 mg twice daily orally <strong>for</strong> 5 days<br />
2. Metronidazole: 400 mg thrice daily orally <strong>for</strong> 5 days (<strong>for</strong> acute <strong>amoebic</strong> dysentery)or 800 mg thrice daily <strong>for</strong> 5<br />
days (<strong>for</strong> other cases)<br />
Treatment period was extended to 10 days in both groups when 5 days treatment was inadequate to relieve symptoms<br />
or clear the stools of E. histolytica<br />
<strong>Anti<strong>amoebic</strong></strong> <strong>drugs</strong> <strong>for</strong> <strong>treating</strong> <strong>amoebic</strong> <strong>colitis</strong> (Review)<br />
Copyright © 2009 <strong>The</strong> <strong>Cochrane</strong> Collaboration. Published by John Wiley & Sons, Ltd.<br />
41