Chem 450—Advanced Organic Chemistry Exam 4
Chem 450—Advanced Organic Chemistry Exam 4
Chem 450—Advanced Organic Chemistry Exam 4
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
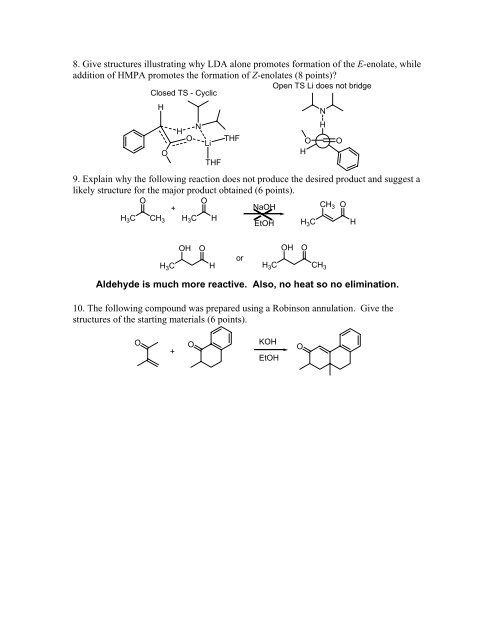
8. Give structures illustrating why LDA alone promotes formation of the E-enolate, while<br />
addition of HMPA promotes the formation of Z-enolates (8 points)?<br />
Open TS Li does not bridge<br />
Closed TS - Cyclic<br />
H<br />
O<br />
H<br />
N<br />
O Li<br />
THF<br />
THF<br />
9. Explain why the following reaction does not produce the desired product and suggest a<br />
likely structure for the major product obtained (6 points).<br />
O<br />
+<br />
O<br />
NaOH<br />
CH3 O<br />
H3C CH3 H3C H<br />
EtOH H3C H<br />
O<br />
H<br />
OH O<br />
OH O<br />
H3C H<br />
or<br />
H3C CH3 Aldehyde is much more reactive. Also, no heat so no elimination.<br />
10. The following compound was prepared using a Robinson annulation. Give the<br />
structures of the starting materials (6 points).<br />
O<br />
+<br />
O<br />
KOH<br />
EtOH<br />
O<br />
N<br />
H<br />
O