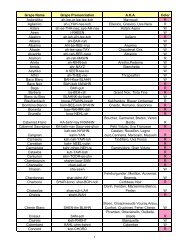
Chapter 38 Workbook Solutions
Chapter 38 Workbook Solutions
Chapter 38 Workbook Solutions
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
<strong>38</strong>-12 cHAPTER <strong>38</strong> . Quantization<br />
40. The longest wavelength in the Balmer series is 656 nm.<br />
a, What transition is this?<br />
3-) )<br />
b. If light of this wavelength shines on a container of hydrogen atoms, will the light be absorbed? Why<br />
or why not?<br />
No. Etse^*,o.\[1 a\\ of t1.," q*ovns in o.<br />
hf{"o5o" .3"t r:t:Jt<br />
thc e,f o.,.nj sta:tc n= [ . J\tcfc. ov. v\o e\eitronS ln tlo n<br />
st..{s 'l;{ ;-(A-o,brocb'+L; o*.ig1 -o,*J<br />
i*r To lho n=3<br />
stdc,<br />
41 . a. How many electrons, protons, and neutrons are in a hydrogen-like 12 C ion ?<br />
(" pr o{ons, G nqv$ron s