Solutions Packet
Solutions Packet
Solutions Packet
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
1 <strong>Solutions</strong>
2 <strong>Solutions</strong>
3 <strong>Solutions</strong>
4 <strong>Solutions</strong>
5 <strong>Solutions</strong>
6 <strong>Solutions</strong>
7 <strong>Solutions</strong>
8 <strong>Solutions</strong>
9 <strong>Solutions</strong>
10 <strong>Solutions</strong>
11 <strong>Solutions</strong>
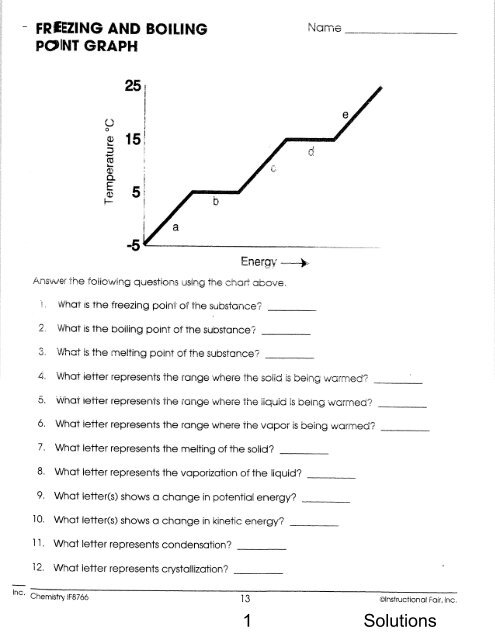
Chemistry: Form WS8.1.1A Name ______________________________<br />
SOLUTIONS Date _________________ Period _____<br />
What’s in the Water?<br />
A solution is a type of mixture. It consists of two or more kinds of matter<br />
each of which retains its own properties. But it is homogeneous. It appears<br />
to be only one substance. This is what distinguishes solutions from<br />
mechanical mixtures. The substance that is dissolved is the solute. The<br />
solvent is the substance that dissolves the solute. It is the continuous phase.<br />
For example, salt dissolved in water appears to be a liquid.<br />
Different solutes dissolve best in different solvents. In order for a<br />
solvent to dissolve a solute, it must exert forces of attraction on the solute.<br />
Polar solvents such as water dissolve polar and ionic solutes well because<br />
they exert mutual attractions that cause their particles to intermingle. Of<br />
course, not all ionic substances dissolve equally well in water. (See Table<br />
F - Table of Solubilities in Water) Nonpolar solvents do NOT dissolve<br />
polar and ionic substances because there is no attraction between them. For<br />
example, oil and water do NOT mix. Nonpolar substances such as fat<br />
dissolve in nonpolar solvents such as benzene because the forces of<br />
attraction are too weak to prevent the particles from freely intermingling.<br />
Answer the questions below based on your reading and on your<br />
knowledge of chemistry.<br />
1. Water is mixed with sugar, resulting in a transparent, colorless liquid.<br />
a. What evidence will there be that this is a mixture rather than a new compound?<br />
b. What evidence shows it is a solution rather than a mechanical mixture?<br />
c. Which is the solute, and which is the solvent? How do you know?<br />
2. Why does table salt dissolve in water, while oil and water don’t mix?<br />
© Evan P. Silberstein, 2003<br />
Water discusses its tastes.<br />
3. Based on Table F indicate which of the following compounds is water soluble and which is insoluble?<br />
a. Li 2CO 3 _______________<br />
b. Fe(OH) 3 _______________<br />
c. CaCrO 4 _______________<br />
d. BaS _______________<br />
e. (NH 4) 3PO 4 _______________<br />
f. Al(ClO 3) 3 _______________<br />
g. PbSO 4 _______________<br />
h. NaOH _______________<br />
i. CuSO 4 _______________<br />
j. KNO 3 _______________<br />
k. AgCl _______________<br />
l. Ba(HCO 3) 2 _______________<br />
12 <strong>Solutions</strong>
Chemistry: Form WS8.1.2A Name ______________________________<br />
SOLUTIONS Date _________________ Period _____<br />
Solubility Curves<br />
The solubility of solid solutes generally increases as temperature increases, while<br />
the solubility of gaseous solutes generally decreases as temperature increases. A<br />
solution that holds as much solute as can dissolve at a given temperature is<br />
saturated. A solution that can dissolve more solute at a given temperature is<br />
unsaturated. A solution that holds more solute than can dissolve at a given<br />
temperature is supersaturated. The amount of solute that is needed to form a<br />
saturated solution at various temperatures can be graphed. This is what is shown<br />
in Table G. The values in Table G are based on solute dissolved in 100 g of water.<br />
Since water has a density of 1 g/mL, the graph can be considered to be based on<br />
100 mL of water. A 200 mL sample of water would be able to dissolve twice as<br />
much at each temperature.<br />
Answer the questions below by referring to Table G.<br />
1. The compound which is the most soluble at 20°C is .<br />
2. The compound which is the least soluble at 10°C is .<br />
3. The compound which is the least soluble at 80°C is .<br />
4. The number of grams of potassium nitrate needed to saturate 100 mL of water<br />
at 70°C is .<br />
5. The formulas of the compounds which vary inversely with the temperature are ,<br />
and .<br />
6. One hundred mL of a sodium nitrate solution is saturated at 10°C. How many additional grams are needed to saturate<br />
the solution at 50°C?<br />
7. One hundred mL of a saturate KCl solution at 80°C will precipitate 10 grams of salt when cooled to what temperature?<br />
8. The two salts that have the same degree of solubility at 70°C are and .<br />
9. The salt with a solubility is least affected by a change in temperature is .<br />
10. The salt that has the greatest increase in solubility in the temperature range between 30°C and 50°C is .<br />
11. The number of grams of sodium nitrate that must be added to 50 mL of water to produce a saturated solution at 50°C is<br />
12. A saturated solution of potassium chlorate is made at 10°C by dissolving the correct mass of salt in 100 mL of water.<br />
When the solution is heated to 90°C, how many grams must be added to saturate the solution?<br />
Continue L<br />
13 <strong>Solutions</strong><br />
.
Chemistry: Form WS8.1.2A Solubility Curves<br />
SOLUTIONS Page 2<br />
13. At what temperature do saturated solutions of sodium chloride and potassium chloride contain the same mass of solute<br />
per 100 mL of water?<br />
14. A saturated solution of potassium nitrate is prepared at 60°C using 200 mL of water. If the solution is cooled to 30°C,<br />
how many grams will precipitate out of the solution?<br />
15. How many more grams of ammonia can be dissolved in 100 mL of water at 10°C than at 90°C?<br />
16. A saturated solution of sodium nitrate in 100 mL of water at 40°C is heated to 50°C. The rate of increase in solubility<br />
grams per degree is .<br />
17. Thirty grams of KCl is dissolved in 100 mL of water at 45°C. The number of additional grams of KCl that would be<br />
needed to make the solution saturated at 80°C is .<br />
© Evan P. Silberstein, 2003<br />
14 <strong>Solutions</strong>
Chemistry: Form WS8.1.3A Name ______________________________<br />
SOLUTIONS Date _________________ Period _____<br />
Dissolving Solids and Gases<br />
A factory releases clean, warm water into a stream. The stream becomes<br />
severely polluted as a result. How does this happen? Fish living in the water<br />
depend on dissolved oxygen in order to breathe. Like other gases, oxygen<br />
molecules tend to spread out. In order to dissolve them, it is necessary to<br />
confine them. Heat speeds the molecules up and makes them spread out<br />
more–exactly the opposite of what is needed to dissolve them. As a result,<br />
heat drives the oxygen out of the water, causing the fish to die. The dead<br />
fish begin to decay. Growing decay bacteria deplete the water of oxygen<br />
even further. In this way, clean warm water can pollute a stream. The<br />
process of dissolving gases is opposite to the process of dissolving solids<br />
because of the differences between gases and solids.<br />
Answer the questions below based on your reading above and on your knowledge of chemistry.<br />
1. A warm can of soda is dropped and bounces down a flight of stairs. When it is opened, carbon dioxide gas<br />
coming out of solution causes it to spray all over. Explain the affect of each of the following:<br />
a. The fact that the soda was warm.<br />
b. The fact that the soda was dropped and bounced down a flight of stairs.<br />
c. The fact that the can was opened.<br />
2. When a gas dissolves, the particles need to be confined. What do the particles of a solid need to do in order to<br />
dissolve?<br />
3. Sugar is added to a hot cup of coffee and stirred. The sugar dissolves. Explain the affect of each of the<br />
following:<br />
a. The fact that the coffee was hot.<br />
b. The fact that the coffee was stirred.<br />
Continue L<br />
15 <strong>Solutions</strong>
Chemistry: Form WS8.1.3A Dissolving Solids and Gases<br />
SOLUTIONS Page 2<br />
4. Which dissolves faster, a teaspoon of sugar or a sugar cube? Why?<br />
5. A solid is added to water and stirred. Some of it dissolves, but not all. What happens to the rate at which the<br />
solid is dissolving between when it was first added and when it stopped dissolving? Explain. (HINT:<br />
Equilibrium!)<br />
6. The table below lists four factors that may effect the rate at which solids and gases dissolve. Fill in the table by<br />
indicating if the rate of dissolving increases, decreases, or is not effected. Then explain why.<br />
Crushing<br />
Stirring<br />
Factor<br />
Increasing the<br />
amount of dissolved<br />
solute<br />
Increasing<br />
Temperature<br />
Affect on Rate of Solution for:<br />
Solid Solutes Gaseous Solutes<br />
© Evan P. Silberstein, 2003<br />
16 <strong>Solutions</strong>
Chemistry: Form WS8.2.1A Name ______________________________<br />
SOLUTIONS Date _________________ Period _____<br />
Finding Concentration<br />
The directions on a can of condensed soup say to mix the can of soup with one can<br />
of water. What would happen to the flavor if it were mixed with two or three cans<br />
of water instead? When two substances are mixed, the amount of one compared to<br />
the amount of the other is known as the concentration. Adding extra water makes<br />
the concentration of the soup lower than what is called for in the recipe–and it tastes<br />
it! There are several ways of measuring concentration–mass per unit volume,<br />
percentage by mass, percentage by volume, and parts per million (ppm). See the<br />
examples below:<br />
Massof solute( g)<br />
Concentration =<br />
Volumeof Solvent or Solution( mL)<br />
Sample Problem<br />
What is the concentration of a solution prepared by<br />
dissolving 25 g of KNO 3 in 100. mL of water?<br />
25g<br />
Concentration = = 025 .<br />
100.<br />
ml<br />
mass( solute)<br />
percent mass = ×<br />
mass( solution)<br />
100%<br />
g<br />
mL<br />
Sample Problem<br />
What is the percent by mass of a solution containing 12.3<br />
g of caffeine dissolved in 100.0 g of water?<br />
Step 1: Find the mass of the solution<br />
100.0 g + 12.3 g = 112.3 g<br />
Step 2: Divide the mass of the solute by the mass of the<br />
solution and multiply by 100 %<br />
12. 3g<br />
percent mass = × 100% = 110% .<br />
112. 3g<br />
Continue L<br />
mass( solute)<br />
ppm = × 1, 000, 000ppm<br />
mass( solution)<br />
Sample Problem<br />
About 0.0047 g of ammonia are dissolved in 20.0 g of<br />
water. Express this in parts per million.<br />
Step 1: Find the mass of the solution<br />
20.0 g + 0.0047 g = 20.0047 g<br />
Step 2: Divide the mass of the solute by the mass of the<br />
solution and multiply by 1,000,000 ppm.<br />
0. 0047g<br />
ppm = × 1, 000, 000ppm = 235ppm<br />
20. 0047g<br />
volume ( solute)<br />
percent volume = ×<br />
volume ( solution)<br />
100%<br />
Sample Problem<br />
What is the percent by volume of a solution containing<br />
18.2 mL of glycerine (C 3H 6O 3) dissolved in 85.0 mL of<br />
water?<br />
Step 1: Find the volume of the solution.<br />
18.2 mL + 85.0 mL = 103.2 mL<br />
Step 2: Divide the volume of the solute by the volume of<br />
the solution and multiply by 100%<br />
18. 2mL<br />
percent volume = × 100% = 17. 6%<br />
1032 . mL<br />
17 <strong>Solutions</strong>
Chemistry: Form WS8.2.1A Finding Concentration<br />
SOLUTIONS Page 2<br />
Answer the questions below based on the sample problems.<br />
1. What is the concentration of 45 mL of a solution<br />
containing 9.0 g of KClO 3?<br />
2. A solution is prepared by mixing 20.0 g of NaNO 3 with<br />
100. mL of water. What is the percentage mass of the<br />
solution? (Assume density of water is 1 g / mL)<br />
3. A 250. mL sample of air at STP contains approximately<br />
52.5 mL of O 2(g). What is the percentage of oxygen in<br />
air?<br />
4. A polar solvent is prepared by mixing 27.5 mL of<br />
propanone with 222.5 mL of water. What is the<br />
percentage by volume of propanone in the mixture?<br />
5. How many parts per million of sulfur dioxide are there<br />
in a solution containing 0.065 g of sulfur dioxide in<br />
5,000 mL of water? (Assume density of water is 1 g / mL)<br />
© Evan P. Silberstein, 2003<br />
6. If 19 mL of alcohol are dissolved in 31 mL of water,<br />
what is the percentage by volume of alcohol?<br />
7. If 0.002 g of PbCl 2 are dissolved in 2.0 L of water, how<br />
many parts per million are dissolved? (Assume density<br />
of water is 1 g / mL)<br />
8. If 15 g of KNO 3 are dissolved in 235 g of water, what is<br />
the percentage of solute by mass?<br />
9. What is the percentage by mass of a solution prepared<br />
with 34 g of KI and 126 g of water?<br />
10. What is the concentration of a solution made with<br />
0.056 g of CO 2(g) and 200 mL of water?<br />
18 <strong>Solutions</strong>
Chemistry: Form WS8.2.2A Name ______________________________<br />
SOLUTIONS Date _________________ Period _____<br />
Molarity<br />
One of the most useful measures of concentration in chemistry is molarity (M). Molarity is the<br />
number of moles of solute per liter of solution. A two molar (2 M) solution contains two moles of<br />
solute per liter of solution.<br />
moles( solute)<br />
M =<br />
L( solution)<br />
Recall that the number of moles is determined by dividing the number of grams by the gram formula<br />
mass (GFM). There are a number of formulas for calculation that come from these relationships:<br />
•<br />
g<br />
• moles = M × L • g = M × GFM × L<br />
M =<br />
GFM × L<br />
Below are some sample problems that show how to apply these formulas.<br />
Sample Problem 1<br />
Find the molarity of 100. mL of a solution that contains<br />
0.25 moles of dissolved solute.<br />
Step 1: Convert all volumes to liters<br />
0001 . L<br />
100.<br />
mL × = 0100 . L<br />
1mL<br />
Step 2: Substitute values into the definitional equation<br />
M mol 025 . mol<br />
= = = 25 . M<br />
L 0100 . L<br />
Sample Problem 3<br />
How many moles of solute are dissolved in 250. mL of a<br />
3.0 M solution?<br />
Step 1: Convert all volumes to liters<br />
0. 001L<br />
250.<br />
mL × = 0250 . L<br />
1mL<br />
Step 2: Substitute values into the correct equation<br />
mol = M × L = 30 . 0. 250L = 0. 75mol<br />
mol ( L)(<br />
)<br />
A two molar solution<br />
Sample Problem 2<br />
Find the molarity of 500. mL of a solution that contains<br />
4.9 g of dissolved sulfuric acid (H 2SO 4).<br />
Step 1: Find the GFM<br />
H = 1 × 2 = 2<br />
S = 32 × 1 = 32<br />
O = 16 × 4 = 64<br />
98<br />
Step 2: Convert all volumes to liters<br />
0001 . L<br />
500.<br />
mL × = 0500 . L<br />
1mL<br />
Step 3: Substitute values into the correct equation<br />
g<br />
M = =<br />
GFM × L 98<br />
49 . g<br />
0500 . L<br />
= 010 . M<br />
g ( mol)(<br />
)<br />
Sample Problem 4<br />
How many grams of sodium carbonate(Na 2CO 3) are<br />
needed to prepare 250 mL of a 0.10 M solution?<br />
Step 1: Find the GFM<br />
Na = 23 × 2 = 46<br />
C = 12 × 1 = 12<br />
O = 16 × 3 = 48<br />
106<br />
Step 2: Convert all volumes to liters<br />
0. 001L<br />
250.<br />
mL × = 0250 . L<br />
1mL<br />
Step 3: Substitute values into the correct equation<br />
g = M × L× GFM = 010 . 106 0. 250L = 2. 7g<br />
mol g ( L)(<br />
mol)(<br />
)<br />
19 <strong>Solutions</strong>
Chemistry: Form WS8.2.2A Molarity<br />
SOLUTIONS Page 2<br />
Answer the questions below based on the reading and the sample problems on the previous page.<br />
1. Determine the molarity of 500. mL of a solution with<br />
0.35 mol of dissolved solute.<br />
2. A 200. mL sample of a solution contains 4.0 g of NaOH.<br />
What is its molarity?<br />
3. How many grams of KNO 3 are needed to prepare 25 mL<br />
of a 2.0 M solution?<br />
4. How many moles of MgSO 4 are contained in 50. mL of<br />
a 3.0 M solution?<br />
5. How many grams of CaCl 2 are dissolved in 80.0 mL of<br />
a 0.75 M solution?<br />
© Evan P. Silberstein, 2003<br />
6. What is the molarity of 300 mL of a solution that<br />
contains 0.60 mol of dissolved ammonia?<br />
7. What is the molarity of 5.0 L of a solution containing<br />
200. g of dissolved CaCO 3?<br />
8. How many grams of NaCl are needed to prepare 500.<br />
mL of a 0.400 M solution?<br />
9. How many moles of solute are contained in 3.0 L of a<br />
1.5 M solution?<br />
10. What is the molarity of 750 mL of a solution that<br />
contains 40.0 g of dissolved CuSO 4?<br />
20 <strong>Solutions</strong>
Chemistry: Form WS8.3.1A Name ______________________________<br />
SOLUTIONS Date _________________ Period _____<br />
Understanding Colligative Properties<br />
After a winter storm, people spread salt on the walks to help melt the ice.<br />
Salt reduces the freezing point of water. Actually, any soluble solute<br />
reduces the freezing point of water by interfering with crystallization. In<br />
this way, antifreeze keeps the water from freezing in an automobile<br />
radiator. This phenomenon is called freezing point depression. Antifreeze<br />
is left in the radiator during the summer. It also prevents the radiator from<br />
boiling over by raising the boiling point. Dissolved solute reduces the<br />
vapor pressure, raising the boiling point. This is called boiling point<br />
elevation.<br />
The amount the freezing point<br />
is depressed or the boiling<br />
point is raised depends on the<br />
concentration of dissolved<br />
solute. The higher the Dad misinterprets freezing point depression.<br />
concentration of dissolved<br />
solute is, the greater the effect on the boiling point or the freezing point is. Only the<br />
concentration of the particles of dissolved solute is important. The nature of the<br />
solute is not. A mole of dissolved sugar has exactly the same effect on the freezing<br />
point and boiling point of 1,000 g of water as a mole of antifreeze because it<br />
contains the same number of particles. Ionic compounds dissociate producing<br />
more particles per mole. One mole of dissolved sodium chloride, for example, produces one mole of aqueous sodium ions and<br />
one mole of aqueous chloride ions for a total of two moles [NaCl(s) ÿ Na + (aq) + Cl – (aq)]. One mole of dissolved sodium<br />
chloride, therefore, has twice the effect on the boiling and freezing points of 1,000 g of water as one mole of dissolved sugar.<br />
It is not the nature of the solute that matters, but only the concentration of dissolved particles that determines how large the<br />
change in freezing point or boiling point will be. Properties of a solution, such as this, which are dependent only on the number<br />
of particles in solution, and not on their nature are called colligative properties.<br />
Answer the questions below based on your reading and on your knowledge of chemistry.<br />
1. Why are boiling point elevation and freezing point depression considered colligative properties?<br />
2. Why is salt put on icy roads and sidewalks in the winter?<br />
3. How will the boiling points of pure water and sea water compare? Why?<br />
Continue L<br />
21 <strong>Solutions</strong>
Chemistry: Form WS8.2.3A Name ______________________________<br />
SOLUTIONS Date _________________ Period _____<br />
Molality<br />
There are two basic ways to prepare solutions quantitatively. Keep in mind that<br />
solute takes up space. Even when the solute is completely dissolved, it affects the<br />
volume of the solution. Preparing solutions based on measuring the amount of<br />
solute per volume of solution is useful, because it is possible to measure out a<br />
sample of the solution and figure out how much solute you have. This is the type<br />
of measure that molarity is. Understanding how solute affects the solution,<br />
however, requires a different type of measure. To compare the solubility of<br />
different substances, it is necessary to know how much solvent is used compared<br />
to solute. To understand how dissolved solute affects the freezing point or boiling<br />
point of a liquid, it is necessary to know how much liquid solvent you have for a<br />
given amount of solute. Molality is very similar to molarity, except that it compares<br />
the moles of solute to kilograms of solvent instead of liters of solution. It is<br />
abbreviated by lower case m.<br />
mol( solute)<br />
m =<br />
kg( solvent)<br />
Sample Problem 1<br />
Find the molality of a solution that contains 0.45 moles of solute<br />
dissolved in 300. g of water .<br />
Step 1: Convert the amount of solvent to kilograms<br />
1kg<br />
300.<br />
g × = 0. 300kg<br />
1000g<br />
Step 2: Substitute values into the definitional equation<br />
045 . mol<br />
m = = 15 . m<br />
0300 . kg<br />
Answer the questions below.<br />
1. Find the molality of a solution that<br />
contains 225 g of Ca(NO 3) 2<br />
dissolved in 400. g of water.<br />
2. Find the molality of a solution that<br />
contains 0.663 mol of solute<br />
dissolved in 300. g of water.<br />
Competition among molar and molal<br />
solutions<br />
Sample Problem 2<br />
Find the molality of a solution that contains 25.57 g of sodium<br />
chloride dissolved in 250. g of water.<br />
Step 1: Find the GFM<br />
Na = 22.99 × 1 = 22.99<br />
Cl = 35.45 × 1 = 35.45<br />
58.44<br />
Step 2: Convert the mass of solute to moles.<br />
⎛ 1mol<br />
⎞<br />
( 2557 . g)<br />
⎜ ⎟ = 0. 4375mol<br />
⎝ 58. 44g<br />
⎠<br />
Step 3: Convert the amount of solvent to kilograms.<br />
1kg<br />
250.<br />
g × = 0. 250kg<br />
1000g<br />
Step 4: Substitute values into the definitional equation.<br />
0. 4375mol<br />
m = = 175 . m<br />
0250 . kg<br />
3. Find the molality of a solution that<br />
contains 1.25 kg of KBr dissolved<br />
in 2.45 kg of water.<br />
© Evan P. Silberstein, 2010<br />
22 <strong>Solutions</strong>
Chemistry: Form WS8.3.1A Understanding Colligative Properties<br />
SOLUTIONS Page 2<br />
Solve the following boiling point elevation problems and the freezing point depression problems as shown in the sample<br />
problems below. [NOTE: At standard pressure, 1 mol of dissolved particles will elevate the boiling point of 1,000 g of<br />
water by 0.52EC and will depress the freezing point of 1,000 g of water by 1.86EC]<br />
Sample Problem<br />
Find the boiling point of a solution containing 1,000 g<br />
of water and 2 mol of dissolved MgF 2.<br />
Step 1: Determine the number of moles of solute<br />
particles<br />
2MgF 2(s) ÿ 2Mg 2+ (aq) + 4F – (aq) mol =6<br />
Step 2: Multiply the boiling point elevation per mole by<br />
the number of moles of solute to find the boiling<br />
point elevation<br />
BPE = 0.52 EC /mol × 3 mol = 3.12EC<br />
Step 3: Add the boiling point elevation to 100EC<br />
BP = 100EC + 3.12EC = 10 3.12EC<br />
Sample Problem<br />
Find the freezing point of a solution containing 1,000 g of<br />
water and 30 g of dissolved antifreeze (C 2H 4O 2).<br />
Step 1: Determine the number of moles of solute particles<br />
C = 12 × 2 = 24 g 30g<br />
H = 1 × 4 = 4<br />
mol = = = 05 . mol<br />
g GFM 60 mol<br />
O = 16 × 2 = 32<br />
60<br />
Step 2: Multiply the freezing point depression per mole by the<br />
number of moles of solute to find the freezing point<br />
depression<br />
FPD = 1.86EC /mol × 0.5 mol = 0.93EC<br />
Step 3: Subtract the freezing point depression from 0EC<br />
FP = 0EC – 0.93EC = – 0.93EC<br />
4. One mole of dissolved particles elevates the boiling point of 1,000 g of water by 0.52EC. At standard pressure, what will<br />
the boiling point of a solution be if it contains 1,000 g of water and:<br />
a. 1 mol of antifreeze (C 2H 4O 2)? _______________<br />
b. 1 mol of salt (NaCl)? _______________<br />
c. 1 mol of ethanol (C 2H 5OH)? _______________<br />
d. 2 mol of glycerol (C 3H 6O 3)? _______________<br />
e. 2 mol of CaCl 2(aq)? _______________<br />
f. 5 mol of sucrose (C 12H 22O 11)? _______________<br />
g. 1 mol of KNO 3(aq)? _______________<br />
h. 3 mol of Ba(NO 3) 2(aq)? _______________<br />
i. 40 g of NaOH(aq)? _______________<br />
j. 270 g of glucose (C 6H 12O 6)? _______________<br />
5. One mole of dissolved particles depresses the freezing point of 1,000 g of water by1.86EC. At standard pressure, what<br />
will the freezing point of a solution be if it contains 1,000 g of water and:<br />
a. 1 mol of glucose (C 6H 12O 6)? _______________<br />
b. 1 mol of BaCl 2(aq)? _______________<br />
c. 2 mol of methanol (CH 3OH)? _______________<br />
d. 3 mol of glycerol (C 3H 6O 3)? _______________<br />
e. 2 mol of CuSO 4(aq)? _______________<br />
© Evan P. Silberstein, 2003<br />
f. 4 mol of sucrose (C 12H 22O 11)? _______________<br />
g. 3 mol of KNO 3(aq)? _______________<br />
h. 2 mol of salt (NaCl)? _______________<br />
i. 150 g of KHCO 3(aq)? _______________<br />
j. 180 g of glucose (C 6H 12O 6)? _______________<br />
23 <strong>Solutions</strong>
Name: _______________________________<br />
Solubility Practice Sheet<br />
1. If 50g of water is saturated with Potassium chlorate at 23°C is evaporated, how<br />
many grams of the salt will be left?<br />
2. What is the smallest mass of water needed to dissolve 23g of ammonium chloride<br />
at 40°C?<br />
3. At what temperature do potassium chlorate and potassium chloride have the same<br />
solubility in water?<br />
4. If you have 30g of potassium chloride already dissolved in 100g of water, how<br />
many more grams are needed to make a saturated solution at 80°C?<br />
5. Which substances decrease in solubility as the temperature increases? Why?<br />
6. Which substance’s solubility is least affected by temperature changes?<br />
7. How much potassium nitrate will dissolve in 300g of water at 40°C?<br />
8. If 400g of water is mixed with 250g of ammonium chloride and is then cooled to<br />
25°C, how many grams will precipitate out of the solution?<br />
24 <strong>Solutions</strong>
Regents Chemistry Review: <strong>Solutions</strong><br />
Solution: a homogenous mixture of substances in the same physical state. <strong>Solutions</strong> contain atoms, ions or molecules of one substance<br />
spread uniformly throughout a second substance. <strong>Solutions</strong> will pass through filter paper and will not settle on standing.<br />
Solute: the substance that is being dissolved and the substance that is typically present in the smaller amount<br />
Solvent: the substance that dissolves the solute, and is typically present in the greater amount.<br />
Solubility: the amount of solute that will dissolve at a certain temperature ( Table G )<br />
Table F tells if a compound is soluble or insoluble based on the ions that make it up.<br />
Factors that determine solubility:<br />
- Nature of solute and solvent: “like dissolves like”<br />
polar dissolves polar, nonpolar dissolves nonpolar<br />
- Temperature- Increase temperature, increase solubility of solids,<br />
Increase temperature, decrease the solubility of gases<br />
- Pressure- only affects gases; as Pressure increases, increase the<br />
solubility of gases in liquids.<br />
Reading Table G: Seven Salts, Three Gases!<br />
Unsaturated: below the curve; a point where the solution holds less<br />
solute than the maximum it can hold<br />
Saturated: on the curve: a solution that contains a maximum amount of solute<br />
that will dissolve at a specific temperature; any additional solute will<br />
not dissolve, it will simply settle to the bottom of the container.<br />
Supersaturated: above the curve: a solution that holds more solute than is present in a<br />
saturated solution at that temperature. These are unstable and temporary .<br />
Concentration of <strong>Solutions</strong>; use Table T to find the formulas<br />
Molarity: the number of moles of solute in 1 L of solution. (Concentration)<br />
**Solution must be in Liters!<br />
Percent by Mass: the mass of an ingredient divided by the total mass, expressed as a percent.<br />
Percent by Volume: the ratio of the volume of an ingredient divided by the total volume and expressed as a percent.<br />
Parts per million: a ratio between the mass of solute and the total mass of solution. The only difference between ppm and<br />
percent by mass is that you multiply by 1,000,000 instead of 100<br />
Preparing a solution of known concentration: Add solute to a small amount of solvent and then top off the solvent to make the given volume of<br />
the solution. First add sufficient grams to the flask, and then add distilled water until the solute is dissolved and thoroughly mixed. Finally, fill with<br />
distilled water to the mark on the neck of the flask, and again stir to make sure the solution is homogeneous. **Remember how we made Kool-aide**<br />
Colligative Properties : the boiling and the freezing point of water change when nonvolatile solutes are added.<br />
Covalent molecules do NOT separate in solution, so they do not lower the freezing point as much as ionic<br />
compounds. The more ions in the solution, the lower the freezing point, the higher the boiling point.<br />
Freezing point depression: when you add moles of particles, the freezing point gets lower<br />
3.0 M MgCl2 lowers the freezing point more than does 3.0 M NaCl because MgCl 2 ionizes into 3 moles of ions, as<br />
opposed to NaCl which only ionizes into 2 moles of ions. 3.0 M C 6H 12O 6 lowers the freezing point, but not as<br />
much as the ionic compounds because covalent molecules do not ionize at all.<br />
Boiling point elevation: when you add moles of particles, the boiling point gets higher<br />
3.0 M MgCl2 increases the boiling point more than does 3.0 M NaCl because MgCl 2 ionizes into 3 moles of ions, as<br />
opposed to NaCl which only ionizes into 2 moles of ions. 3.0 M C6H 12O 6 lowers the freezing point, but not as<br />
much as the ionic compounds because covalent molecules do not ionize at all.<br />
25 <strong>Solutions</strong>
26 <strong>Solutions</strong>