application of x-ray diffraction and scanning electron ... - OpenDrive
application of x-ray diffraction and scanning electron ... - OpenDrive
application of x-ray diffraction and scanning electron ... - OpenDrive
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
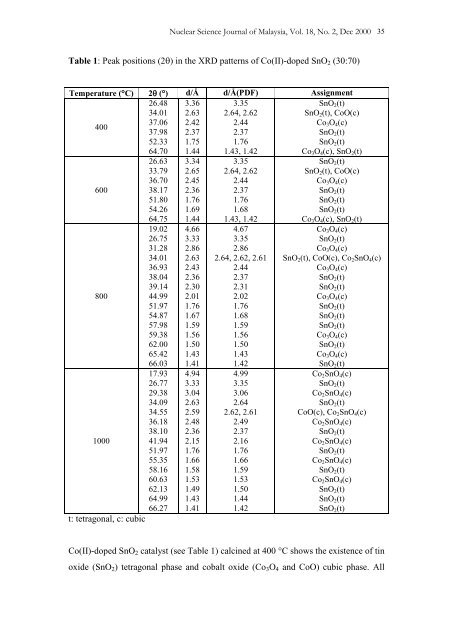
Nuclear Science Journal <strong>of</strong> Malaysia, Vol. 18, No. 2, Dec 2000 35<br />
Table 1: Peak positions (2) in the XRD patterns <strong>of</strong> Co(II)-doped SnO 2 (30:70)<br />
Temperature (C) 2 () d/Å d/Å(PDF) Assignment<br />
26.48<br />
34.01<br />
3.36<br />
2.63<br />
3.35<br />
2.64, 2.62<br />
SnO 2 (t)<br />
SnO 2 (t), CoO(c)<br />
400<br />
37.06 2.42 2.44<br />
Co 3 O 4 (c)<br />
37.98 2.37 2.37<br />
SnO 2 (t)<br />
52.33<br />
64.70<br />
1.75<br />
1.44<br />
1.76<br />
1.43, 1.42<br />
SnO 2 (t)<br />
Co 3 O 4 (c), SnO 2 (t)<br />
600<br />
800<br />
1000<br />
t: tetragonal, c: cubic<br />
26.63<br />
33.79<br />
36.70<br />
38.17<br />
51.80<br />
54.26<br />
64.75<br />
19.02<br />
26.75<br />
31.28<br />
34.01<br />
36.93<br />
38.04<br />
39.14<br />
44.99<br />
51.97<br />
54.87<br />
57.98<br />
59.38<br />
62.00<br />
65.42<br />
66.03<br />
17.93<br />
26.77<br />
29.38<br />
34.09<br />
34.55<br />
36.18<br />
38.10<br />
41.94<br />
51.97<br />
55.35<br />
58.16<br />
60.63<br />
62.13<br />
64.99<br />
66.27<br />
3.34<br />
2.65<br />
2.45<br />
2.36<br />
1.76<br />
1.69<br />
1.44<br />
4.66<br />
3.33<br />
2.86<br />
2.63<br />
2.43<br />
2.36<br />
2.30<br />
2.01<br />
1.76<br />
1.67<br />
1.59<br />
1.56<br />
1.50<br />
1.43<br />
1.41<br />
4.94<br />
3.33<br />
3.04<br />
2.63<br />
2.59<br />
2.48<br />
2.36<br />
2.15<br />
1.76<br />
1.66<br />
1.58<br />
1.53<br />
1.49<br />
1.43<br />
1.41<br />
3.35<br />
2.64, 2.62<br />
2.44<br />
2.37<br />
1.76<br />
1.68<br />
1.43, 1.42<br />
4.67<br />
3.35<br />
2.86<br />
2.64, 2.62, 2.61<br />
2.44<br />
2.37<br />
2.31<br />
2.02<br />
1.76<br />
1.68<br />
1.59<br />
1.56<br />
1.50<br />
1.43<br />
1.42<br />
4.99<br />
3.35<br />
3.06<br />
2.64<br />
2.62, 2.61<br />
2.49<br />
2.37<br />
2.16<br />
1.76<br />
1.66<br />
1.59<br />
1.53<br />
1.50<br />
1.44<br />
1.42<br />
SnO 2 (t)<br />
SnO 2 (t), CoO(c)<br />
Co 3 O 4 (c)<br />
SnO 2 (t)<br />
SnO 2 (t)<br />
SnO 2 (t)<br />
Co 3 O 4 (c), SnO 2 (t)<br />
Co 3 O 4 (c)<br />
SnO 2 (t)<br />
Co 3 O 4 (c)<br />
SnO 2 (t), CoO(c), Co 2 SnO 4 (c)<br />
Co 3 O 4 (c)<br />
SnO 2 (t)<br />
SnO 2 (t)<br />
Co 3 O 4 (c)<br />
SnO 2 (t)<br />
SnO 2 (t)<br />
SnO 2 (t)<br />
Co 3 O 4 (c)<br />
SnO 2 (t)<br />
Co 3 O 4 (c)<br />
SnO 2 (t)<br />
Co 2 SnO 4 (c)<br />
SnO 2 (t)<br />
Co 2 SnO 4 (c)<br />
SnO 2 (t)<br />
CoO(c), Co 2 SnO 4 (c)<br />
Co 2 SnO 4 (c)<br />
SnO 2 (t)<br />
Co 2 SnO 4 (c)<br />
SnO 2 (t)<br />
Co 2 SnO 4 (c)<br />
SnO 2 (t)<br />
Co 2 SnO 4 (c)<br />
SnO 2 (t)<br />
SnO 2 (t)<br />
SnO 2 (t)<br />
Co(II)-doped SnO 2 catalyst (see Table 1) calcined at 400 C shows the existence <strong>of</strong> tin<br />
oxide (SnO 2 ) tetragonal phase <strong>and</strong> cobalt oxide (Co 3 O 4 <strong>and</strong> CoO) cubic phase. All