BCA table lecture
BCA table lecture
BCA table lecture
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
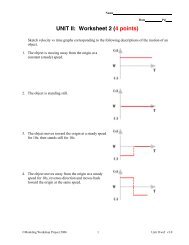
Stoichiometry 1<br />
<strong>BCA</strong> Method<br />
1.Lead will react with hydrochloric acid to<br />
produce lead chloride and hydrogen gas.<br />
How many moles of hydrochloric acid are<br />
needed to completely react with 4.0 mole<br />
of lead?<br />
Equation:<br />
Before:<br />
Change:<br />
After:<br />
1. Lead will react with hydrochloric acid<br />
to produce lead chloride and hydrogen<br />
gas. How many moles of hydrochloric<br />
acid are needed to completely react with<br />
4.0 mole of lead?<br />
1.Lead will react with hydrochloric acid to<br />
produce lead chloride and hydrogen gas.<br />
How many moles of hydrochloric acid are<br />
needed to completely react with 4.0 mole<br />
of lead?<br />
Equation:<br />
Pb +<br />
2 HCl<br />
→ PbCl 2<br />
+ H 2<br />
Equation:<br />
Pb +<br />
2 HCl<br />
→ PbCl 2<br />
+ H 2<br />
Before:<br />
Before:<br />
4.0 mol<br />
XS<br />
0 mol<br />
0 mol<br />
Change:<br />
Change:<br />
After:<br />
After:<br />
1
What are the ratios?<br />
Equation: Pb + 2 HCl → PbCl 2 + H 2<br />
Pb : HCl is 1:2 ratio (you need twice (2/1) as much HCl)<br />
Pb : PbCl 2 is 1:1 ratio<br />
Pb : H 2 is 1:1 ratio<br />
HCl : PbCl 2 is 2:1 ratio<br />
HCl : H 2 is 2:1 ratio<br />
PbCl 2 : H 2 is 1:1 ratio<br />
1.Lead will react with hydrochloric acid to<br />
produce lead chloride and hydrogen gas.<br />
How many moles of hydrochloric acid are<br />
needed to completely react with 4.0 mole<br />
of lead?<br />
Equation:<br />
Before:<br />
Change:<br />
After:<br />
Pb +<br />
4.0 mol<br />
-4.0 mol<br />
2 HCl<br />
XS<br />
-8.0 mol<br />
TWICE AS<br />
MANY AS Pb<br />
→ PbCl 2<br />
0 mol<br />
4.0 mol<br />
+ H 2<br />
0 mol<br />
4.0 mol<br />
Complete calculations on the side<br />
1.Lead will react with hydrochloric acid to<br />
produce lead chloride and hydrogen gas.<br />
How many moles of hydrochloric acid are<br />
needed to completely react with 4.0 mole<br />
of lead?<br />
In this case, desired answer is in<br />
moles<br />
If mass is required, convert moles<br />
to grams in the usual way<br />
Equation:<br />
Before:<br />
Change:<br />
After:<br />
Pb +<br />
4.0 mol<br />
-4.0 mol<br />
0<br />
2 HCl<br />
XS<br />
-8.0 mol<br />
XS<br />
→ PbCl 2<br />
0 mol<br />
4.0 mol<br />
4.0 mol<br />
+ H 2<br />
0 mol<br />
4.0 mol<br />
4.0 mol<br />
36.46g<br />
8 .0moles HCl × = 291.68g<br />
1mole<br />
Sig Figs: 2.9 x 10 2 g<br />
2
2. How many moles of hydrogen gas will be<br />
produced if 2.5 moles of calcium hydride<br />
react according to the following equation?<br />
Equation:<br />
Before:<br />
Change:<br />
After:<br />
CaH 2 +<br />
2 H 2 O →<br />
Ca(OH) 2<br />
+2 H 2<br />
2. How many moles of hydrogen gas will be<br />
produced if 2.5 moles of calcium hydride<br />
react according to the following equation?<br />
Equation:<br />
Before:<br />
Change:<br />
After:<br />
CaH 2 +<br />
2.5 mol<br />
2 H 2 O →<br />
XS<br />
Ca(OH) 2<br />
0 mol<br />
+2 H 2<br />
0 mol<br />
What are the ratios?<br />
Equation: CaH 2 + 2 H 2 O → Ca(OH) 2 +2 H 2<br />
CaH 2 : H 2 O is 1:2 ratio<br />
CaH 2 : Ca(OH) 2 is 1:1 ratio<br />
CaH 2 : H 2 is 1:2 ratio (you produce twice (2/1) as much H 2 )<br />
H 2 O : Ca(OH) 2 is 2:1 ratio<br />
H 2 O : H 2 is 2:2 ratio which reduces to 1:1<br />
H 2 : Ca(OH) 2 is 2:1 ratio<br />
2. How many moles of hydrogen gas will be<br />
produced if 2.5 moles of calcium hydride<br />
react according to the following equation?<br />
Equation:<br />
Before:<br />
Change:<br />
After:<br />
CaH 2 +<br />
2.5 mol<br />
-2.5 mol<br />
2 H 2 O →<br />
XS<br />
-5.0 mol<br />
Ca(OH) 2<br />
0 mol<br />
2.5 mol<br />
+2 H 2<br />
0 mol<br />
5.0 mol<br />
TWICE AS<br />
MUCH AS CaH 2<br />
3
2. How many moles of hydrogen gas will be<br />
produced if 2.5 moles of calcium hydride<br />
react according to the following equation?<br />
Equation:<br />
Before:<br />
Change:<br />
After:<br />
CaH 2 +<br />
2.5 mol<br />
-2.5 mol<br />
0<br />
2 H 2 O →<br />
XS<br />
-5.0 mol<br />
XS<br />
Ca(OH) 2<br />
0 mol<br />
2.5 mol<br />
2.5 mol<br />
+2 H 2<br />
0 mol<br />
5.0 mol<br />
5.0 mol<br />
Equation:<br />
Before:<br />
Change:<br />
After:<br />
3. How many moles of water will be<br />
produced if 0.45 mol of oxygen reacts<br />
according to the following equation?<br />
2 C 6 H 6 +<br />
15 O 2<br />
→ 12 CO 2<br />
+ 6 H 2 O<br />
What are the ratios?<br />
Equation:<br />
Before:<br />
Change:<br />
After:<br />
3. How many moles of water will be<br />
produced if 0.45 mol of oxygen reacts<br />
according to the following equation?<br />
2 C 6 H 6 +<br />
XS<br />
15 O 2<br />
0.45 mol<br />
→ 12 CO 2<br />
0 mol<br />
+ 6 H 2 O<br />
0 mol<br />
Equation: 2 C 6 H 6 + 15 O 2 → 12 CO 2 + 6 H 2 O<br />
C 6 H 6 : O 2 is 2:15 ratio<br />
C 6 H 6 : CO 2 is 2:12 ratio = 1:6<br />
C 6 H 6 : H 2 O is 2:6 ratio = 1:3<br />
O 2 : CO 2 is 15:12 ratio<br />
O 2 : H 2 O is 15:6 ratio = 5:2 (you produce 2/5 as much H 2 O)<br />
CO 2 : H 2 O is 12:6 ratio = 2:1<br />
4
Use Mole Ratios to Calculate<br />
2 C 6 H 6 + 15 O 2 → 12 CO 2 + 6<br />
H 2 O<br />
15 mol O 2 will produce 6 mol H 2 O<br />
6mol 2mol<br />
H<br />
O<br />
0 .45mol<br />
O<br />
×<br />
2<br />
=<br />
0.18mol<br />
H<br />
O<br />
2<br />
15mol<br />
O<br />
2<br />
2<br />
Equation:<br />
Before:<br />
Change:<br />
After:<br />
3. How many moles of water will be<br />
produced if 0.45 mol of oxygen reacts<br />
according to the following equation?<br />
2 C 6 H 6 +<br />
XS<br />
-0.06 mol<br />
15 O 2<br />
0.45 mol<br />
-0.45 mol<br />
→ 12 CO 2<br />
0 mol<br />
0.36 mol<br />
+ 6 H 2 O<br />
0 mol<br />
0.18 mol<br />
2/5 AS MANY<br />
AS O 2<br />
Equation:<br />
3. How many moles of water will be<br />
produced if 0.45 mol of oxygen reacts<br />
according to the following equation?<br />
2 C 6 H 6 +<br />
15 O 2<br />
→ 12 CO 2<br />
+ 6 H 2 O<br />
Before:<br />
XS<br />
0.45 mol<br />
0 mol<br />
0 mol<br />
Change:<br />
-0.06 mol<br />
-0.45 mol<br />
0.36 mol<br />
0.18 mol<br />
After:<br />
XS<br />
0<br />
0.36 mol<br />
0.18 mol<br />
5