White Rust on Galvanized Steel
White Rust on Galvanized Steel
White Rust on Galvanized Steel
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
Zinc Passivati<strong>on</strong><br />
Like many metals, the good corrosi<strong>on</strong> properties<br />
of zinc are due to a thin film of corrosi<strong>on</strong><br />
product that forms <strong>on</strong> the surface of the metal.<br />
This tightly-adherent corrosi<strong>on</strong> film is called a<br />
passivati<strong>on</strong> layer. The formati<strong>on</strong> of the<br />
passivati<strong>on</strong> layer is essential to achieving l<strong>on</strong>glife<br />
with a galvanized product.<br />
Zinc will naturally form a passivati<strong>on</strong> layer<br />
when exposed to weathering over an extended<br />
period of time. Rain combined with air drying<br />
will, over the course of a year or more form a<br />
passivati<strong>on</strong> layer <strong>on</strong> all areas of the zinc which<br />
do not come in c<strong>on</strong>tact with recirculating water.<br />
Thus, the exterior of evaporative equipment and<br />
all interior areas above the drift eliminators will<br />
naturally develop a passivati<strong>on</strong> film.<br />
For the comp<strong>on</strong>ents of the system that are<br />
exposed to or immersed in recirculating water,<br />
the water chemistry must be carefully c<strong>on</strong>trolled<br />
until a passivati<strong>on</strong> film forms. Once the film<br />
forms, the recirculating water chemistry, though<br />
still important, is less critical for zinc corrosi<strong>on</strong>.<br />
If the passivati<strong>on</strong> film is damaged by abrasi<strong>on</strong> or<br />
chemical attack, it must be re-formed by again<br />
carefully c<strong>on</strong>trolling the recirculating water<br />
chemistry.<br />
It is very difficult to c<strong>on</strong>trol these water<br />
parameters and form a passivati<strong>on</strong> layer in a<br />
system started under load due to the higher<br />
temperature and the c<strong>on</strong>tinually changing water<br />
chemistry. Therefore, if possible, the equipment<br />
should be commissi<strong>on</strong>ed with water circulati<strong>on</strong><br />
and without a heat load. The critical parameters<br />
to form the passivati<strong>on</strong> film are described in the<br />
following table. It generally takes 4 to 12 weeks<br />
to passivate zinc with pH c<strong>on</strong>trol the most<br />
critical parameter and with the first few weeks<br />
of water exposure being the most critical time.<br />
Figure 2<br />
Bright and Shiny<br />
Fresh Zinc<br />
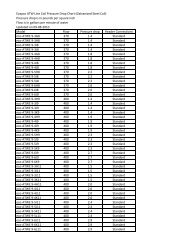
Parameter Passivati<strong>on</strong> Value<br />
pH 7.0 – 8.0<br />
C<strong>on</strong>ductivity 50 ppm<br />
Chlorides<br />