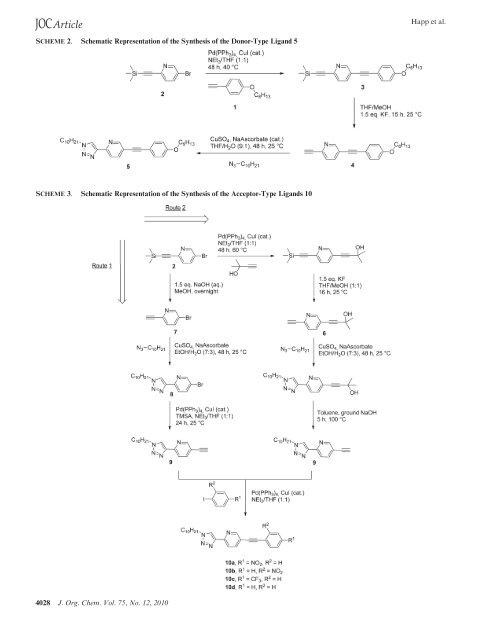
JOCArticle Happ et al. SCHEME 2. Schematic Representati<strong>on</strong> of the Synthesis of the <str<strong>on</strong>g>D<strong>on</strong>or</str<strong>on</strong>g>-<str<strong>on</strong>g>Type</str<strong>on</strong>g> Lig<str<strong>on</strong>g>and</str<strong>on</strong>g> 5 SCHEME 3. Schematic Representati<strong>on</strong> of the Synthesis of the <str<strong>on</strong>g>Acceptor</str<strong>on</strong>g>-<str<strong>on</strong>g>Type</str<strong>on</strong>g> Lig<str<strong>on</strong>g>and</str<strong>on</strong>g>s 10 4028 J. Org. Chem. Vol. 75, No. 12, 2010
Happ et al. SCHEME 4. Schematic Representati<strong>on</strong> of the Synthesis of the <str<strong>on</strong>g>Acceptor</str<strong>on</strong>g>-<str<strong>on</strong>g>Type</str<strong>on</strong>g> Lig<str<strong>on</strong>g>and</str<strong>on</strong>g> 12 Bearing an Aromatic Moiety JOCArticle SCHEME 5. Schematic Representati<strong>on</strong> of the Synthesis of a 2-(<strong>1H</strong>-[1,2,3]Triazol-4-yl)pyridyl Lig<str<strong>on</strong>g>and</str<strong>on</strong>g> with an <str<strong>on</strong>g>Acceptor</str<strong>on</strong>g>-Unit Attached <strong>on</strong> the Triazole Ring acceptor-functi<strong>on</strong>alized compound 15 was synthesized in moderate yield (50%). However, the implementati<strong>on</strong> of the O-alkyl d<strong>on</strong>or system in analogy to 5 was not yet successful. The heteroleptic ruthenium(II) complexes 16 to 18 of the general structure [(dmbpy) 2 RuL](PF 6 ) 2 (dmbpy =4,4 0 -dimethyl-2,2 0 -bipyridine) were synthesized by heating Ru- (dmbpy) 2 Cl 2 8,11 <str<strong>on</strong>g>and</str<strong>on</strong>g> the appropriate lig<str<strong>on</strong>g>and</str<strong>on</strong>g>s (L = 5, 10, 12, 15) under microwave irradiati<strong>on</strong> (Table 1). After 2 h the reacti<strong>on</strong>s were completed <str<strong>on</strong>g>and</str<strong>on</strong>g> a 10-fold excess of NH 4 PF 6 was added to precipitate the products. In most cases, precipitati<strong>on</strong> occurred after 15 min <str<strong>on</strong>g>and</str<strong>on</strong>g> the pure complex was isolated after washing with ethanol <str<strong>on</strong>g>and</str<strong>on</strong>g> diethyl ether in moderate to very good yields (Table 1). Only 16c <str<strong>on</strong>g>and</str<strong>on</strong>g> 17 had to be recrystallized from ethanol/water. The latter fact may explain the moderate yields obtained for these complexes. The verificati<strong>on</strong> of the structures of 16-18 was carried out by 1 H <str<strong>on</strong>g>and</str<strong>on</strong>g> 13 C NMR spectroscopy as well as HR-ESI mass spectrometry. (11) Rau, S.; Ruben, M.; B€uttner, T.; Temme, C.; Dautz, S.; G€orls, H.; Rudolph, M.; Walther, D.; Vos, J. G. J. Chem. Soc., Dalt<strong>on</strong> Trans. 2000, 3649–3657. Crystallographic Analysis. For the heteroleptic Ru II complex 17 single crystals suitable for X-ray crystallographic analysis were obtained by slow diethyl ether diffusi<strong>on</strong> revealing the coordinati<strong>on</strong> of the two 4,4 0 -dimethyl-2,2 0 -bipyridine <str<strong>on</strong>g>and</str<strong>on</strong>g> the 2-(<strong>1H</strong>-[1,2,3]triazol-4-yl)pyridine lig<str<strong>on</strong>g>and</str<strong>on</strong>g> 12 to the Ru II core. The molecule crystallizes in a triclinic system with P1 symmetry (for ORTEP see the Supporting Informati<strong>on</strong>). The structural parameters observed from 17 were compared to the corresp<strong>on</strong>ding structurally related homoleptic complex [Ru(bpy) 3 ](PF 6 ) 2 12 (Figure 2). It is noteworthy that the interlig<str<strong>on</strong>g>and</str<strong>on</strong>g> angle of the nitrogen atoms in the square plane were similar for both complexes (17: 170.40° <str<strong>on</strong>g>and</str<strong>on</strong>g> 174.40°; [Ru(bpy) 3 ](PF 6 ) 2 : 172.3°; see Figure 2) resulting in a comparable distorti<strong>on</strong> of the ideal octahedral geometry. The b<strong>on</strong>d length from ruthenium to the coordinating nitrogen atom of the triazole ring (N 0 : 2.028(2), Figure 2) was shortened significantly compared (12) Biner, M.; Buergi, H. B.; Ludi, A.; Roehr, C. J. Am. Chem. Soc. 1992, 114, 5197–5203. (13) Egbe, D. A. M.; Bader, C.; Nowotny, J.; G€unther, W.; Klemm, E. Macromolecules 2003, 36, 5459–5469. J. Org. Chem. Vol. 75, No. 12, 2010 4029