KEY-FinalExamStudyGuide_Fall2013
KEY-FinalExamStudyGuide_Fall2013
KEY-FinalExamStudyGuide_Fall2013
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
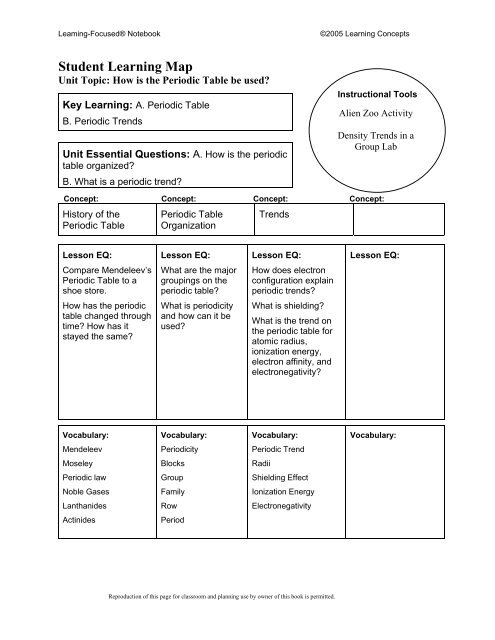
Leaming-Focused® Notebook<br />
©2005 Learning Concepts<br />
Student Learning Map<br />
Unit Topic: How is the Periodic Table be used?<br />
Key Learning: A. Periodic Table<br />
B. Periodic Trends<br />
Unit Essential Questions: A. How is the periodic<br />
table organized?<br />
B. What is a periodic trend?<br />
Instructional Tools<br />
Alien Zoo Activity<br />
Density Trends in a<br />
Group Lab<br />
Concept: Concept: Concept: Concept:<br />
History of the<br />
Periodic Table<br />
Periodic Table<br />
Organization<br />
Trends<br />
Lesson EQ:<br />
Lesson EQ:<br />
Lesson EQ:<br />
Lesson EQ:<br />
Compare Mendeleev’s<br />
Periodic Table to a<br />
shoe store.<br />
What are the major<br />
groupings on the<br />
periodic table?<br />
How does electron<br />
configuration explain<br />
periodic trends?<br />
How has the periodic<br />
table changed through<br />
time? How has it<br />
stayed the same?<br />
What is periodicity<br />
and how can it be<br />
used?<br />
What is shielding?<br />
What is the trend on<br />
the periodic table for<br />
atomic radius,<br />
ionization energy,<br />
electron affinity, and<br />
electronegativity?<br />
Vocabulary:<br />
Vocabulary:<br />
Vocabulary:<br />
Vocabulary:<br />
Mendeleev<br />
Periodicity<br />
Periodic Trend<br />
Moseley<br />
Blocks<br />
Radii<br />
Periodic law<br />
Group<br />
Shielding Effect<br />
Noble Gases<br />
Family<br />
Ionization Energy<br />
Lanthanides<br />
Row<br />
Electronegativity<br />
Actinides<br />
Period<br />
Reproduction of this page for classroom and planning use by owner of this book is permitted.