Safe and Suitable Ingredients - GoodFood World
Safe and Suitable Ingredients - GoodFood World
Safe and Suitable Ingredients - GoodFood World
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
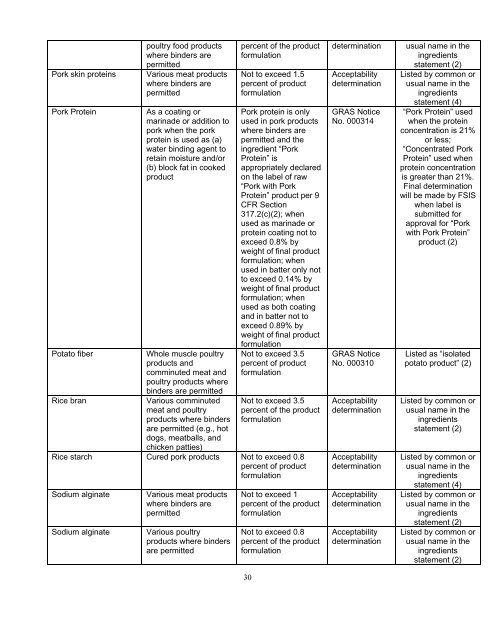
Pork skin proteins<br />
Pork Protein<br />
Potato fiber<br />
Rice bran<br />
poultry food products<br />
where binders are<br />
permitted<br />
Various meat products<br />
where binders are<br />
permitted<br />
As a coating or<br />
marinade or addition to<br />
pork when the pork<br />
protein is used as (a)<br />
water binding agent to<br />
retain moisture <strong>and</strong>/or<br />
(b) block fat in cooked<br />
product<br />
Whole muscle poultry<br />
products <strong>and</strong><br />
comminuted meat <strong>and</strong><br />
poultry products where<br />
binders are permitted<br />
Various comminuted<br />
meat <strong>and</strong> poultry<br />
products where binders<br />
are permitted (e.g., hot<br />
dogs, meatballs, <strong>and</strong><br />
percent of the product<br />
formulation<br />
Not to exceed 1.5<br />
percent of product<br />
formulation<br />
Pork protein is only<br />
used in pork products<br />
where binders are<br />
permitted <strong>and</strong> the<br />
ingredient “Pork<br />
Protein” is<br />
appropriately declared<br />
on the label of raw<br />
“Pork with Pork<br />
Protein” product per 9<br />
CFR Section<br />
317.2(c)(2); when<br />
used as marinade or<br />
protein coating not to<br />
exceed 0.8% by<br />
weight of final product<br />
formulation; when<br />
used in batter only not<br />
to exceed 0.14% by<br />
weight of final product<br />
formulation; when<br />
used as both coating<br />
<strong>and</strong> in batter not to<br />
exceed 0.89% by<br />
weight of final product<br />
formulation<br />
Not to exceed 3.5<br />
percent of product<br />
formulation<br />
Not to exceed 3.5<br />
percent of the product<br />
formulation<br />
chicken patties)<br />
Rice starch Cured pork products Not to exceed 0.8<br />
percent of product<br />
formulation<br />
Sodium alginate<br />
Sodium alginate<br />
Various meat products<br />
where binders are<br />
permitted<br />
Various poultry<br />
products where binders<br />
are permitted<br />
Not to exceed 1<br />
percent of the product<br />
formulation<br />
Not to exceed 0.8<br />
percent of the product<br />
formulation<br />
30<br />
determination<br />
Acceptability<br />
determination<br />
GRAS Notice<br />
No. 000314<br />
GRAS Notice<br />
No. 000310<br />
Acceptability<br />
determination<br />
Acceptability<br />
determination<br />
Acceptability<br />
determination<br />
Acceptability<br />
determination<br />
usual name in the<br />
ingredients<br />
statement (2)<br />
Listed by common or<br />
usual name in the<br />
ingredients<br />
statement (4)<br />
“Pork Protein” used<br />
when the protein<br />
concentration is 21%<br />
or less;<br />
“Concentrated Pork<br />
Protein” used when<br />
protein concentration<br />
is greater than 21%.<br />
Final determination<br />
will be made by FSIS<br />
when label is<br />
submitted for<br />
approval for “Pork<br />
with Pork Protein”<br />
product (2)<br />
Listed as “isolated<br />
potato product” (2)<br />
Listed by common or<br />
usual name in the<br />
ingredients<br />
statement (2)<br />
Listed by common or<br />
usual name in the<br />
ingredients<br />
statement (4)<br />
Listed by common or<br />
usual name in the<br />
ingredients<br />
statement (2)<br />
Listed by common or<br />
usual name in the<br />
ingredients<br />
statement (2)