Chapter 4.pdf
Chapter 4.pdf
Chapter 4.pdf
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
4 CHAPTER REVIEW<br />
ACHIEVEMENT CHART CATEGORIES<br />
k Knowledge and understanding t Thinking and investigation<br />
c Communication<br />
a Application<br />
Key Concept Review<br />
1. What is the main difference between a pure<br />
substance and a mixture Name an example<br />
of each. k<br />
2. What is a chemical change<br />
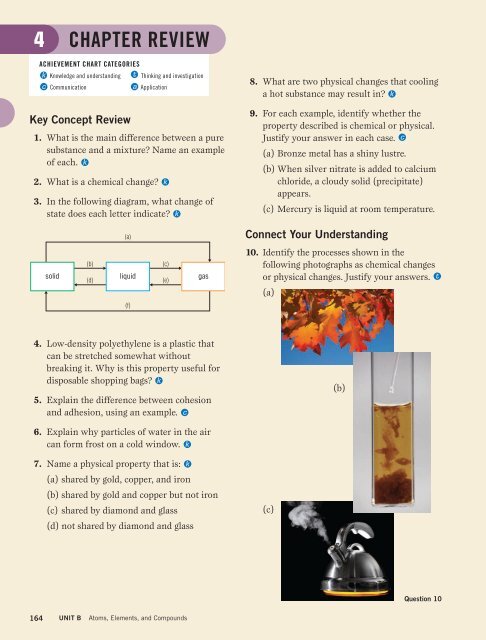
3. In the following diagram, what change of<br />
state does each letter indicate k<br />
k<br />
8. What are two physical changes that cooling<br />
a hot substance may result in k<br />
9. For each example, identify whether the<br />
property described is chemical or physical.<br />
Justify your answer in each case. c<br />
(a) Bronze metal has a shiny lustre.<br />
(b) When silver nitrate is added to calcium<br />
chloride, a cloudy solid (precipitate)<br />
appears.<br />
(c) Mercury is liquid at room temperature.<br />
(a)<br />
Connect Your Understanding<br />
solid<br />
(b)<br />
(d)<br />
liquid<br />
(c)<br />
(e)<br />
gas<br />
10. Identify the processes shown in the<br />
following photographs as chemical changes<br />
or physical changes. Justify your answers.<br />
(a)<br />
t<br />
(f)<br />
4. Low-density polyethylene is a plastic that<br />
can be stretched somewhat without<br />
breaking it. Why is this property useful for<br />
disposable shopping bags k<br />
5. Explain the difference between cohesion<br />
and adhesion, using an example. c<br />
(b)<br />
6. Explain why particles of water in the air<br />
can form frost on a cold window. k<br />
7. Name a physical property that is:<br />
(a) shared by gold, copper, and iron<br />
(b) shared by gold and copper but not iron<br />
(c) shared by diamond and glass<br />
(d) not shared by diamond and glass<br />
k<br />
(c)<br />
Question 10<br />
164 UNIT B Atoms, Elements, and Compounds