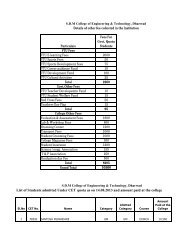
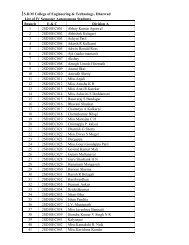
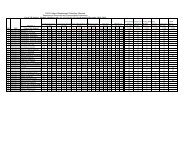
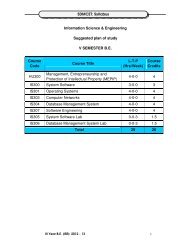
<strong>SDMCET</strong>: <strong>Syllabus</strong>cathodic), inorganic coatings(anodized & phosphate), cathodic protection(sacrificial & impressed current methods) & anodic protection. 6 Hrs.4) Chemical Energy Sources: Fuels--Introduction, Classification, Calorific value,Determination <strong>of</strong> calorific value using Bomb calorimeter, Numerical problems,Cracking—Introduction, fixed bed and fluidized bed catalytic cracking;advantages <strong>of</strong> moving bed catalytic cracking over fixed bed catalytic cracking,Synthesis <strong>of</strong> petrol by Bergius process & Fishcher-Tropsch process, Reforming<strong>of</strong> petrol—significance & reactions; gasoline & diesel knocking & theirmechanism, , anti knocking agents; unleaded petrol; Octane number; Cetanenumber; Power alcohol & Biodiesel.7 Hrs.5) High Polymers: Introduction, Types <strong>of</strong> polymerization – Addition andcondensation polymerization with examples; Methods <strong>of</strong> polymerization- bulk,solution, suspension and emulsion; Mechanism <strong>of</strong> polymerization - Free radicaltaking ethylene as a monomer; Glass Transition temperature, factorsinfluencing T g –flexibility, inter molecular forces, branching and cross-linking,molecular mass, stereo regularity and presence <strong>of</strong> plasticizers; Significance <strong>of</strong>T g ; Synthesis, properties & applications <strong>of</strong> Polycarbonate, Polymethylmethacrylate and Polyurethane; Elastomers—introduction, synthesis, &applications <strong>of</strong> butyl rubber and silicone rubber. Adhesives--introduction,synthesis, & applications <strong>of</strong> epoxy resins; conducting polymers- Introduction,Synthesis and applications <strong>of</strong> conducting polyaniline.7 Hrs.6) Water Technology: Introduction, sources and impurities <strong>of</strong> water; Hardness <strong>of</strong>water, Units <strong>of</strong> Hardness, Determination <strong>of</strong> Hardness and Alkalinity. Numericalproblems on hardness and alkalinity. Determination <strong>of</strong> Chloride & Fluoride.Determination <strong>of</strong> DO, COD, BOD, Numerical problems on BOD & COD,Desalination <strong>of</strong> water- Multistage flash evaporation, Reverse osmosis and Ionselective Electrodialysis method.6 Hrs.7) Phase Equilibria: Phase rule – statement, Terms- definition and ex<strong>plan</strong>ation<strong>of</strong> phase, component and degree <strong>of</strong> freedom. Applications <strong>of</strong> phase rule overone component systems- water system, over two component systems- types<strong>of</strong> equilibria, Lead- silver system Formation <strong>of</strong> compounds with congruentmelting points: Ferric chloride- water system, with incongruent melting points:Sodium sulphate – water system.5 Hrs.8) Inorganic cementing materials:a) Cement: Classification- Natural, puzzolana, slag, Portland and special cements.Portland cement- Raw materials, manufacture by wet and dry processes,chemical constitution <strong>of</strong> Portland cement. Setting and hardening <strong>of</strong> Portlandcement, functions <strong>of</strong> gypsum in cement.b) Refractories: Definition, physical and chemical characteristics-Refractoriness,strength, spalling, thermal conductivity, Chemical porosity and chemicalcomposition. Classification- Acidic, basic and neutral. Preparation, propertiesI Year B. E: 2013 – 1418
<strong>SDMCET</strong>: <strong>Syllabus</strong>and uses <strong>of</strong> silica, magnesite, carbon, and carborundom.6 Hrs.Books:1) Jain P.C. & Monika Jain, Engineering Chemistry, Dhanpat Rai & Sons, Delhi.2) A Text Book <strong>of</strong> Engineering Chemistry – M.M. Uppal, Khanna Publishers.3) Puri B.R., Sharma L.R. and M.S. Pathania, Principles <strong>of</strong> Physical Chemistry, S.N.Chand & Co., Jalandhar.4) J.C. Kuriacose, J. Rajaram, Chemistry in Engineering and Technology, Volume I& II, Tata McGraw-Hill Publishing Company Limited, New Delhi.5) R. Narayan & B. Vishwanathan, Chemical and Electrochemical Energy Systems,Universities Press (India) Limited.6) Mars G. Fontana, Corrosion Engineering, McGraw-Hill International Edition.7) Gowarikar V.R., Viswanathan, N.V. & Jayadev Shreedharan, Polymer Science,New Age International Publishers.8) Billmeyer F.W., Textbook <strong>of</strong> Polymer Science, John Wiley & Sons.9) Will Mitchell J.R., Fuel Cells, Academic Press, New York and London.10) C.D. Verghese, Electroplating and other surface treatments, Tata McGraw-HillPublishing Company Ltd.11UCYL100/11UCYL200 Engineering Chemistry Lab (0–0–3-0) 1.5List <strong>of</strong> ExperimentsVolumetry:1) Estimation <strong>of</strong> total hardness <strong>of</strong> water by EDTA method.2) Determination <strong>of</strong> alkalinity (hydroxide, carbonate and bicarbonate) <strong>of</strong> watersample.3) Estimation <strong>of</strong> calcium present in the given sample <strong>of</strong> cement solution by EDTAmethod.4) Estimation <strong>of</strong> percentage <strong>of</strong> copper in brass by iodometric method.5) Estimation <strong>of</strong> amount <strong>of</strong> iron in haematite ore solution by external indicatormethod.Instrumental:1) Estimation <strong>of</strong> hydrochloric acid & acetic acid from their mixture conductometrically.2) Estimation <strong>of</strong> FAS Potentiometrically.3) Estimation <strong>of</strong> copper colorimetrically.4) Determination <strong>of</strong> pK a value <strong>of</strong> a given weak acid using pH-meter.5) Determination <strong>of</strong> viscosity coefficient <strong>of</strong> a given liquid using Ostwald’sviscometer.I Year B. E: 2013 – 1419