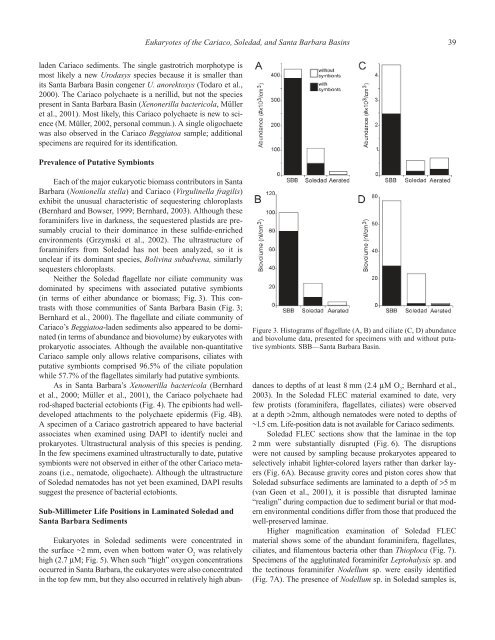
Eukaryotes of the Cariaco, Soledad, <strong>and</strong> Santa Barbara Basins 39laden Cariaco sediments. The single gastrotrich morphotype ismost likely a new Urodasys species because it is smaller thanits Santa Barbara Basin congener U. anorektoxys (Todaro et al.,2000). The Cariaco polychaete is a nerillid, but not the speciespresent in Santa Barbara Basin (Xenonerilla bactericola, Mülleret al., 2001). Most likely, this Cariaco polychaete is new to science(M. Müller, 2002, personal commun.). A single oligochaetewas also observed in the Cariaco Beggiatoa sample; additionalspecimens are required for its identification.Prevalence of Putative SymbiontsEach of the major eukaryotic biomass contributors in SantaBarbara (Nonionella stella) <strong>and</strong> Cariaco (Virgulinella fragilis)exhibit the unusual characteristic of sequestering chloroplasts(Bernhard <strong>and</strong> Bowser, 1999; Bernhard, 2003). Although theseforaminifers live in darkness, the sequestered plastids are presumablycrucial to their dominance in these sulfide-enrichedenvironments (Grzymski et al., 2002). The ultrastructure offoraminifers from Soledad has not been analyzed, so it isunclear if its dominant species, Bolivina subadvena, similarlysequesters chloroplasts.Neither the Soledad flagellate nor ciliate community wasdominated by specimens with associated putative symbionts(in terms of either abundance or biomass; Fig. 3). This contrastswith those communities of Santa Barbara Basin (Fig. 3;Bernhard et al., 2000). The flagellate <strong>and</strong> ciliate community ofCariaco’s Beggiatoa-laden sediments also appeared to be dominated(in terms of abundance <strong>and</strong> biovolume) by eukaryotes withprokaryotic associates. Although the available non-quantitativeCariaco sample only allows relative comparisons, ciliates withputative symbionts comprised 96.5% of the ciliate populationwhile 57.7% of the flagellates similarly had putative symbionts.As in Santa Barbara’s Xenonerilla bactericola (Bernhardet al., 2000; Müller et al., 2001), the Cariaco polychaete hadrod-shaped bacterial ectobionts (Fig. 4). The epibionts had welldevelopedattachments to the polychaete epidermis (Fig. 4B).A specimen of a Cariaco gastrotrich appeared to have bacterialassociates when examined using DAPI to identify nuclei <strong>and</strong>prokaryotes. Ultrastructural analysis of this species is pending.In the few specimens examined ultrastructurally to date, putativesymbionts were not observed in either of the other Cariaco metazoans(i.e., nematode, oligochaete). Although the ultrastructureof Soledad nematodes has not yet been examined, DAPI resultssuggest the presence of bacterial ectobionts.Sub-Millimeter Life Positions in Laminated Soledad <strong>and</strong>Santa Barbara SedimentsFigure 3. Histograms of flagellate (A, B) <strong>and</strong> ciliate (C, D) abundance<strong>and</strong> biovolume data, presented for specimens with <strong>and</strong> without putativesymbionts. SBB—Santa Barbara Basin.Eukaryotes in Soledad sediments were concentrated inthe surface ~2 mm, even when bottom water O 2was relativelyhigh (2.7 µM; Fig. 5). When such “high” oxygen concentrationsoccurred in Santa Barbara, the eukaryotes were also concentratedin the top few mm, but they also occurred in relatively high abundancesto depths of at least 8 mm (2.4 µM O 2; Bernhard et al.,2003). In the Soledad FLEC material examined to date, veryfew protists (foraminifera, flagellates, ciliates) were observedat a depth >2mm, although nematodes were noted to depths of~1.5 cm. Life-position data is not available for Cariaco sediments.Soledad FLEC sections show that the laminae in the top2 mm were substantially disrupted (Fig. 6). The disruptionswere not caused by sampling because prokaryotes appeared toselectively inhabit lighter-colored layers rather than darker layers(Fig. 6A). Because gravity cores <strong>and</strong> piston cores show thatSoledad subsurface sediments are laminated to a depth of >5 m(van Geen et al., 2001), it is possible that disrupted laminae“realign” during compaction due to sediment burial or that modernenvironmental conditions differ from those that produced thewell-preserved laminae.Higher magnification examination of Soledad FLECmaterial shows some of the abundant foraminifera, flagellates,ciliates, <strong>and</strong> filamentous bacteria other than Thioploca (Fig. 7).Specimens of the agglutinated foraminifer Leptohalysis sp. <strong>and</strong>the tectinous foraminifer Nodellum sp. were easily identified(Fig. 7A). The presence of Nodellum sp. in Soledad samples is,
40 J.M. Bernhard <strong>and</strong> K.R. BuckFigure 4. Transmission electron micrographs of the undescribed Cariaco polychaete. (A) View including the polychaete epidermis <strong>and</strong> attachedbacterial ectobionts. (B) Higher-magnification view showing morphological modifications at attachment points (arrows). m—mitochondria.Scale bars: A =1 µm; B = 200 nm.to our knowledge, the first recorded occurrence of this tectinousgenus in laminated sediments.Sub-millimeter life positions of Santa Barbara Basineukaryotes have recently been described (Bernhard et al., 2003).Examination of additional material indicates that metazoans didnot occur exclusively at the sediment-water interface even whenoxygen was nearly undetectable (Fig. 8; 0.1 µM O 2; Bernhard etal., 2003). More specifically, polychaetes <strong>and</strong> nematodes wereboth observed at depths of at least 6 mm (Fig. 8) in laminatedsediments (see Figure 2D in Bernhard et al., 2003).Given the apparently strict occurrence of Soledad eukaryotesin the surface 1–2 mm (Fig. 5), abundance data illustrated in Figure2 was recalculated to determine the density in the top 2 mmof Soledad sediments. When considered in this manner, Soledadeukaryote densities <strong>and</strong> biovolume in Soledad exceed those in thesurface centimeter of Santa Barbara Basin (i.e., Soledad density~55.9 × 10 4 cm −3 versus Santa Barbara ~43.3 × 10 4 cm −3 ; Soledadbiovolume ~1101 nl cm −3 versus Santa Barbara ~967 nl cm −3 ).Such a perspective suggests that ecological approaches at thecentimeter scale are not always representative of in situ conditionsat scales relevant to the microorganisms.DISCUSSIONOur observations suggest that, in general, the eukaryoticfauna of deep-water sulfidic habitats is relatively diverse, with ahigh dominance of few or one species, <strong>and</strong> a high total abundance,if considered at the appropriate scale. In addition, protists typicallydominate abundance <strong>and</strong> biovolume, <strong>and</strong> most eukaryotictaxa have bacterial associates. Taxonomic comparisons indicatesimilarities in metazoan taxa, with nematodes in all three basins<strong>and</strong> gastrotrichs <strong>and</strong> primitive polychaetes occurring in two of thebasins (Table 2). In at least two of the three basins, the dominantforaminifer sequesters chloroplasts, even though the basin seafloorlies far below the maximum extent of the photic zone.Although little is known regarding the taxonomy of flagellates<strong>and</strong> ciliates from oxygen-depleted deep-water sediments(or their associated putative symbionts), much is known aboutthe comparable fauna from shallow-water environments (e.g.,Fenchel <strong>and</strong> Finlay, 1995). Given the recent <strong>and</strong> controversialassertion that protistan species are cosmopolitan <strong>and</strong>, by extrapolation,of limited diversity (Finlay, 2002), deep-water nanobiotaare crucial communities with which to test this hypothesis. Our
- Page 2: Sulfur Biogeochemistry—Past and P
- Page 8: Preface viisulfurization on the pre
- Page 11 and 12: 2 V. BrüchertThe resulting sulfur
- Page 13 and 14: 4 V. BrüchertFigure 1. Phylogeneti
- Page 15 and 16: 6 V. Brüchertnot yet been resolved
- Page 17 and 18: 8 V. BrüchertEFFECT OF ELECTRON DO
- Page 19 and 20: 10 V. Brüchertfractionation factor
- Page 21 and 22: 12 V. BrüchertFigure 4. Depth prof
- Page 23 and 24: 14 V. Brüchertto derive isotope fr
- Page 25 and 26: 16 V. BrüchertRudnicki, M.D., Elde
- Page 28 and 29: Microbially mediated sulfur-redox 1
- Page 30 and 31: Microbially mediated sulfur-redox 2
- Page 32 and 33: Microbially mediated sulfur-redox 2
- Page 34 and 35: Microbially mediated sulfur-redox 2
- Page 36 and 37: Microbially mediated sulfur-redox 2
- Page 38 and 39: Microbially mediated sulfur-redox 2
- Page 40 and 41: Microbially mediated sulfur-redox 3
- Page 42 and 43: Microbially mediated sulfur-redox 3
- Page 44 and 45: Geological Society of AmericaSpecia
- Page 46 and 47: Eukaryotes of the Cariaco, Soledad,
- Page 50 and 51: Eukaryotes of the Cariaco, Soledad,
- Page 52 and 53: Eukaryotes of the Cariaco, Soledad,
- Page 54 and 55: Eukaryotes of the Cariaco, Soledad,
- Page 56: Eukaryotes of the Cariaco, Soledad,
- Page 59 and 60: 50 A. SchippersTABLE 1. DETECTION O
- Page 61 and 62: 52 A. SchippersS 4O 62−+ H 2O →
- Page 63 and 64: 54 A. Schippersthe oxygen molecule
- Page 65 and 66: 56 A. Schippersprohibits biological
- Page 67 and 68: 58 A. SchippersSampleTABLE 4. SULFU
- Page 69 and 70: 60 A. SchippersGeochimica et Cosmoc
- Page 71 and 72: 62 A. SchippersSasaki, K., Tsunekaw
- Page 73 and 74: 64 B.B. Jørgensen and D.C. NelsonI
- Page 75 and 76: 66 B.B. Jørgensen and D.C. NelsonF
- Page 77 and 78: 68 B.B. Jørgensen and D.C. Nelsonq
- Page 79 and 80: 70 B.B. Jørgensen and D.C. Nelsons
- Page 81 and 82: 72 B.B. Jørgensen and D.C. Nelsonp
- Page 83 and 84: 74 B.B. Jørgensen and D.C. NelsonP
- Page 85 and 86: 76 B.B. Jørgensen and D.C. Nelsont
- Page 87 and 88: 78 B.B. Jørgensen and D.C. NelsonF
- Page 89 and 90: 80 B.B. Jørgensen and D.C. NelsonJ
- Page 92 and 93: Geological Society of AmericaSpecia
- Page 94 and 95: Formation and degradation of seafl
- Page 96 and 97: Formation and degradation of seafl
- Page 98 and 99:
Formation and degradation of seafl
- Page 100 and 101:
Formation and degradation of seafl
- Page 102 and 103:
Formation and degradation of seafl
- Page 104 and 105:
Formation and degradation of seafl
- Page 106 and 107:
Geological Society of AmericaSpecia
- Page 108 and 109:
Distribution and fate of sulfur int
- Page 110 and 111:
Distribution and fate of sulfur int
- Page 112 and 113:
Distribution and fate of sulfur int
- Page 114 and 115:
Distribution and fate of sulfur int
- Page 116 and 117:
Distribution and fate of sulfur int
- Page 118 and 119:
Distribution and fate of sulfur int
- Page 120 and 121:
Distribution and fate of sulfur int
- Page 122 and 123:
Distribution and fate of sulfur int
- Page 124 and 125:
Distribution and fate of sulfur int
- Page 126 and 127:
Geological Society of AmericaSpecia
- Page 128 and 129:
- HSO 4SO 2-4H 2 SCO 32-HCO 3-HCO 3
- Page 130 and 131:
Sedimentary pyrite formation 121mar
- Page 132 and 133:
Sedimentary pyrite formation 123The
- Page 134 and 135:
Sedimentary pyrite formation 125Ben
- Page 136 and 137:
Sedimentary pyrite formation 127TAB
- Page 138 and 139:
Sedimentary pyrite formation 129The
- Page 140 and 141:
Sedimentary pyrite formation 131Dek
- Page 142 and 143:
Sedimentary pyrite formation 133Mor
- Page 144 and 145:
Geological Society of AmericaSpecia
- Page 146 and 147:
Recent advances and future research
- Page 148 and 149:
Recent advances and future research
- Page 150 and 151:
Recent advances and future research
- Page 152 and 153:
Recent advances and future research
- Page 154 and 155:
Recent advances and future research
- Page 156 and 157:
Recent advances and future research
- Page 158 and 159:
Recent advances and future research
- Page 160 and 161:
Geological Society of AmericaSpecia
- Page 162 and 163:
Using sulfur isotopes to elucidate
- Page 164 and 165:
Using sulfur isotopes to elucidate
- Page 166 and 167:
Using sulfur isotopes to elucidate
- Page 168 and 169:
Using sulfur isotopes to elucidate
- Page 170 and 171:
Geological Society of AmericaSpecia
- Page 172 and 173:
Sites of anomalous organic reminera
- Page 174 and 175:
Sites of anomalous organic reminera
- Page 176 and 177:
Sites of anomalous organic reminera
- Page 178 and 179:
Sites of anomalous organic reminera
- Page 180 and 181:
Sites of anomalous organic reminera
- Page 182 and 183:
Sites of anomalous organic reminera
- Page 184 and 185:
Sites of anomalous organic reminera
- Page 186 and 187:
Geological Society of AmericaSpecia
- Page 188 and 189:
Sulfur isotope composition of carbo
- Page 190 and 191:
Sulfur isotope composition of carbo
- Page 192 and 193:
Sulfur isotope composition of carbo
- Page 194 and 195:
Sulfur isotope composition of carbo
- Page 196 and 197:
Sulfur isotope composition of carbo
- Page 198 and 199:
Sulfur isotope composition of carbo
- Page 200 and 201:
Sulfur isotope composition of carbo
- Page 202 and 203:
Sulfur isotope composition of carbo
- Page 204 and 205:
Geological Society of AmericaSpecia
- Page 206 and 207:
4 Ga of seawater evolution 197brine
- Page 208 and 209:
4 Ga of seawater evolution 199Figur
- Page 210 and 211:
4 Ga of seawater evolution 201data
- Page 212 and 213:
4 Ga of seawater evolution 203notab
- Page 214:
4 Ga of seawater evolution 205et Co