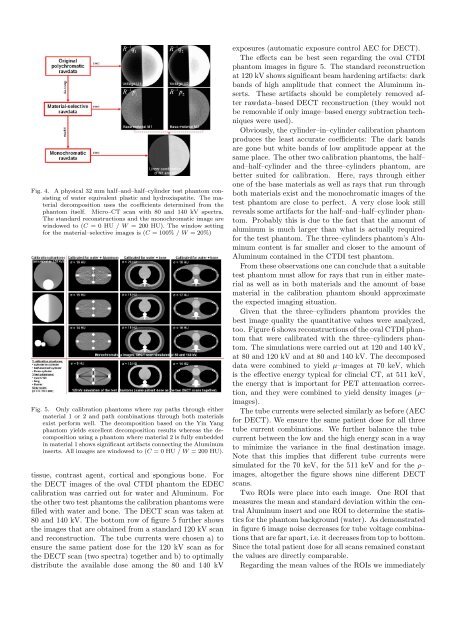
Fig. 4. A physical 32 mm half–and–half–cylinder test phantom consistingof water equivalent plastic and hydroxiapatite. The materialdecomposition uses the coefficients determined from thephantom itself. Micro–CT scan with 80 and 140 kV spectra.The standard reconstructions and the monochromatic image arewindowed to (C = 0 HU / W = 200 HU). The window setting<strong>for</strong> the material–selective images is (C = 100% / W = 20%)Fig. 5. Only calibration phantoms where ray paths through eithermaterial 1 or 2 and path combinations through both materialsexist per<strong>for</strong>m well. The decomposition based on the Yin Yangphantom yields excellent decomposition results whereas the decompositionusing a phantom where material 2 is fully embeddedin material 1 shows significant artifacts connecting the Aluminuminserts. All images are windowed to (C = 0 HU / W = 200 HU).tissue, contrast agent, cortical and spongious bone. Forthe DECT images of the oval CTDI phantom the <strong>EDEC</strong>calibration was carried out <strong>for</strong> water and Aluminum. Forthe other two test phantoms the calibration phantoms werefilled with water and bone. The DECT scan was taken at80 and 140 kV. The bottom row of figure 5 further showsthe images that are obtained from a standard 120 kV scanand reconstruction. The tube currents were chosen a) toensure the same patient dose <strong>for</strong> the 120 kV scan as <strong>for</strong>the DECT scan (two spectra) together and b) to optimallydistribute the available dose among the 80 and 140 kVexposures (automatic exposure control AEC <strong>for</strong> DECT).The effects can be best seen regarding the oval CTDIphantom images in figure 5. The standard reconstructionat 120 kV shows significant beam hardening artifacts: darkbands of high amplitude that connect the Aluminum inserts.These artifacts should be completely removed afterrawdata–based DECT reconstruction (they would notbe removable if only image–based energy subtraction techniqueswere used).Obviously, the cylinder–in–cylinder calibration phantomproduces the least accurate coefficients: The dark bandsare gone but white bands of low amplitude appear at thesame place. The other two calibration phantoms, the half–and–half–cylinder and the three–cylinders phantom, arebetter suited <strong>for</strong> calibration. Here, rays through eitherone of the base materials as well as rays that run throughboth materials exist and the monochromatic images of thetest phantom are close to perfect. A very close look stillreveals some artifacts <strong>for</strong> the half–and–half–cylinder phantom.Probably this is due to the fact that the amount ofaluminum is much larger than what is actually required<strong>for</strong> the test phantom. The three–cylinders phantom’s Aluminumcontent is far smaller and closer to the amount ofAluminum contained in the CTDI test phantom.From these observations one can conclude that a suitabletest phantom must allow <strong>for</strong> rays that run in either materialas well as in both materials and the amount of basematerial in the calibration phantom should approximatethe expected imaging situation.Given that the three–cylinders phantom provides thebest image quality the quantitative values were analyzed,too. Figure 6 shows reconstructions of the oval CTDI phantomthat were calibrated with the three–cylinders phantom.The simulations were carried out at 120 and 140 kV,at 80 and 120 kV and at 80 and 140 kV. The decomposeddata were combined to yield µ–images at 70 keV, whichis the effective energy typical <strong>for</strong> clincial CT, at 511 keV,the energy that is important <strong>for</strong> PET attenuation correction,and they were combined to yield density images (ρ–images).The tube currents were selected similarly as be<strong>for</strong>e (AEC<strong>for</strong> DECT). We ensure the same patient dose <strong>for</strong> all threetube current combinations. We further balance the tubecurrent between the low and the high energy scan in a wayto minimize the variance in the final destination image.Note that this implies that different tube currents weresimulated <strong>for</strong> the 70 keV, <strong>for</strong> the 511 keV and <strong>for</strong> the ρ–images, altogether the figure shows nine different DECTscans.Two ROIs were place into each image. One ROI thatmeasures the mean and standard deviation within the centralAluminum insert and one ROI to determine the statistics<strong>for</strong> the phantom background (water). As demonstratedin figure 6 image noise decreases <strong>for</strong> tube voltage combinationsthat are far apart, i.e. it decreases from top to bottom.Since the total patient dose <strong>for</strong> all scans remained constantthe values are directly comparable.Regarding the mean values of the ROIs we immediately
Fig. 6. Quantitative results achieved at various tube voltage combinations(rows) <strong>for</strong> images of the attenuation coefficient at 70and 511 keV and <strong>for</strong> density images (columns). The tube currentwas chosen to keep the total patient dose <strong>for</strong> all three voltagepairs constant, hence the noise levels are directly comparable.The values given in the images are mean ± standard deviationmeasured in an ROI centered in the central Aluminum insert andin an ROI in the water background close to the upper right edgeof the phantom. The ratios of Aluminum and water values arevery close to the theoretical values. As expected, image noise islowest <strong>for</strong> tube voltage combinations that are far apart (here <strong>for</strong>the 80 and 140 kV combination).find that a very high accuracy is achieved. Water shouldbe at 1000 since the images are normalized to the theoreticalwater value. The deviation of the reconstructed watervalue is less 0.2% in all cases. Given the theoretical attenuationand density values we find that Aluminum shouldbe at 3224 <strong>for</strong> the 70 keV, at 2355 <strong>for</strong> the 511 keV and at2699 <strong>for</strong> the density image. The values actually measuredare very close to the theoretical predictions. Obviously, the25 calibration coefficients determined by <strong>EDEC</strong> from thethree–cylinders calibration phantom scans are highly accurateand our choice K = L = 4 appears to be sufficient.V. Summary and ConclusionThe empirical dual energy calibration is a simple, effectiveand accurate way to calibrate dual energy CT. It relieson a calibration phantom that must provide path lengthvariations through material 1, material 2 and combinationsof path lengths through both materials. In contrastto other rawdata–based calibration techniques the exactshape, position and orientation of the phantom does nothave to be known. <strong>EDEC</strong> determines the material decompositioncoefficients in the image domain. The coefficientsare then typically used in rawdata domain by passing pairsof attenuation values q 1 and q 2 (same ray at two tube voltages)through a simple polynomial:The rawdata p 1 <strong>for</strong> base material 1 and p 2 <strong>for</strong> base material2 then undergo the standard image reconstruction procedure(e.g. a Feldkamp–type cone–beam reconstruction).If desired, one may avoid products of q 1 and q 2 in thebase functions and may then per<strong>for</strong>m image–based DECTdecomposition (not shown in this paper). This kind ofenergy subtraction calibration is likely to be more efficientand more accurate than today’s procedure of simply havingtwo water–precorrected images (at two tube voltages) beinglinearly combined.We have demonstrated that the quality of <strong>EDEC</strong> decompositionslightly depends on the choice of the calibrationphantom and we have identified phantom designs that arewell suited. It was further shown that the sets of cofficientsobtained achieve precise quantitative results.Whenever detector position–dependent corrections arerequired where the spectrum w(L, E) varies with each rayL — this is, <strong>for</strong> example, the case with bow–tie prefiltration— one may combine <strong>EDEC</strong> with analytical precorrectionor material decomposition techniques and obtain a hybridapproach similar to the technique presented in reference[10].Altogether one may regard <strong>EDEC</strong> as being a simple andversatile method to obtain the desired material decompositionfunctions.References[1] R. Alvarez and A. Macovski, “<strong>Energy</strong>–selective reconstructionsin x–ray CT,” Phys. Med. Biol., vol. 21, no. 5, pp. 733–744,1976.[2] R. Alvarez and E. Seppi, “A comparison of noise and dose inconventional and energy selective computed tomography,” IEEETransactions on Nuclear Science, vol. NS–26, no. 2, pp. 2853–2856, Apr. 1979.[3] A. Coleman and M. Sinclair, “A beam–hardening correctionusing dual–energy computed tomography,” Phys. Med. Biol.,vol. 30, no. 11, pp. 1251–1256, 1985.[4] W. A. Kalender, W. Perman, J. Vetter, and E. Klotz, “Evaluationof a prototype dual–energy computed tomographic apparatus.I. Phantom studies,” Med. Phys., vol. 13, no. 3, pp. 334–339,May/June 1986.[5] J. Vetter, W. Perman, W. A. Kalender, R. Mazess, andJ. Holden, “Evaluation of a prototype dual–energy computedtomographic apparatus. II. Determination of vertebral bone mineralcontent,” Med. Phys., vol. 13, no. 3, pp. 340–343, May/June1986.[6] W. A. Kalender, E. Klotz, and L. Kostaridou, “An algorithm <strong>for</strong>noise suppression in dual energy CT material density images,”IEEE Transactions on Medical Imaging, vol. 7, no. 3, pp. 218–224, Sept. 1988.[7] K. Sourbelle, M. Kachelrieß, and W. A. Kalender, “<strong>Empirical</strong>water precorrection <strong>for</strong> cone–beam computed tomography,”IEEE Medical Imaging Conference Record, pp. 1871–1875, Oct.2005.[8] M. Kachelrieß, K. Sourbelle, and W. Kalender, “<strong>Empirical</strong> cuppingcorrection: A first order rawdata precorrection <strong>for</strong> cone–beam computed tomography,” Med. Phys., vol. 33, no. 5, pp.1269–1274, May 2006.[9] D. M. Tucker, G. T. Barnes, and D. P. Chakraborty, “Semiempiricalmodel <strong>for</strong> generating tungsten target x–ray spectra,” Med.Phys., vol. 18, no. 3, pp. 211–218, Mar./Apr. 1991.[10] K. Sourbelle, M. Kachelrieß, M. Karolczak, and W. A. Kalender,“Hybrid cupping correction (HCC) <strong>for</strong> quantitative cone–beamCT,” Radiology, vol. 237(P), p. 525, Nov. 2005.K∑ L∑p i = c in q1q k 2 l with n = k(L + 1) + l.k=0 l=0