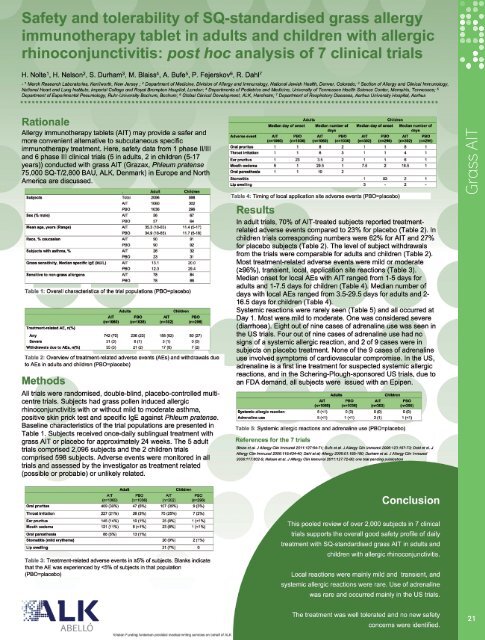
Safety and tolerability of SQ-standardised grass allergy immunotherapy tabletin adults and children with allergic rhinoconjunctivitis: post hoc analysis of7 clinical trialsH Nolte 1 , HS Nelson 2 , S Durham 3 , MS Blaiss 4 , A Bufe 5 , P Fejerskov 6 , R Dahl 71Merck & Co, Kenilworth, United States; 2 National Jewish Medical and Research Center, Denver, United States; 3 National Heart and Lung Institute, ImperialCollege, London, United Kingdom; 4 University of Tennessee Health Science Center, Memphis, United States; 5 Experimental Pneumology, Ruhr University Bochum,Bochum, Germany; 6 International Pharmacovigilance, <strong>ALK</strong>, Hørsholm, Denmark; 7 Aarhus University Hospital, Aarhus, DenmarkGrass AITBackgroundAllergy immunotherapy tablets (AITs) may provide a saferand more convenient alternative to subcutaneous specificimmunotherapy treatment. Here, we discuss safety datafrom 1 phase II/III and 6 phase III clinical trials (5 in adults, 2in children (5-17 years)) conducted with grass AIT (Grazax ® ,Phleum pratense, 75,000 SQ-T/2,800 BAU, <strong>ALK</strong>, Denmark)in Europe and North America.MethodAll trials were randomised, double-blind, placebocontrolled,multi-centre trials. The subjects had grass polleninduced allergic rhinoconjunctivitis with or without asthma,positive skin prick test and specific IgE against Phleumpratense. Subjects received once daily sublingual treatmentwith grass AIT or placebo for approximately 24 weeks. The5 adult trials comprised 2,096 subjects and the 2 childrentrials comprised 598 subjects. Adverse events weremonitored in all trials and assessed by the investigator astreatment related (possible or probable) or unlikely related.ResultIn the adult trials, 70% of the AIT treated subjects reportedtreatment related adverse events compared with 23% forthe placebo group. In the children trials, correspondingnumbers were 62% for AIT and 27% for placebo subjects.Most treatment related adverse events were mild ormoderate (≥96%), transient, local, application sitereactions (adults: oral pruritus (39%), throat irritation(21%), ear pruritus (14%); children: oral pruritus (35%),throat irritation (25%), stomatitis (mild erythema) (9%)).Median onset for local AEs with AIT ranged from 1-5 daysfor adults and 1-7.5 days for children. Median number ofdays with local AEs ranged from 3.5-29.5 days for adultsand 2-16.5 days for children. Systemic reactions were seenin
Grass AIT21