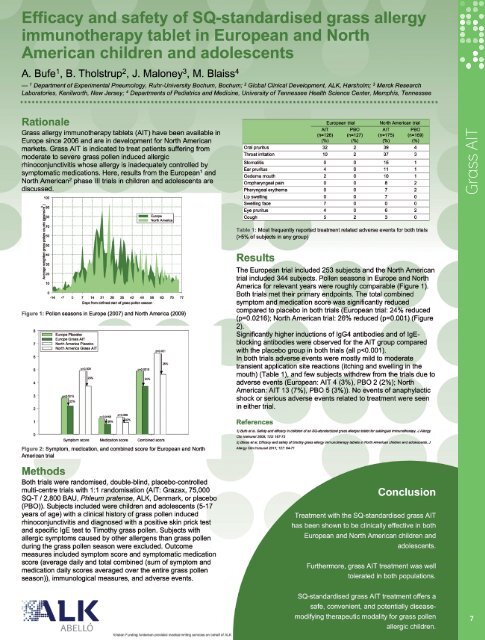
Efficacy and safety of SQ-standardised grass allergy immunotherapy tabletin European and North American children and adolescentsA Bufe 1 , B Tholstrup 2 , J Maloney 3 , MS Blaiss 41Experimental Pneumology, Ruhr University Bochum, Bochum, Germany; 2 Global Clinical Development, <strong>ALK</strong>, Hørsholm, Denmark; 3 Merck & Co, Kenilworth, UnitedStates; 4 University of Tennessee Health Science Center, Memphis, United StatesGrass AITBackgroundGrass allergy immunotherapy tablets (AITs) have beenavailable in Europe since 2006 and are in development forthe North American (NA) markets. Grass AIT is indicated totreat patients with moderate to severe grass pollen inducedallergic rhinoconjunctivitis whose allergy is inadequatelycontrolled by symptomatic medication. Here, we discussthe results from the European (EU) and NA phase III trials inchildren and adolescents.MethodBoth trials were randomised, double-blind, placebocontrolled,multi-centre trials with 1:1 randomisation (AIT:Grazax ® , Phleum pratense, 75,000 SQ-T/2,800 BAU, <strong>ALK</strong>,Denmark, or placebo). The subjects included were childrenand adolescents (5-17 years of age) with a clinical historyof (northern) grass pollen induced rhinoconjunctivitisand diagnosed with a positive skin prick test and specificIgE test to (northern) grass pollen. Subjects with allergicsymptoms caused by other allergens than grass pollenduring the grass pollen season were excluded. Outcomemeasures included symptom and medication scores(total combined and average daily), global evaluation/rhinoconjunctivitis quality of life (RQLQ), immunologicalmeasures and adverse events.ResultThe EU trial included 253 and the NA trial 344 subjects.Both trials met their primary endpoints. The total combinedsymptom and medication score was significantly reducedcompared with placebo in both trials (EU trial: 24% reduced(p=0.0216); NA trial: 26% reduced (p=0.001). The EUglobal evaluation showed that 74% of AIT subjects ratedan improved or much improved response compared withprevious pollen season (66% in placebo group, p=0.164).For the NA trial, an 18% reduction (p=0.042) relative toplacebo in RQLQ score was seen for AIT subjects during thegrass pollen season. Significantly higher inductions of IgG4antibodies and of IgE-blocking antibodies were observedfor the AIT group compared with the placebo group. Inboth trials, adverse events were mostly mild to moderatetransient application site reactions (itching and swellingin the mouth), and only few subjects withdrew from thetrials due to adverse events (EU: AIT group 3%, placebogroup 2%; NA: AIT group 7%, placebo group 3%). Systemicadverse events were rare in both trials.ConclusionSQ-standardised grass AIT treatment has been shown tobe clinically effective and well tolerated in both Europeanand North American children and adolescents.6Abstract number: 503. Session date and time: Sunday 12 June, 12.00 - 13.30Session: Poster session – Pediatric aspects of immunotherapy. Session room: Rumeli Hall
Grass AIT7