Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
Table 1I.Results otfeeding<br />
Animal<br />
Ni<br />
229<br />
II .pakai- .n d nymphon hrte rem<br />
State led<br />
r tNlll 30<br />
243 resulialil s<br />
23l utcecpliblc i,<br />
24! siiscejiiiptie .47<br />
251, sis1epihlc " 43<br />
Coniitrtl ra i t i<br />
( "o rtnd r,,hb it<br />
.<br />
Iur lies dislphiccl. iull litcks liM<br />
had moulted, to pick tip7.parva.The fail ore of 2/3 of the<br />
susceptibles to become infected also cast serious doubts on<br />
the infectivity of the ticks, and thus detracted from the<br />
apparently favourable outcome with the tick-resist ant<br />
cattle. There is,however, tit) doubt that we succceded in<br />
rendering the group I cattle resistant, which confirrins our<br />
findiniiethe ill li~ild that 11.illI('it s dlCchp -CistanoCe<br />
tituile sIsssl" uid less taikCtikh (h,tid ti h\ ,t te1t)1<br />
0,1IAN Ill IIAI ION AND (tARA(IFRISA I ION (11- ANI( d NS<br />
I-t( )M SAI.IVARfY i.ANt)S, R t 'ROII(VI IVI- SYSI I M.<br />
(itt ANt) tIAMt)IYNIt'lI it PAR IIAtH F I)<br />
I I N,1I.I V I (KS 0 - R/II'I( TI.I ,/S. ,'#'ll It<br />
Tlhe studies undertaken involved immunis,tion of animals<br />
with crude antigen extracts derived from unfed, partially fed<br />
or fully fed ticks. Initial information on protein components<br />
oh R?. aippendicrlatis organs producing the antigens<br />
responsible for the resistance response against ticks is<br />
described in this summnary,.<br />
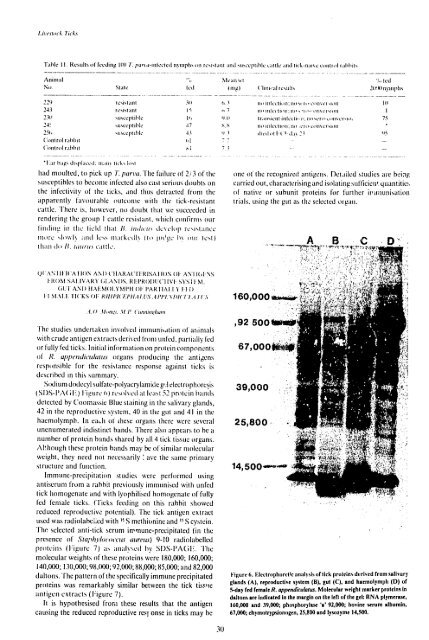
Soditit doidecyl sulfate-polyacrylamide g,:l electrophorcsis<br />
SI)S-PA( I-) :igure 0) relsolsedIt las,t 52 proIteit bands<br />
detected by C(omssie Blue staining in the salivary glands,<br />
42 in the reproductive system, 40 in the gut and 41 inthe<br />
haemolymph. Itneah of these organs there were several<br />
unenumerated indistinct hands. There also appears to be a<br />
number of protein bands shared by all 4 tick tissue organs.<br />
Although these protein hands may be of similar molecular<br />
weight, they need not necessarily ave the same primary<br />
structure and fuiction.<br />
Immune-precipitation sttidies were performed using<br />
antiserum from a rabbit previously immiunised with unfed<br />
tick homogenate and with lyophilised homogenate of fully<br />
fed female ticks. (Ticks feeding on this rabbit showed<br />
reduced reproductive potential). The tick antigen extract<br />
used was radiolabeLed with IsS methionine and 11S cystein.<br />
The selected anti-tick serum immune-precipitated (in the<br />
presence of Stap/hi'loco'cus aureao) 9-10 radiolabelled<br />
proteins (Figure 7) as analveld by SI)S-l'A(.L The<br />
molecular weights of these proteins were 180,000; 160,000;<br />
140,000; 130,000; 98,000; 92,000; 88,000; 85,000; and 82,000<br />
daltons. The pattern of the specifically immune precipitated<br />
proteins was remarkably similar between the tick tissie<br />
antigen extracts (Figure 7).<br />
It is hypothesised from these results that the antigen<br />
causing the reduced reproductive resI onse in ticks may be<br />
u 11 l ,tSCe'ibt li' 'attle'1odltick-mmi<br />
\1calI't<br />
4I1ig) ( litijil restil,<br />
i . no icci n IolaS lo<br />
, 7 n., ilctuil; [I' .1--<br />
7 -<br />
? .<br />
Crlr, I 10ti1,<br />
uclll t'01<br />
o i 'luc y<br />
0 I fu i t % nI n1c cll 'iin sercm-soin% ci<br />
X.S Io Ilnlslt l , no I i c- 'l l<br />
l hd t I '0 [Lis2l<br />
30<br />
2 1<br />
o', 75<br />
I.d<br />
) Iillpli<br />
one o lthe recognized antigetis. Detailed studies ale bei,-g<br />
carried out, characterising and isolating sufficien, quantitie,<br />
ol native or subunit proteins for further i',imunisatioit<br />
trials, using the gut as the selected otgait.<br />
160,000 wthwi,<br />
,92 500WM<br />
67,0000<br />
39000<br />
25,800,<br />
14,500- e<br />
.W1<br />
'15<br />
A B C D "<br />
i1 !<br />
Figure 6. Electrophoretic anallsis of tick priteins derived from salt vary<br />
glands (A), reproductive system (1), gut (C), and haemolymph (D) of<br />
I-day fed female R. appendiculatus. Molecular weight marker proteins In<br />
dallons areIndicated in themargin on tlie left of the gel: RNA plymerase,<br />
160,000 and 39,000; phoslphorylase 'a'92,000; bovine serum albumin,<br />
67,000; chymotrypslonogen, 25,800 and lysozyme 14,500.<br />
.,<br />
V