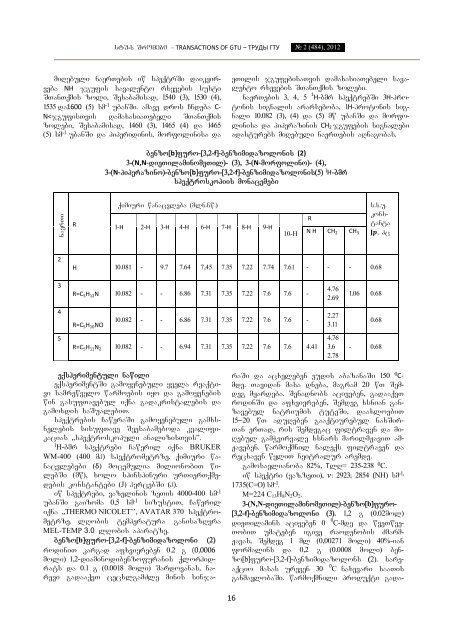
stu-s Sromebi – TRANSACTIONS OF GTU – ТРУДЫ ГТУ № 2 (484), 2012 miRebuli naerTebis iw speqtrSi daikvirveba NH jgufis savalento rxevebis susti STanTqmis zoli, Sesabamisad, 1540 (3), 1530 (4), 1535 da1600 (5) sm -1 ubanSi. amave dros Cndeba C‐ N
stu-s Sromebi – TRANSACTIONS OF GTU – ТРУДЫ ГТУ № 2 (484), 2012 aqvT wylian WiqaSi da amuSaveben amiakiani wyliT. warmoqmnil naleqs filtraven da recxaven wyliT neitralur aremde. gamosavlianoba 80% TlR=238-239 0C. iw speqtri (vaz.zeTi), : 1530 (NH ); 1700 (C=O); 1457 sm-1 (C-N) . M=309 C18H19N3O2. 3-(N‐morfolino)-benzo[b]furo-[3,2-f]benzimidazoloni (4). 1,2 g (0.02mol) morfolins aciveben 0 0 C-mde da wveTwveTobiT umateben igive raodenobis ZmarmJavas, Semdeg 1 ml ( 0,00271 moli) 40%-ian formalins da 0,2 g (0.0008 moli) benzo[b]furo-[3,2-f]-benzimidazolons (2). sareaqcio masas ureven 30 0 C naxevari saaTis ganmavlobaSi. warmoqmnili produqti gadaaqvT wylian WiqaSi da amuSaveben amiakiani wyliT. gamoyofil naleqs filtraven Sotis filtrze da recxaven wyliT neitralur reaqciamde. gamosavlianoba 82_85%, TlR=205-207 0C. iw speqtri, (vaz.zeTi), : 1528 sm-1 (NH) 1465 sm-1 (C-N). M=322 C18H17N3O3. 3-(N‐piperazino)-benzo[b]furo-[3,2-f]benzimidazoloni (5) 1,2 g (0.02mol) piperazins aciveben 0 0 C-mde da wveTwveTobiT umateben igive raodenobis ZmarmJavas, Semdeg 1 ml (0,00271 moli) 40%-ian formalins da 0,2 g (0.0008 moli) benzo[b]furo-[3,2-f]-benzimidazolons (2). sareaqcio masas ureven 30 0 C naxevari saaTis ganmavlobaSi. warmoqmnili produqti gadaaqvT wylian WiqaSi da amuSaveben amiakiani wyliT. damuSavebul produqts filtraven da recxaven neitralur aremde. gamosavali 83% TlR=135-137 0 C. iw speqtri (vaz.zeTi), :1530 1600 sm -1 (NH) 1470 sm -1 (C-N) . M=321 C18H19N4O2. 3. daskvna axali sinTezuri samkurnalo preparatebis SeqmnisaTvis mniSvnelovania cnobili biologiuri aqtiurobis mqone naerTebis modifikacia an gaerTianeba. am mizniT, axali, mosalodneli biologiuri aqtiurobis mqone niv- Tierebebis misaRebad dasinTezebulia benzo[b]furo[3,2-f]benzimidazolinebiswarmoebulebi dieTilaminTan, morfolinsa da piperazinTan. damuSavebulia maTi miRebis preparatuli meTodebi da Seswavlilia infrawiTeli da 1H-bmr speqtroskopiis saSualebiT. literatura 1. Солдатенко А.Т., Колядина Н.М., Шендрик Н.В. Основы органической химии лекарственных веществ. Москва: Химия, 2001. 2. Pozharsky A.F, Anisimova V.A, Tsupak V.A "Practical Works in The Chemistry of Heterocycles" Rostov 1988. 3. Симонов А.М., Анисимова В.А. Синтез и превра‐ щения 2‐аминобензимидазолов//ХГС, 1979, 7. С‐867. 4. M. Poje, B. Rocic, M. Sikirica, I. Vickovic, M. Bruvo; Oxidation of uric acid. 4.Synthesis, structure, and di‐ abetogenic action of 5‐imino‐2,4,6(1H,3H,5H)‐py‐ rimidinetrione salts and their alloxan‐like covalent adducts J. Med. Chem.,1983, 26 (6), pp 861–864 5. Влик Ф.Ф. Реакция Манниха. Органические реак‐ ции//Сб. 10 М., 1948, с. 146‐259. 6. Брестер Д.Х., Эшель Э.Д. Алкилирование аминами и аммонийными солями. Т. 7. М., 1956, с. 146‐259. 7. Hiremath S, Sidapp S. A conventient synthesis of aminotriptaminn analogous tu serotonin. J. Med. Chem., 1965, 8. P. 142‐143/ 8. Преображенская М.Н., Федотова М.В., Суворов Н.Н. Применение индолил‐индольного метода для синтеза производных триптамина и гете‐ роауксина//ЖОрХ, 1964, Т.34, вып. 4б. С 1310‐1314. 9. Федотова М.В., Суворов Н.Н., Огарева О.Б. Новый синтез 6‐метоксмитриптамина // ЖОрХ/Т. 30, 1960. UDC 66.098 SOME DERIVATIVES OF BENZO‐[B]‐FURO‐[3,2‐F]‐BENZIMIDAZOLONE OBTAINED WITH MANNICH REACTION M. Maisuradze*, N. Gakhokidze, S. Tsqvitaia Department of chemical and biological technologies, Georgian Technical University, 77, Kostava str, Tbilisi, 0175, Georgia Resume: There are considered new heterocyclic condensed systems on the base of benzo[b]furo‐[3,2‐f]‐ benzimidazolone and derivatives with expected biological activity 3‐(N,N‐diethilaminomethil)‐, 3‐(N‐morpholino)‐ 3‐ (N‐piperazino)‐benzo‐[b]‐furo‐[3,2‐f]‐benzimidazolone, that are obtained with Mannich reaction. Key words: dibenzofurane; benzimidazolone; Manich reaction 17