TerraTox - HIV-1 structure file, May 2004. Copyright ... - TerraBase Inc.
TerraTox - HIV-1 structure file, May 2004. Copyright ... - TerraBase Inc.
TerraTox - HIV-1 structure file, May 2004. Copyright ... - TerraBase Inc.
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
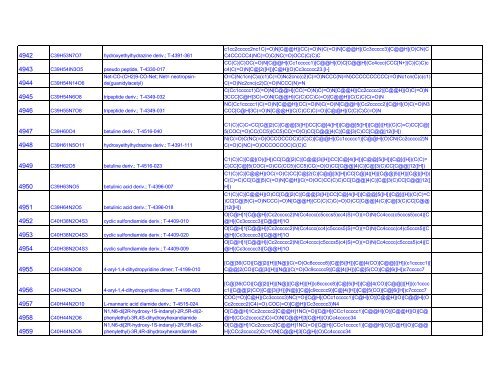
4942 C39H53N7O7 hydroxyethylhydrazine deriv.; T-4391-361<br />
4945 C39H54N6O8 tripeptide deriv.; T-4349-032<br />
4946 C39H55N7O8 tripeptide deriv.; T-4349-031<br />
4947 C39H60O4 betuline deriv.; T-4516-040<br />
4948 C39H61N5O11 hydroxyethylhydrazine deriv.; T-4391-111<br />
4949 C39H62O5 betuline deriv.; T-4516-023<br />
4950 C39H63NO5 betulinic acid deriv.; T-4396-007<br />
4951 C39H64N2O5 betulinic acid deriv.; T-4396-018<br />
4952 C40H38N2O4S3 cyclic sulfondiamide deriv.; T-4409-010<br />
4953 C40H38N2O4S3 cyclic sulfondiamide deriv.; T-4409-020<br />
4954 C40H38N2O4S3 cyclic sulfondiamide deriv.; T-4409-009<br />
4955 C40H38N2O8 4-aryl-1,4-dihydropyridine dimer; T-4199-010<br />
4956 C40H42N2O4 4-aryl-1,4-dihydropyridine dimer; T-4199-003<br />
4943 C39H54IN3O5 pseudo peptide, T-4330-017<br />
Net-CO-(CH2)9-CO-Net; Net= neotropsinde(guanidylacetyl)<br />
4944 C39H54N14O6<br />
4957 C40H44N2O10 L-mannaric acid diamide deriv.; T-4515-024<br />
N1,N6-di[2R-hydroxy-1S-indanyl)-2R,5R-di(2-<br />
4958 C40H44N2O6 phenylethyl)-3R,4S-dihydroxyhexandiamide<br />
N1,N6-di[2R-hydroxy-1S-indanyl)-2R,5R-di(2-<br />
4959 C40H44N2O6 phenylethyl)-3R,4R-dihydroxyhexandiamide<br />
c1cc2ccccc2nc1C(=O)N[C@@H](CC(=O)N)C(=O)N[C@@H](Cc3ccccc3)[C@@H](O)CN(C<br />
C4CCCCC4)NC(=O)C(NC(=O)OCC)C(C)C<br />
CC(C)(C)OC(=O)N[C@@H](Cc1ccccc1)[C@@H](O)C[C@@H](Cc4ccc(CCC[N+](C)(C)C)c<br />
c4)C(=O)N[C@]2([H])[C@H](O)Cc3ccccc23.[I-]<br />
O=C(Nc1cn(C)c(c1)C(=O)Nc2cnc(c2)C(=O)NCCC(N)=N)CCCCCCCCCC(=O)Nc1cn(C)c(c1)<br />
C(=O)Nc2cnc(c2)C(=O)NCCC(N)=N<br />
C(Cc1ccccc1)C(=O)N[C@@H](CC(=O)N)C(=O)N[C@@H](Cc2ccccc2)[C@@H](O)C(=O)N<br />
3CCC[C@H]3C(=O)N[C@@H](C(C)CC)C(=O)[C@@H](C(C)C)C(=O)N<br />
NC(Cc1ccccc1)C(=O)N[C@@H](CC(=O)N)C(=O)N[C@@H](Cc2ccccc2)[C@H](O)C(=O)N3<br />
CCC[C@H]3C(=O)N[C@@H](C(C)CC)C(=O)[C@@H](C(C)C)C(=O)N<br />
C1(C)(C)C=CC[C@]2(C)[C@@]3([H])CC[C@]4([H])[C@@]5([H])[C@]([H])(C(C)=C)CC[C@]<br />
5(COC(=O)CC(CC5)(CC5)CC(=O)O)CC[C@@]4(C)[C@]3(C)CC[C@@]12([H])<br />
N(C(=O)C(NC(=O)OCCOCCOC)C(C)C)[C@@H](Cc1ccccc1)[C@@H](O)CN(Cc2ccccc2)N<br />
C(=O)C(NC(=O)OCCOCCOC)C(C)C<br />
C1(C)(C)[C@](O)([H])CC[C@]2(C)[C@@]3([H])CC[C@]4([H])[C@@]5([H])[C@]([H])(C(C)=<br />
C)CC[C@]5(COC(=O)CC(CC5)(CC5)CC(=O)O)CC[C@@]4(C)[C@]3(C)CC[C@@]12([H])<br />
C1(C)(C)[C@@H](OC(=O)C)CC[C@]2(C)[C@@]3([H])CC[C@]4([H])[C@@]5([H])[C@]([H])(<br />
C(C)=C)CC[C@]5(C(=O)N[C@H](C(=O)OC)CC(C)C)CC[C@@]4(C)[C@]3(C)CC[C@@]12([<br />
H])<br />
C1(C)(C)[C@@H](O)CC[C@]2(C)[C@@]3([H])CC[C@]4([H])[C@@]5([H])[C@]([H])(C(C)=C<br />
)CC[C@]5(C(=O)NCCC(=O)N[C@@H](CC(C)C)C(=O)O)CC[C@@]4(C)[C@]3(C)CC[C@@<br />
]12([H])<br />
O[C@H]1[C@@H](Cc2ccccc2)N(Cc4ccc(c5cccs5)cc4)S(=O)(=O)N(Cc4ccc(c5cccs5)cc4)[C<br />
@H](Cc3ccccc3)[C@@H]1O<br />
O[C@H]1[C@@H](Cc2ccccc2)N(Cc4ccc(cc4)c5cccs5)S(=O)(=O)N(Cc4cccc(c4)c5cccs5)[C<br />
@H](Cc3ccccc3)[C@@H]1O<br />
O[C@H]1[C@@H](Cc2ccccc2)N(Cc4cccc(c5cccs5)c4)S(=O)(=O)N(Cc4cccc(c5cccs5)c4)[C<br />
@H](Cc3ccccc3)[C@@H]1O<br />
[C@]36(CO)[C@]2([H])[N@](C(=O)Oc8ccccc8)[C@]5([H])[C@]4(CO)[C@@]([H])(c1ccccc1)[<br />
C@@]2(CO)[C@]3([H])[N@](C(=O)Oc9ccccc9)[C@]4([H])[C@]5(CO)[C@]6([H])c7ccccc7<br />
[C@]36(CO)[C@]2([H])[N@]([C@H]([H])c8ccccc8)[C@]5([H])[C@]4(CO)[C@@]([H])(c1cccc<br />
c1)[C@@]2(CO)[C@]3([H])[N@]([C@]c9ccccc9)[C@]4([H])[C@]5(CO)[C@]6([H])c7ccccc7<br />
COC(=O)[C@H](Cc3ccccc3)NC(=O)[C@H](OCc1ccccc1)[C@H](O)[C@@H](O)[C@@H](O<br />
Cc2ccccc2)C4(=O).COC(=O)[C@H](Cc3ccccc3)N4<br />
O[C@@H]1Cc2ccccc2[C@@H]1NC(=O)[C@H](CCc1ccccc1)[C@@H](O)[C@@H](O)[C@<br />
@H](CCc2ccccc2)C(=O)N[C@@H]3[C@H](O)Cc4ccccc34<br />
O[C@@H]1Cc2ccccc2[C@@H]1NC(=O)[C@H](CCc1ccccc1)[C@@H](O)[C@H](O)[C@@<br />
H](CCc2ccccc2)C(=O)N[C@@H]3[C@H](O)Cc4ccccc34