TerraTox - HIV-1 structure file, May 2004. Copyright ... - TerraBase Inc.
TerraTox - HIV-1 structure file, May 2004. Copyright ... - TerraBase Inc.
TerraTox - HIV-1 structure file, May 2004. Copyright ... - TerraBase Inc.
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
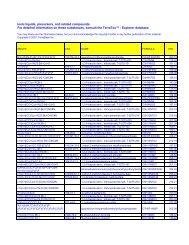
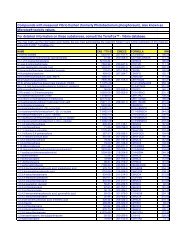
5168 C48H82N2O5 betulinic acid deriv.; T-4396-017<br />
5169 C49H44N8O5 SD-146<br />
5170 C49H56N18O8<br />
Dist-CO-(trans-1,2-bicyclo[2.2.1]hept-4-ene)-CO-Dist;<br />
Dist= distamicynyl<br />
C1(C)(C)[C@@H](O)CC[C@]2(C)[C@@]3([H])CC[C@]4([H])[C@@]5([H])[C@]([H])(C(C)=C<br />
)CC[C@]5(C(=O)NCCCCCCCCCCC(=O)N[C@@H](CC(C)C)C(=O)OC)CC[C@@]4(C)[C@]<br />
3(C)CC[C@@]12([H])<br />
O[C@H]1[C@@H](Cc2ccccc2)N(Cc4cc(C(=O)Nc6nc7ccccc7n6)ccc4)C(=O)N(Cc4cc(C(=O)<br />
Nc6nc7ccccc7n6)ccc4)[C@H](Cc3ccccc3)[C@@H]1O<br />
O=C(Nc1cn(C)c(c1)C(=O)Nc2cn(C)c(c2)C(=O)Nc3cnc(c3)C(=O)NCCC(N)=N)[C@@H]4(C5<br />
C=CC(C5)[C@H]4C(=O)Nc1cn(C)c(c1)C(=O)Nc2cn(C)c(c2)C(=O)Nc3cnc(c3)C(=O)NCCC(<br />
N)=N<br />
5171 C49H68N6O7 Indinavir-(Prd)-(Boc)Val(14)<br />
c1ncccc1CN2CCN([C@@H](C2)C(=O)NC(C)(C)C)C[C@@H](OC(=O)C(C(C)C)NC(=O)OC(<br />
C)(C)C)C[C@@H](Cc3ccccc3)C(=O)N(C6(C)C)[C@@H]4[C@H](O6)Cc5ccccc45<br />
5172 C50H64N18O8 Dist-CO-(CH2)8-CO-Dist; Dist= distamicynyl<br />
5173 C50H64N6O10 1,6-diphenyl-3,4-diol deriv.; T-4679-012<br />
5174 C50H70O11<br />
5175 C50H73N5O5 Indinavir-Myr(8)<br />
3,28-di-O-(3',4',5'-trimethoxybenzoyl)-dihydrobetulinic<br />
acid<br />
5176 C50H74O8 betuline deriv.; T-4516-013<br />
5177 C50H75N9O8 <strong>HIV</strong> protease inhibitor; 4069-001<br />
5178 C50H76O8 dihydrobetuline deriv.; T-4516-021<br />
5179 C50H82O9 phorbol deriv.; T-4227-084<br />
O=C(Nc1cn(C)c(c1)C(=O)Nc2cn(C)c(c2)C(=O)Nc3cnc(c3)C(=O)NCCC(N)=N)CCCCCCCCC<br />
(=O)Nc1cn(C)c(c1)C(=O)Nc2cn(C)c(c2)C(=O)Nc3cnc(c3)C(=O)NCCC(N)=N<br />
N(C(=O)C(NC(=O)CN(C)C(=O)OCc3ccccc3)C(C)C)[C@@H](Cc1ccccc1)[C@@H](O)[C@H<br />
](O)[C@H](Cc2ccccc2)NC(=O)C(NC(=O)C(C)NC(=O)OCc4ccccc4)C(C)C<br />
C1(C)(C)[C@](OC(=O)c6cc(OC)c(OC)c(OC)c6)([H])CC[C@]2(C)[C@@]3([H])CC[C@]4([H])[<br />
C@@]5([H])[C@]([H])(C(C)C)CC[C@]5(C(=O)OC(=O)c7cc(OC)c(OC)c(OC)c7)CC[C@@]4(<br />
C)[C@]3(C)CC[C@@]12([H])<br />
c1ncccc1CN2CCN([C@@H](C2)C(=O)NC(C)(C)C)C[C@@H](O)C[C@@H](Cc3ccccc3)C(=<br />
O)N[C@@H]4[C@H](OC(=O)CCCCCCCCCCCCC)Cc5ccccc45<br />
C1(C)(C)[C@](OC(=O)C56OC(=O)C(C)(C5(C)C)CC6)([H])CC[C@]2(C)[C@@]3([H])CC[C@<br />
]4([H])[C@@]5([H])[C@]([H])(C(C)=C)CC[C@]5(COC(=O)C56OC(=O)C(C)(C5(C)C)CC6)CC<br />
[C@@]4(C)[C@]3(C)CC[C@@]12([H])<br />
CC(C)(C)OC(=O)N1CCCC1C(=O)N[C@@H](Cc2ccccc2)C(=O)N(C)[C@@H](Cc3cncn3)C(<br />
=O)N[C@@H](CC(C)C)[C@H](O)C[C@H](C(C)C)C(=O)N[C@@H](C(C)CC)C(=O)NCc4ncc<br />
cc4<br />
C1(C)(C)[C@](OC(=O)C56OC(=O)C(C)(C5(C)C)CC6)([H])CC[C@]2(C)[C@@]3([H])CC[C@<br />
]4([H])[C@@]5([H])[C@]([H])(C(C)C)CC[C@]5(COC(=O)C56OC(=O)C(C)(C5(C)C)CC6)CC[<br />
C@@]4(C)[C@]3(C)CC[C@@]12([H])<br />
O=C1C(C)=C[C@@]2([H])[C@@]3(O)[C@H](C)[C@@H](OC(=O)C)[C@@](OC(=O)CCCC<br />
CCCCCCCCC)(C4(C)C)[C@]4([H])[C@]3([H])C=C(COC(=O)CCCCCCCCCCCCC)C[C@]12<br />
O<br />
5180 C51H40N6O23S6 suramin<br />
5181 C51H58O14 3,7,12-tricaffeoylcholic acid<br />
C1=CC=C(C=C1C(=O)NC=4C=C(C(=O)NC=3C2=C([S](O)(=O)=O)C=C([S](O)(=O)=O)C=C2<br />
C([S](=O)(O)=O)=CC=3)C=CC=4C)NC(NC=5C=C(C=CC=5)C(NC=8C(C)=CC=C(C(NC7=C6<br />
C([S](O)(=O)=O)=CC([S](O)(=O)=O)=CC6=C([S](O)(=O)=O)C=C7)=O)C=8)=O)=O 145-63-1<br />
C1[C@H](OC(=O)/C=C/c5ccc(O)c(O)c5)CC[C@]2(C)[C@H]3C[C@H](OC(=O)/C=C/c6ccc(<br />
O)c(O)c6)[C@]4(C)C(C(C)CCC(=O)O)CC[C@H]4[C@@H]3[C@H](OC(=O)/C=C/c7ccc(O)c(<br />
O)c7)C[C@@H]12