TNT: Trinitrotoluenes and Mono and Dinitrotoluenes
TNT: Trinitrotoluenes and Mono and Dinitrotoluenes
TNT: Trinitrotoluenes and Mono and Dinitrotoluenes
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
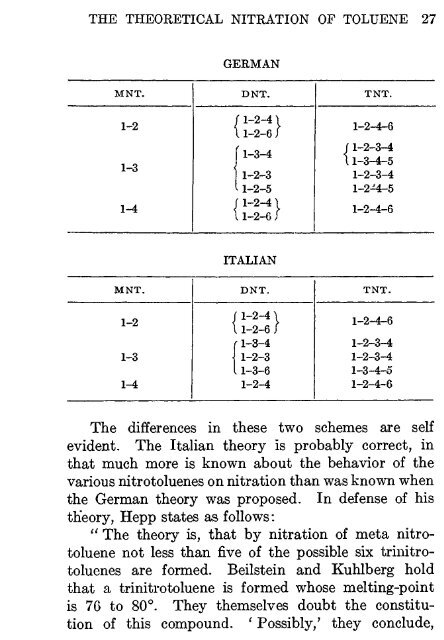
THE THEORETICAL NITRATION OF TOLUENE 27<br />
MNT.<br />
1-2<br />
1-3<br />
1-4<br />
MNT.<br />
1-2<br />
1-3<br />
1-4<br />
GERMAN<br />
DNT.<br />
r1-2-41<br />
\ 1-2-6 /<br />
11-3-4<br />
I 1-2-3<br />
11-2-5<br />
f1-2-41<br />
11-2-6 i<br />
ITALIAN<br />
DNT.<br />
r1-2-41<br />
11-2-6 J<br />
r 1-3-4<br />
| 1-2-3<br />
11-3-6<br />
1-2-4<br />
<strong>TNT</strong>.<br />
1-2-4-6<br />
(1-2-3-4<br />
11-3-4-5<br />
1-2-3-4<br />
1-2-4-5<br />
1-2-4-6<br />
<strong>TNT</strong>.<br />
1-2-4-6<br />
1-2-3-4<br />
1-2-3-4<br />
1-3-4-5<br />
1-2-4-6<br />
The differences in these two schemes are self<br />
evident. The Italian theory is probably correct, in<br />
that much more is known about the behavior of the<br />
various nitrotoluenes on nitration than was known when<br />
the German theory was proposed. In defense of his<br />
theory, Hepp states as follows:<br />
" The theory is, that by nitration of meta nitrotoluene<br />
not less than five of the possible six trinitrotoluenes<br />
are formed. Beilstein <strong>and</strong> Kuhlberg hold<br />
that a trinitrotoluene is formed whose melting-point<br />
is 76 to 80°. They themselves doubt the constitution<br />
of this compound. ' Possibly/ they conclude,