Brochure for Business Partners - Kyocera
Brochure for Business Partners - Kyocera
Brochure for Business Partners - Kyocera
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
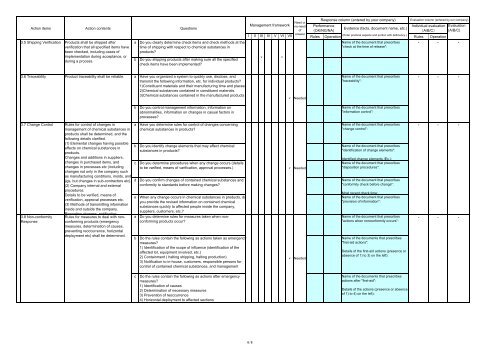
Action items Action contents Questions<br />
3.5 Shipping Verification<br />
3.6 Traceability Product traceability shall be reliable.<br />
3.7 Change Control<br />
3.8 Non-con<strong>for</strong>mity<br />
Response<br />
Products shall be shipped after<br />
verification that all specified items have<br />
been checked, including cases of<br />
implementation during acceptance, or<br />
during a process.<br />
Rules <strong>for</strong> control of changes in<br />
management of chemical substances in<br />
products shall be determined, and the<br />
following details clarified.<br />
(1) Elemental changes having possible<br />
effects on chemical substances in<br />
products.<br />
Changes and additions in suppliers,<br />
changes in purchased items, and<br />
changes in processes etc (including<br />
changes not only in the company such<br />
as manufacturing conditions, molds, and<br />
jigs, but changes in sub-contractors etc).<br />
(2) Company internal and external<br />
procedures.<br />
Details to be verified, means of<br />
verification, approval processes etc.<br />
(3) Methods of transmitting in<strong>for</strong>mation<br />
inside and outside the company.<br />
Recording changes notification<br />
Rules <strong>for</strong> measures to deal with noncon<strong>for</strong>ming<br />
products (emergency<br />
measures, determination of causes,<br />
preventing reoccurrence, horizontal<br />
deployment etc) shall be determined.<br />
a Do you clearly determine check items and check methods at the<br />
time of shipping with respect to chemical substances in<br />
products?<br />
b Do you shipping products after making sure all the specified<br />
check items have been implemented?<br />
a Have you organized a system to quickly use, disclose, and<br />
transmit the following in<strong>for</strong>mation, etc. <strong>for</strong> individual products?<br />
1)Constituent materials and their manufacturing time and places.<br />
2)Chemical substances contained in constituent materials.<br />
3)Chemical substances contained in the manufactured products<br />
b Do you control management in<strong>for</strong>mation, in<strong>for</strong>mation on<br />
abnormalities, in<strong>for</strong>mation on changes in casual factors in<br />
processes?<br />
a Have you determine rules <strong>for</strong> control of changes concerning<br />
chemical substances in products?<br />
b Do you identify change elements that may affect chemical<br />
substances in products?<br />
c Do you determine procedures when any change occurs (details<br />
to be varified, means of varification, approval processes.)<br />
d Do you confirm changes of contained chemical substances and<br />
con<strong>for</strong>mity to standards be<strong>for</strong>e making changes?<br />
e When any change occurs in chemical substances in products, do<br />
you provide the revised in<strong>for</strong>mation on contained chemical<br />
substances quickly to affected people inside the company,<br />
suppliers, customers, etc.?<br />
a Do you determine rules <strong>for</strong> measures taken when noncon<strong>for</strong>ming<br />
products occur?<br />
b Do the rules contain the following as actions taken as emergency<br />
measures?<br />
1) Identification of the scope of influence (identification of the<br />
affected lot, equipment involved, etc.)<br />
2) Containment ( halting shipping, halting production)<br />
3) Notification to in-house, customers, responsible persons <strong>for</strong><br />
control of contained chemical substances, and management<br />
c Do the rules contain the following as actions after emergency<br />
measures?<br />
1) Identification of causes<br />
2) Determination of necessary measures<br />
3) Prevention of reoccurrence<br />
4) Horizontal deployment to affected sections<br />
Management framework<br />
Need or<br />
no-need<br />
of<br />
Response column (entered by your company)<br />
Per<strong>for</strong>mance<br />
Evidence (facts, document name, etc.)<br />
(OK/NG/NA)<br />
Evaluation column (entered by our company)<br />
Individual evaluation Evaluation<br />
(A/B/C) (A/B/C)<br />
I II III IV V VI VII<br />
answer<br />
Rules<br />
(Enter practical aspects and portion with deficiency.)<br />
Operation Rules Operation<br />
Name of the document that prescribes<br />
"check at the time of release":<br />
- - -<br />
6/8<br />
v v<br />
v Needed<br />
v<br />
v<br />
Needed<br />
Needed<br />
Name of the document that prescribes<br />
"traceability":<br />
Name of the document that prescribes<br />
"in<strong>for</strong>mation control":<br />
Name of the document that prescribes<br />
"change control":<br />
Name of the document that prescribes<br />
"identification of change elements":<br />
Identified change elements (Ex.):<br />
Name of the document that prescribes<br />
"disposition procedures":<br />
Name of the document that prescribes<br />
"con<strong>for</strong>mity check be<strong>for</strong>e change":<br />
Most recent check time:<br />
Name of the document that prescribes<br />
"provision of in<strong>for</strong>mation":<br />
Name of the document that prescribes<br />
"actions when noncon<strong>for</strong>mity occurs":<br />
Name of the documents that prescribes<br />
"first-aid actions":<br />
Details of the first-aid actions (presence or<br />
absence of 1) to 3) on the left):<br />
Name of the documents that prescribes<br />
actions after "first-aid":<br />
Details of the actions (presence or absence<br />
of 1) to 4) on the left):<br />
-<br />
- -<br />
- - -<br />
- -<br />
-