Cleveland Clinic Brain Tumor and Neuro-Oncology Center Clinical ...
Cleveland Clinic Brain Tumor and Neuro-Oncology Center Clinical ...
Cleveland Clinic Brain Tumor and Neuro-Oncology Center Clinical ...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
<strong>Clevel<strong>and</strong></strong> <strong>Clinic</strong> <strong>Brain</strong> <strong>Tumor</strong> <strong>and</strong> <strong>Neuro</strong>-<strong>Oncology</strong> <strong>Center</strong>:Patient Data<br />
http://bti.ccf.org/cgi-bin/bti/auth/detailLAMINATE.cgi?id=613<br />
Page 3 of 4<br />
4/11/2013<br />
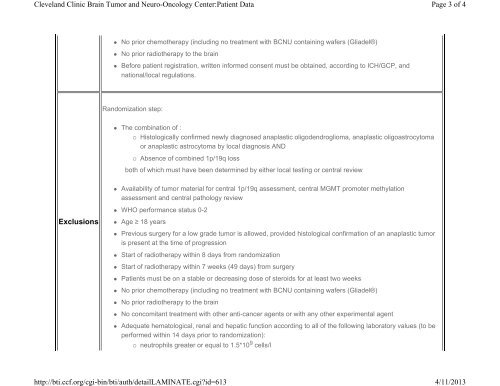
• No prior chemotherapy (including no treatment with BCNU containing wafers (Gliadel®)<br />
• No prior radiotherapy to the brain<br />
• Before patient registration, written informed consent must be obtained, according to ICH/GCP, <strong>and</strong><br />
national/local regulations.<br />
R<strong>and</strong>omization step:<br />
• The combination of :<br />
◦ Histologically confirmed newly diagnosed anaplastic oligodendroglioma, anaplastic oligoastrocytoma<br />
or anaplastic astrocytoma by local diagnosis AND<br />
◦ Absence of combined 1p/19q loss<br />
both of which must have been determined by either local testing or central review<br />
• Availability of tumor material for central 1p/19q assessment, central MGMT promoter methylation<br />
assessment <strong>and</strong> central pathology review<br />
• WHO performance status 0-2<br />
Exclusions<br />
• Age ≥ 18 years<br />
• Previous surgery for a low grade tumor is allowed, provided histological confirmation of an anaplastic tumor<br />
is present at the time of progression<br />
• Start of radiotherapy within 8 days from r<strong>and</strong>omization<br />
• Start of radiotherapy within 7 weeks (49 days) from surgery<br />
• Patients must be on a stable or decreasing dose of steroids for at least two weeks<br />
• No prior chemotherapy (including no treatment with BCNU containing wafers (Gliadel®)<br />
• No prior radiotherapy to the brain<br />
• No concomitant treatment with other anti-cancer agents or with any other experimental agent<br />
• Adequate hematological, renal <strong>and</strong> hepatic function according to all of the following laboratory values (to be<br />
performed within 14 days prior to r<strong>and</strong>omization):<br />
◦ neutrophils greater or equal to 1.5*10 9 cells/l