Sumoylation in Aspergillus nidulans: sumO inactivation ...
Sumoylation in Aspergillus nidulans: sumO inactivation ...
Sumoylation in Aspergillus nidulans: sumO inactivation ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
732 K.H. Wong et al. / Fungal Genetics and Biology 45 (2008) 728–737<br />
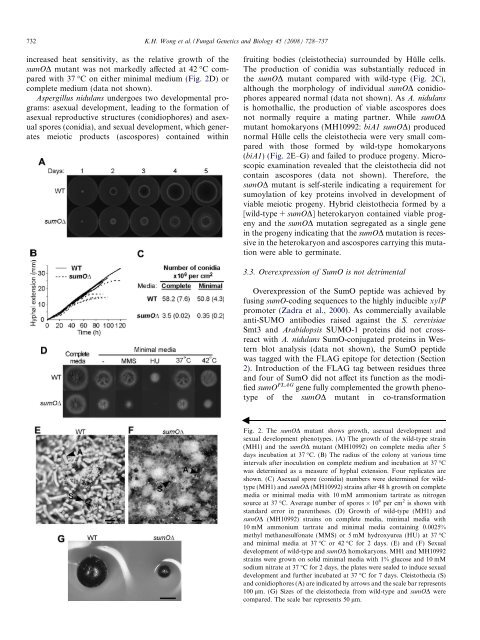
<strong>in</strong>creased heat sensitivity, as the relative growth of the<br />
<strong>sumO</strong>D mutant was not markedly affected at 42 °C compared<br />
with 37 °C on either m<strong>in</strong>imal medium (Fig. 2D) or<br />
complete medium (data not shown).<br />
<strong>Aspergillus</strong> <strong>nidulans</strong> undergoes two developmental programs:<br />
asexual development, lead<strong>in</strong>g to the formation of<br />
asexual reproductive structures (conidiophores) and asexual<br />
spores (conidia), and sexual development, which generates<br />
meiotic products (ascospores) conta<strong>in</strong>ed with<strong>in</strong><br />
fruit<strong>in</strong>g bodies (cleistothecia) surrounded by Hülle cells.<br />
The production of conidia was substantially reduced <strong>in</strong><br />
the <strong>sumO</strong>D mutant compared with wild-type (Fig. 2C),<br />
although the morphology of <strong>in</strong>dividual <strong>sumO</strong>D conidiophores<br />
appeared normal (data not shown). As A. <strong>nidulans</strong><br />
is homothallic, the production of viable ascospores does<br />
not normally require a mat<strong>in</strong>g partner. While <strong>sumO</strong>D<br />
mutant homokaryons (MH10992: biA1 <strong>sumO</strong>D) produced<br />
normal Hülle cells the cleistothecia were very small compared<br />
with those formed by wild-type homokaryons<br />
(biA1) (Fig. 2E–G) and failed to produce progeny. Microscopic<br />
exam<strong>in</strong>ation revealed that the cleistothecia did not<br />
conta<strong>in</strong> ascospores (data not shown). Therefore, the<br />
<strong>sumO</strong>D mutant is self-sterile <strong>in</strong>dicat<strong>in</strong>g a requirement for<br />
sumoylation of key prote<strong>in</strong>s <strong>in</strong>volved <strong>in</strong> development of<br />
viable meiotic progeny. Hybrid cleistothecia formed by a<br />
[wild-type + <strong>sumO</strong>D] heterokaryon conta<strong>in</strong>ed viable progeny<br />
and the <strong>sumO</strong>D mutation segregated as a s<strong>in</strong>gle gene<br />
<strong>in</strong> the progeny <strong>in</strong>dicat<strong>in</strong>g that the <strong>sumO</strong>D mutation is recessive<br />
<strong>in</strong> the heterokaryon and ascospores carry<strong>in</strong>g this mutation<br />
were able to germ<strong>in</strong>ate.<br />
3.3. Overexpression of SumO is not detrimental<br />
Overexpression of the SumO peptide was achieved by<br />
fus<strong>in</strong>g <strong>sumO</strong>-cod<strong>in</strong>g sequences to the highly <strong>in</strong>ducible xylP<br />
promoter (Zadra et al., 2000). As commercially available<br />
anti-SUMO antibodies raised aga<strong>in</strong>st the S. cerevisiae<br />
Smt3 and Arabidopsis SUMO-1 prote<strong>in</strong>s did not crossreact<br />
with A. <strong>nidulans</strong> SumO-conjugated prote<strong>in</strong>s <strong>in</strong> Western<br />
blot analysis (data not shown), the SumO peptide<br />
was tagged with the FLAG epitope for detection (Section<br />
2). Introduction of the FLAG tag between residues three<br />
and four of SumO did not affect its function as the modified<br />
<strong>sumO</strong> FLAG gene fully complemented the growth phenotype<br />
of the <strong>sumO</strong>D mutant <strong>in</strong> co-transformation<br />
3<br />
Fig. 2. The <strong>sumO</strong>D mutant shows growth, asexual development and<br />
sexual development phenotypes. (A) The growth of the wild-type stra<strong>in</strong><br />
(MH1) and the <strong>sumO</strong>D mutant (MH10992) on complete media after 5<br />
days <strong>in</strong>cubation at 37 °C. (B) The radius of the colony at various time<br />
<strong>in</strong>tervals after <strong>in</strong>oculation on complete medium and <strong>in</strong>cubation at 37 °C<br />
was determ<strong>in</strong>ed as a measure of hyphal extension. Four replicates are<br />
shown. (C) Asexual spore (conidia) numbers were determ<strong>in</strong>ed for wildtype<br />
(MH1) and <strong>sumO</strong>D (MH10992) stra<strong>in</strong>s after 48 h growth on complete<br />
media or m<strong>in</strong>imal media with 10 mM ammonium tartrate as nitrogen<br />
source at 37 °C. Average number of spores 10 6 per cm 2 is shown with<br />
standard error <strong>in</strong> parentheses. (D) Growth of wild-type (MH1) and<br />
<strong>sumO</strong>D (MH10992) stra<strong>in</strong>s on complete media, m<strong>in</strong>imal media with<br />
10 mM ammonium tartrate and m<strong>in</strong>imal media conta<strong>in</strong><strong>in</strong>g 0.0025%<br />
methyl methanesulfonate (MMS) or 5 mM hydroxyurea (HU) at 37 °C<br />
and m<strong>in</strong>imal media at 37 °C or42°C for 2 days. (E) and (F) Sexual<br />
development of wild-type and <strong>sumO</strong>D homokaryons. MH1 and MH10992<br />
stra<strong>in</strong>s were grown on solid m<strong>in</strong>imal media with 1% glucose and 10 mM<br />
sodium nitrate at 37 °C for 2 days, the plates were sealed to <strong>in</strong>duce sexual<br />
development and further <strong>in</strong>cubated at 37 °C for 7 days. Cleistothecia (S)<br />
and conidiophores (A) are <strong>in</strong>dicated by arrows and the scale bar represents<br />
100 lm. (G) Sizes of the cleistothecia from wild-type and <strong>sumO</strong>D were<br />
compared. The scale bar represents 50 lm.