HACCP Plan Pork, Sheep, Goat Slaughter 04/11/2003 Version 1
HACCP Plan Pork, Sheep, Goat Slaughter 04/11/2003 Version 1
HACCP Plan Pork, Sheep, Goat Slaughter 04/11/2003 Version 1
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
<strong>HACCP</strong> <strong>Plan</strong> <strong>Pork</strong>, <strong>Sheep</strong>, <strong>Goat</strong> <strong>Slaughter</strong><br />
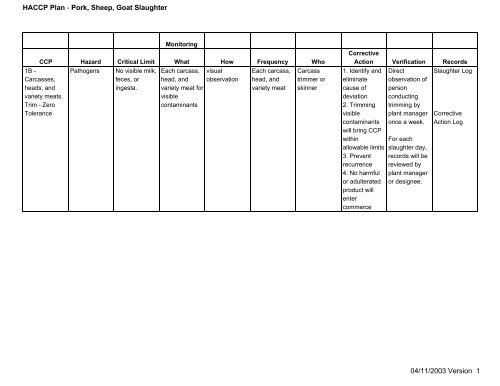
Monitoring<br />
CCP Hazard Critical Limit What How Frequency Who<br />
1B <br />
Carcasses,<br />
heads, and<br />
variety meats.<br />
Trim Zero<br />
Tolerance<br />
Pathogens<br />
No visible milk,<br />
feces, or<br />
ingesta.<br />
Each carcass,<br />
head, and<br />
variety meat for<br />
visible<br />
contaminants<br />
visual<br />
observation<br />
Each carcass,<br />
head, and<br />
variety meat<br />
Carcass<br />
trimmer or<br />
skinner<br />
Corrective<br />
Action Verification Records<br />
1. Identify and<br />
eliminate<br />
cause of<br />
deviation<br />
2. Trimming<br />
visible<br />
contaminants<br />
will bring CCP<br />
within<br />
allowable limits<br />
3. Prevent<br />
recurrence<br />
4. No harmful<br />
or adulterated<br />
product will<br />
enter<br />
commerce<br />
Direct<br />
observation of<br />
person<br />
conducting<br />
trimming by<br />
plant manager<br />
once a week.<br />
For each<br />
slaughter day,<br />
records will be<br />
reviewed by<br />
plant manager<br />
or designee.<br />
<strong>Slaughter</strong> Log<br />
Corrective<br />
Action Log<br />
<strong>04</strong>/<strong>11</strong>/<strong>2003</strong> <strong>Version</strong> 1
<strong>HACCP</strong> <strong>Plan</strong> <strong>Pork</strong>, <strong>Sheep</strong>, <strong>Goat</strong> <strong>Slaughter</strong><br />
Product Description<br />
COMMON NAME:<br />
HOW IS IT TO BE<br />
USED?<br />
TYPE OF<br />
PACKAGE?<br />
LENGTH OF SHELF<br />
LIFE?<br />
AT WHAT<br />
TEMPERATURE?<br />
pork, sheep, goat, lamb carcass halves or quarters; whole heads; head and cheek meat; (species) variety meats<br />
(heart, liver, tongue)<br />
Further processing<br />
No packaging is used for carcass wholes and halves; butcher paper, freezer wrap, or cryovac bag for head and<br />
cheek meat; butcher paper or cryovac for heart, liver, and tongue<br />
Carcass halves and quarters: 14 days<br />
under refrigeration (< 41°F) Head and cheek meat: 7 days under refrigeration, 6 months frozen. Heart, liver,<br />
tongue: 7 days under refrigeration, 6 months frozen.<br />
WHERE WILL IT BE<br />
SOLD?<br />
LABELING<br />
INSTRUCTIONS:<br />
IS SPECIAL<br />
DISTRIBUTION<br />
CONTROL<br />
NEEDED?<br />
Further processed in our plant<br />
Carcasses have ID tag and inspection legend; carcass quarters have inspection legend.Head and cheek meat are<br />
labeled with appropriate product label including safe handling instructions. Heart, liver, tongue (if not subdivided)<br />
have inspection legend; if subdivided, these products would have an appropriate product label including safe<br />
handling instructions.<br />
Lot code based on production date applied along with appropriate product label.<br />
<strong>04</strong>/<strong>11</strong>/<strong>2003</strong> <strong>Version</strong> 2
<strong>HACCP</strong> <strong>Plan</strong> <strong>Pork</strong>, <strong>Sheep</strong>, <strong>Goat</strong> <strong>Slaughter</strong><br />
<strong>04</strong>/<strong>11</strong>/<strong>2003</strong> <strong>Version</strong> 3
<strong>HACCP</strong> <strong>Plan</strong> <strong>Pork</strong>, <strong>Sheep</strong>, <strong>Goat</strong> <strong>Slaughter</strong><br />
1. Process Step 2. Food Safety Hazard 3. Reasonably likely to<br />
occur<br />
1. Receiving live animals Biological – Pathogens<br />
(Salmonella) carried on<br />
hide and in intestinal<br />
tract<br />
HAZARD ANALYSIS – PORK, SHEEP, GOAT SLAUGHTER –<br />
Carcass halves and quarters, whole head, head meat, heart, liver, tongue<br />
Yes (Pathogens)<br />
4. Basis of Reasonably<br />
likely to occur<br />
Livestock are a known<br />
source of Salmonella<br />
5. If Yes in Column 3,<br />
What Measures Could<br />
be Applied to Prevent,<br />
Eliminate, or Reduce<br />
the Hazard to an<br />
Acceptable Level?<br />
Trim Zero Tolerance step<br />
(CCP later in the<br />
process) controls<br />
pathogens.<br />
6. Critical Control<br />
Point<br />
Chemical – Drug<br />
residues<br />
No<br />
Low risk according to<br />
USDA Residue<br />
Monitoring Program<br />
Physical – Buckshot,<br />
needles, bullets<br />
No<br />
SSOPs make hazard<br />
unlikely.<br />
2. Stunning/Bleeding OR<br />
Shooting/Bleeding<br />
Biological Pathogens<br />
(see list above)<br />
introduced into animal’s<br />
circulatory system by<br />
the sticking knife<br />
Yes<br />
(Pathogens)<br />
Sticking knife will be<br />
heatsanitized prior to<br />
sticking. Operational<br />
SSOP will be followed.<br />
However, pathogen<br />
transfer may still occur.<br />
Trim Zero Tolerance step<br />
(CCP later in the<br />
process) controls<br />
pathogens. Stick wound<br />
is trimmed on every<br />
animal.<br />
Chemical – None<br />
No<br />
3. Head Removal<br />
(optional)<br />
Physical fragments– Bullet<br />
No If broken) frangible bullets (easily are used,<br />
the head will be<br />
discarded.<br />
Biological –Pathogens<br />
(see list above)<br />
introduced by knife<br />
Yes<br />
Hide opening and<br />
removal of head may<br />
introduce pathogens<br />
onto the carcass.<br />
Trim Zero Tolerance step<br />
(CCP later in the<br />
process) controls<br />
pathogens.<br />
<strong>04</strong>/<strong>11</strong>/<strong>2003</strong> <strong>Version</strong> 4
<strong>HACCP</strong> <strong>Plan</strong> <strong>Pork</strong>, <strong>Sheep</strong>, <strong>Goat</strong> <strong>Slaughter</strong><br />
Chemical – None<br />
No<br />
Physical – None<br />
No<br />
4. Head processing<br />
(optional)<br />
Biological: Presence or<br />
growth of pathogens (see<br />
list above)<br />
Yes<br />
(Presence)<br />
No<br />
(Growth)<br />
Raw meat is a known<br />
source of pathogens.<br />
Trim Zero Tolerance step<br />
(CCP later in the<br />
process) controls<br />
pathogens.<br />
Chemical – None<br />
No<br />
Physical – None<br />
No<br />
5. Skinning Biological – Presence or<br />
growth of pathogens (see<br />
list above)<br />
Chemical – None<br />
Yes (Presence)<br />
No<br />
(Growth)<br />
No<br />
The hide is a known<br />
source of pathogens.<br />
Trim Zero Tolerance step<br />
(CCP later in the<br />
process) controls<br />
pathogens.<br />
Physical None<br />
No<br />
6. Scalding / Dehairing Biological –<br />
Contamination by<br />
pathogens when tissue<br />
is exposed during<br />
process<br />
No<br />
Exposed subcutaneous<br />
tissue is trimmed using<br />
a sanitized knife.<br />
Chemical None<br />
Physical None<br />
No<br />
No<br />
7. Evisceration Biological – Presence or<br />
growth of pathogens (see<br />
list above)<br />
Yes<br />
(Presence)<br />
No<br />
(Growth)<br />
The intestinal tract of<br />
animals is a known<br />
source of pathogens.<br />
Trim Zero Tolerance step<br />
(CCP later in the<br />
process) controls<br />
pathogens.<br />
<strong>04</strong>/<strong>11</strong>/<strong>2003</strong> <strong>Version</strong> 5
<strong>HACCP</strong> <strong>Plan</strong> <strong>Pork</strong>, <strong>Sheep</strong>, <strong>Goat</strong> <strong>Slaughter</strong><br />
Chemical – None<br />
No<br />
Physical – None<br />
No<br />
8. Variety Meats<br />
Processing (optional)<br />
Biological Presence or<br />
growth of pathogens (see<br />
list above)<br />
Chemical –None<br />
Yes<br />
(Presence)<br />
No<br />
(Growth)<br />
No<br />
Livestock are a known<br />
source of pathogens.<br />
Heart, liver, tongue may<br />
be contaminated via<br />
tools, employees, hides<br />
and gastrointestinal<br />
tract, although<br />
Operational SSOP<br />
reduces this risk.<br />
Trim Zero Tolerance step<br />
(CCP later in the<br />
process) controls<br />
pathogens.<br />
Physical – None<br />
No<br />
9. Splitting (optional) Biological – Presence or<br />
growth of pathogens (see<br />
list above)<br />
Yes (Presence)<br />
No<br />
(Growth)<br />
Pathogens are known to<br />
be present on livestock<br />
carcasses; splitting saw<br />
may transfer pathogens<br />
from carcass to carcass<br />
or from location to<br />
location on one carcass.<br />
Trim Zero Tolerance step<br />
(CCP later in the<br />
process) controls<br />
pathogens.<br />
Chemical –None<br />
No<br />
Physical – Metal or bone<br />
fragments<br />
No<br />
Visual observation for<br />
foreign materials during<br />
processing, inspection of<br />
equipment during<br />
cleaning make hazard<br />
unlikely.<br />
<strong>04</strong>/<strong>11</strong>/<strong>2003</strong> <strong>Version</strong> 6
<strong>HACCP</strong> <strong>Plan</strong> <strong>Pork</strong>, <strong>Sheep</strong>, <strong>Goat</strong> <strong>Slaughter</strong><br />
10. Trim Zero Tolerance<br />
(may be done<br />
concurrently with step 4<br />
or step 8 for head meat<br />
and variety meats,<br />
respectively)<br />
Biological – Presence or<br />
growth of pathogens (see<br />
list above)<br />
Chemical – None<br />
Yes (Presence)<br />
No<br />
(Growth)<br />
No<br />
Pathogens are known to<br />
be present on livestock<br />
carcasses, and are also<br />
reasonably likely to be<br />
present on head meat<br />
and variety meats.<br />
All visible fecal material,<br />
milk, ingesta is trimmed<br />
off carcass halves and<br />
quarters, whole heads,<br />
head meat, and variety<br />
meats. Stick wound is<br />
trimmed on every animal.<br />
1B<br />
Physical – None<br />
No<br />
<strong>11</strong>. Final Wash Biological – Presence or<br />
growth of pathogens (see<br />
list above)<br />
Chemical – None<br />
No (Presence)<br />
No<br />
(Growth)<br />
No<br />
Prior step (Trim Zero<br />
Tolerance) reduced<br />
likelihood of hazard<br />
occurring to an<br />
acceptable level.<br />
Physical None.<br />
No<br />
12. Chilling Biological – Biological<br />
–Presence or growth of<br />
pathogens (see list<br />
above)<br />
Chemical – None<br />
No (Presence)<br />
No<br />
(Growth)<br />
No<br />
Pathogens adequately<br />
controlled at preceding<br />
CCP, SOP for final<br />
product storage makes<br />
growth of pathogens (if<br />
present) unlikely 2Tompkin .<br />
Physical None<br />
No<br />
<strong>04</strong>/<strong>11</strong>/<strong>2003</strong> <strong>Version</strong> 7