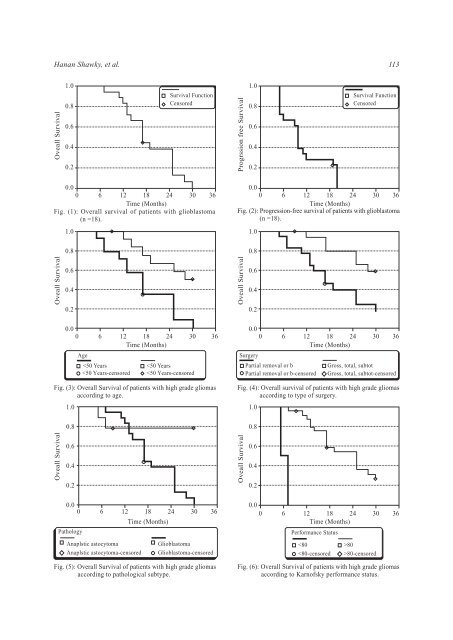
112 <strong>The</strong> <strong>Outcomes</strong> <strong>of</strong> <strong>Concomitant</strong> <strong>Radiation</strong> <strong>Plus</strong> <strong>Temozolomide</strong> week <strong>of</strong> the concomitant treatment. During the adjuvant temozolomide phase, 33.3% <strong>of</strong> patients required antiemetic therapy. One patient experienced a treatment-induced rash that resulted in early discontinuation <strong>of</strong> temozolomide after 7 days <strong>of</strong> CCRT. Moderate to severe fatigue was reported in 5 patients during the CCRT phase (grade 3; one patient) and in 5 patients during the adjuvant temozolomide phase (grade 3; one patient). <strong>The</strong> short duration <strong>of</strong> follow-up precludes definitive assessment <strong>of</strong> late radiation toxicity; only 9 patients were alive with a follow-up longer than 24 months. However, signs <strong>of</strong> leukoencephalopathy, without evident clinical impairment, were apparent on MRI in all <strong>of</strong> these patients. One patient developed intracranial hypertension, refractory seizures, and loss <strong>of</strong> vision 25 months after beginning RT. <strong>The</strong> loss <strong>of</strong> vision may in part be due to prior RT. Subsequent work-up indicated a spinal dissemination <strong>of</strong> the disease with positive CSF cytology and no evidence <strong>of</strong> local recurrence. A second patient developed neurologic deterioration with progressive short-term memory loss and hemiplegia 17 months after beginning RT. At 26 months, this patient was still alive without evidence <strong>of</strong> tumor progression. <strong>The</strong> remaining patients with follow-up longer than 18 months are doing well without any clinical signs <strong>of</strong> neurologic impairment. Thromboembolic events occurred in 3 patients (11.1%). Two patients died <strong>of</strong> cerebral hemorrhage in the absence <strong>of</strong> a coagulation disorder or thrombocytopenia. Survival: At the time <strong>of</strong> this analysis, 19 patients had died. <strong>The</strong> median duration <strong>of</strong> follow-up was 17 months, (range, 5-30 months). On the basis <strong>of</strong> Kaplan-Meier estimates, the median overall survival for the all patients with high grade gliomas (n=27) was 19 months (95% confidence interval, 13.58-24.42) (Table 5). About 33.3% <strong>of</strong> our patients were alive at 2 years, (Table 5). <strong>The</strong> median progression-free survival was 11 months (95% confidence interval, 8.56- 13.44) (Table 5). <strong>The</strong> two-year progression-free survival rate was 26.1%. <strong>The</strong> 18 patients with glioblastoma were analyzed separately from the other patients with high grade gliomas, and the median overall survival was 17 months (95% confidence interval, 13.9-20.1). <strong>The</strong> one-year and two-year overall survival rate were 83.3% and 38.1% respectively (Fig. 1). <strong>The</strong> median progression-free survival for the 18 patients with glioblastoma was 10 months (95% confidence interval, 8.99-11.01) (Fig. 2). <strong>The</strong> six-month progression-free survival rate was 72.22%. Prognostic factors: We analyzed the median overall survival and survival rates <strong>of</strong> the eligible patient populations in relation to prognostic indicators. In patients younger than 50 years old, the median survival was not reached at 24 months, with 66.7% <strong>of</strong> these patients still alive at 24 months. In patients 50 years old, the 24 months overall survival was only 35.7% months (p=0.005), (Fig. 3). <strong>The</strong> prognosis by surgical respectability was also analyzed in these eligible patients. Patients who underwent gross total resection, near-total removal and subtotal resection had 24 months overall survival <strong>of</strong> 57%. However, for patients who underwent partial removal or biopsy, the 24 months overall survival was 26.8% (p=0.0017), (Fig. 4). As regard to pathological type, the 24 months overall survival for patients with glioblastoma and anaplastic astrocytoma (WHO grade III), was 38.1% and 77.8% respectively (p=0.0025), (Fig. 5). Survival according to other possible prognostic factors were included, Karn<strong>of</strong>sky Table (5): Overall Survival and progression-free survival <strong>of</strong> all patients with high grade gliomas (n=27). Variable Overall Survival (months): Median 95% confidence interval 12-month 24-month Progression-free survival (months): Median 95% confidence interval 12-month 24-month Survival 19.00 months 13.58-24.42 months 81.16% 33.3% 11.00 8.56-13.44 months 43.14% 26.14%
Hanan Shawky, et al. 113 Oveall Survival 1.0 0.8 0.6 0.4 0.2 Survival Function Censored Progrssion free Survival 1.0 0.8 0.6 0.4 0.2 Survival Function Censored 0.0 0 6 12 18 24 30 36 Time (Months) Fig. (1): Overall survival <strong>of</strong> patients with glioblastoma (n =18). 1.0 0.8 0.0 0 6 12 18 24 30 36 Time (Months) Fig. (2): Progression-free survival <strong>of</strong> patients with glioblastoma (n =18). 1.0 0.8 Oveall Survival 0.6 0.4 0.2 Oveall Survival 0.6 0.4 0.2 0.0 0 6 12 18 24 30 36 Time (Months) Age