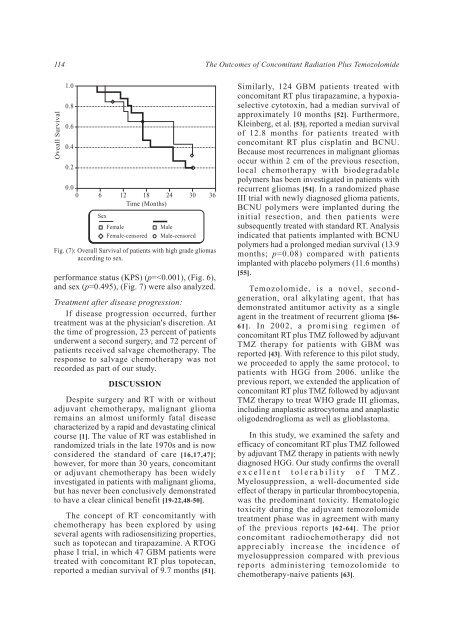
114 <strong>The</strong> <strong>Outcomes</strong> <strong>of</strong> <strong>Concomitant</strong> <strong>Radiation</strong> <strong>Plus</strong> <strong>Temozolomide</strong> Oveall Survival 1.0 0.8 0.6 0.4 0.2 0.0 0 6 12 18 24 30 36 Time (Months) Sex Female Female-censored Fig. (7): Overall Survival <strong>of</strong> patients with high grade gliomas according to sex. performance status (KPS) (p=
Hanan Shawky, et al. 115 In this study, lymphocytopenia is <strong>of</strong>ten observed with TMZ treatment but may, in part, be due to the frequent administration <strong>of</strong> corticosteroids as was described in another phase I trial [36]. Although lymphocytopenia occurs frequently, it is not typically associated with clinical sequelae. However, one <strong>of</strong> the 27 patients we treated with concomitant RT plus TMZ therapy developed pneumonia. <strong>The</strong> frequency <strong>of</strong> opportunistic infections in a similar patient population treated with RT alone is unknown. Nausea and vomiting, the most frequently reported non-hematologic adverse events, were also mild to moderate and could be readily controlled with the administration <strong>of</strong> standard antiemetics. Non-hematologic toxicity observed with temozolomide treatment was in agreement with the report published by Stupp, et al. [43]. Late toxicity resulting from exposure to alkylating agents or combined modality treatment remains a concern. <strong>Concomitant</strong> RT plus TMZ therapy did not increase late toxicities associated with RT during our follow-up period; however, follow-up remains too short to make any conclusions with regard to late toxicities resulting from treatment with TMZ. Some reports <strong>of</strong> concomitant RT plus TMZ followed by adjuvant TMZ therapy for GBM were published. In 2005, the efficacy <strong>of</strong> postoperative TMZ radiochemotherapy in malignant glioma was reported in Germany [65]. According to that report, median PFS time was 7.3 months for primary glioblastoma, treated with concomitant RT plus TMZ. That study also reported that the median overall survival time for patients with glioblastoma was 14.6 months. Another randomized prospective study <strong>of</strong> concomitant RT plus TMZ was reported in 2005 proving that this protocol is more effective than RT alone in patients with newly diagnosed glioblastoma [66]. This randomized study compared concomitant RT plus TMZ with RT alone in patients with newly diagnosed glioblastoma. It reported that the median overall survival time for patients with glioblastoma was 14.6 months with RT plus TMZ and 12.1 months with RT alone. <strong>The</strong> 2-year overall survival rate was 26.5% with RT plus TMZ and 10.4% with RT alone. That study confirmed the effectiveness <strong>of</strong> concomitant RT plus TMZ for glioblastoma patients. Survival results in our study are encouraging. Indeed, the median PFS time <strong>of</strong> 10 months, the median overall survival <strong>of</strong> 17 months and the 2-year overall survival rate <strong>of</strong> 38.1% for the 18 patients with glioblastoma in our series treated with concomitant RT plus TMZ followed by adjuvant TMZ therapy compares favorably with the other previous reported protocols. At present concomitant RT plus TMZ followed by adjuvant TMZ therapy is widely accepted as the current standard care for patients with glioblastoma [23,32,42,44,67-72]. Unlike glioblastoma, there is no definite consensus about the standard regimen for WHO grade III gliomas, such as anaplastic astrocytoma and anaplastic oligodendroglioma. For example, a phase III trial <strong>of</strong> RT plus chemotherapy using procarbazine, lomustine and vincristine (PCV) to treat anaplastic oligodendroglioma was published in 2006 [73]. This study concluded that PCV plus RT did not prolong the survival <strong>of</strong> patients with anaplastic oligodendroglioma and the longer PFS was associated with significant toxicity. <strong>The</strong>refore, it is essential to verify the role <strong>of</strong> TMZ in the treatment WHO grade III glioma. Since the above-mentioned landmark study <strong>of</strong> Stupp, et al. [66] in 2005, there have been many phase II and III clinical studies <strong>of</strong> the treatment <strong>of</strong> HGG with TMZ in adults [67,68,74-77]. <strong>The</strong>se trials have reported good outcomes, and our present results for patients with WHO grade III and IV gliomas are also favorable, with tolerable toxicity. In our study the median PFS time was 11 months, and the median overall survival was 19 months for the 27 patients with HGG treated with concomitant RT plus TMZ followed by adjuvant TMZ therapy. Our results are consistent with most <strong>of</strong> these reports, confirming that concomitant RT plus TMZ followed by adjuvant TMZ therapy <strong>of</strong>fers good clinical outcomes in the treatment <strong>of</strong> HGG. Subanalyses performed to determine the existence <strong>of</strong> prognostic factors in the patient population under evaluation revealed that baseline KPS was an important prognostic factor that correlated meaningfully with median survival (p=