Chemical Industries NewsletterâJanuary 2009 - Chemical Insight ...
Chemical Industries NewsletterâJanuary 2009 - Chemical Insight ...
Chemical Industries NewsletterâJanuary 2009 - Chemical Insight ...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
August 2008<br />
January <strong>2009</strong><br />
A monthly compilation of SRIC<br />
A monthly report abstracts compilation and news of SRIC<br />
report abstracts and news<br />
CEH Marketing Research Report Abstract<br />
CYCLOHEXANOL AND CYCLOHEXANONE<br />
By Michael Malveda with Hiroaki Mori<br />
Cyclohexanol and cyclohexanone are primarily captively consumed, either isolated or as a mixture, in the production of nylon<br />
intermediates (adipic acid and caprolactam); less than 5% is consumed in markets other than nylon. About 80% of production<br />
is based on cyclohexane raw material; the remainder is based on phenol (and 2% on toluene).<br />
Since most cyclohexanol and cyclohexanone is captively consumed on site for the production of adipic acid and caprolactam<br />
(over 95%), only minor amounts enter the world’s trade markets (cyclohexane, a precursor, is more widely traded). Major<br />
world consumption areas are Western Europe, the United States and Other Asia. For the next several years, Other Asia (in<br />
particular, China) is expected to have the highest average annual growth rate for cyclohexanol and cyclohexanone<br />
consumption.<br />
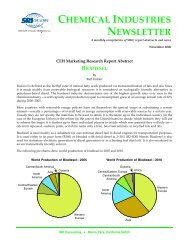
The following pie chart shows world consumption of cyclohexanol and cyclohexanone:<br />
World Consumption of Cyclohexanol and Cyclohexanone—2008<br />
Canada<br />
Central/South America<br />
Central/Eastern<br />
Europe<br />
Japan<br />
Mexico<br />
Western<br />
Europe<br />
Other<br />
Asia<br />
United<br />
States<br />
With the global economic downturn, declines and slowdowns in a variety of industries will limit cyclohexanol and<br />
cyclohexanone growth. For example, in the United States, the housing and construction slumps will negatively affect the<br />
carpet and rug markets, both for residential and commercial businesses. This market accounts for the largest segment of<br />
nylon fiber demand, fibers that are made from adipic acid and caprolactam, and that, in turn, are the drivers of the<br />
cyclohexanol and cyclohexanone products.<br />
(For the complete marketing research report on CYCLOHEXANOL AND CYCLOHEXANONE, visit this report’s home page or see p. 638.7000<br />
A of the <strong>Chemical</strong> Economics Handbook.)<br />
SRI Consulting ● Menlo Park, California 94025
January <strong>2009</strong><br />
<strong>Chemical</strong> <strong>Industries</strong> Newsletter<br />
CEH Product Review Abstract<br />
ETHANOLAMINES<br />
By Elvira O. Camara Greiner with Milen Blagoev and Kazuteru Yokose<br />
Ethanolamines are a class of organic compounds that include monoethanolamine (MEA), diethanolamine (DEA) and<br />
triethanolamine (TEA). They are used in surfactants, gas purification, herbicides and wood preservatives. Commercially,<br />
ethanolamines are created by reacting an appropriate amount of an amine (either ammonia, MEA or DEA) with ethylene oxide.<br />
Over 27% of ethanolamine consumption in 2007 was for the production of surfactants. Herbicides (which may include some<br />
ethanolamines consumed for other agricultural chemicals) accounted for over 14% of total consumption in 2007, followed by<br />
gas treatment applications (10%).<br />
The following pie chart shows world consumption of ethanolamines:<br />
World Consumption of Ethanolamines—2007<br />
Rep. of Korea<br />
Middle East<br />
Canada<br />
Mexico<br />
Central/Eastern Europe<br />
Japan<br />
Central/South<br />
America<br />
India<br />
Taiwan<br />
Other<br />
United<br />
States<br />
China<br />
Western<br />
Europe<br />
In North America, ethanolamines consumption is forecast to grow at a rate of about 3% annually between 2007 and 2013.<br />
Herbicides will drive ethanolamines consumption in North America and the world, followed by surfactants (to a lesser extent).<br />
Gas treatment applications will also grow at a healthy rate of almost 4% per year through 2013. Overall, world ethanolamines<br />
consumption is forecast to grow at an average annual rate of 3.5–4.0% during 2007–2013.<br />
Key findings and future implications for the ethanolamines market include:<br />
• The market tightness experienced during 2004 and 2005 has eased since additional world capacity started coming<br />
on stream in 2006 (in Europe) and 2007 (in the United States).<br />
• The increase in the production of ammoniacal (or alkaline) copper quaternary (ACQ) wood preservatives (a major<br />
MEA consumer) in the United States has leveled off. Nearly all of the growth for ACQ wood preservatives occurred<br />
in late 2003 and 2004.<br />
• Several capacity expansions in various regions are planned in order to meet future demand:<br />
Given current market conditions, all of the planned capacity may not come on stream by the planned on-stream dates. It may<br />
be difficult for market demand to absorb these planned new capacities, especially with the lingering world economic crisis. The<br />
markets with the most growth potential will be herbicides (for DEA), ethyleneamines (for MEA) and ester quats (for TEA).<br />
Prices have risen significantly in the last few years as a result of high energy and feedstock costs.<br />
(For the complete product review on ETHANOLAMINES, visit this report’s home page or see p. 642.5000 A of the <strong>Chemical</strong> Economics<br />
Handbook.)<br />
2 Visit us at www.sriconsulting.com
<strong>Chemical</strong> <strong>Industries</strong> Newsletter January <strong>2009</strong><br />
CEH Marketing Research Report Abstract<br />
INDUSTRIAL PHOSPHATES<br />
By Bala Suresh with Chiyo Funada<br />
The industrial phosphates market has been influenced to a large extent by the dramatic rise in raw material prices, especially<br />
during the past year. The price of purified phosphoric acid has been increasing dramatically as a result of the increase in the<br />
price of phosphate rock, which is the raw material used for producing the acid. That, coupled with the spike in sulfur prices<br />
caused by supply shortages, along with increasing utility costs, has increased the price of phosphate products enormously.<br />
Traditionally, phosphate rock producers have sold the product at relatively lower prices and value was added by the producer<br />
of downstream products. This trend has shifted suddenly wherein the maximum value generation has been moved to the<br />
phosphate rock producers. Phosphate rock prices have escalated fourfold in the past year.<br />
Additionally, China, which is a high-volume exporting country, raised its export tax to between 100% and 175% in April and<br />
September 2008. This was valid for the remainder of 2008. As a result, the gap between the domestic and international prices<br />
increased and exports also increased. This caused some degree of uncertainty in the market as the tax measures could cause<br />
supply shortages and consequently higher prices. However, phosphate rock prices started to stabilize in November 2008 and<br />
this could stabilize the market.<br />
The following pie chart shows consumption of industrial phosphates by major region:<br />
Consumption of Industrial Phosphates by Major Region—2008<br />
Japan<br />
Brazil<br />
Canada<br />
Mexico<br />
Western<br />
Europe<br />
China<br />
United<br />
States<br />
STPP use as a detergent builder no longer accounts for the majority of phosphoric acid consumption in the United States,<br />
Canada or Japan; however, it was the largest market in Western Europe and in Mexico in 2007. Phosphates are excellent<br />
builders, but they are also plant nutrients, and phosphate wastes entering surface water systems can result in excessive<br />
growth of algae. This eutrophication is a significant problem in a number of regions and in particular in the developed<br />
countries. Although detergent phosphates are not normally the major source of phosphate contamination (fertilizer runoff and<br />
animal or human wastes are typically larger sources), they can be readily controlled by restricting use.<br />
Environmental concerns will continue as a strong determinant of phosphate demand in detergents in Western Europe and<br />
North America and accordingly, consumption will stagnate, if not decline. Growth is expected in India, China, other Asian<br />
countries and South America, where increasingly detergents in the mid and premium segments gain market share and these<br />
typically contain phosphate builder systems.<br />
(For the complete marketing research report on INDUSTRIAL PHOSPHATES, visit this report’s home page or see p. 760.2500 A of the<br />
<strong>Chemical</strong> Economics Handbook.)<br />
Visit us at www.sriconsulting.com 3
January <strong>2009</strong><br />
<strong>Chemical</strong> <strong>Industries</strong> Newsletter<br />
CEH Marketing Research Report Abstract<br />
LIGNOSULFONATES<br />
By Sebastian N. Bizzari with Hossein Janshekar and Kazuteru Yokose<br />
Lignosulfonates are complex polymeric materials obtained as coproducts of wood pulping; they consist of a mixture of<br />
sulfonated lignin, sugars, sugar acids, resins and inorganic chemicals. Most lignosulfonates are obtained from the spent<br />
pulping liquor of sulfite pulping operations, although some are also produced by postsulfonation of lignins obtained by sulfate<br />
pulping (kraft process). Recovered coproduct lignosulfonates may be used with little or no additional treatment or they may be<br />
converted to specialty materials with the chemical and physical properties adjusted for specific end-use markets.<br />
Lignosulfonates function primarily as dispersants and binders. Concrete admixtures are the leading market for lignosulfonates,<br />
accounting for 38% of world consumption in 2008. Consumption as binders in copper mining, carbon black and coal is the next<br />
largest world market, accounting for 12% of world consumption in 2008.<br />
The following pie chart shows world consumption of lignosulfonates:<br />
World Consumption of Lignosulfonates—2008<br />
Taiwan<br />
India<br />
Mexico<br />
Canada<br />
Middle East<br />
Central/South America<br />
Rep. of Korea<br />
Japan<br />
Africa<br />
Other<br />
Western<br />
Europe<br />
China<br />
United<br />
States<br />
Central/Eastern<br />
Europe<br />
Dispersant applications of lignosulfonates accounted for 67.5% of world consumption in 2008 followed by binder and adhesive<br />
applications at 32.5%. Major end-use markets include construction, mining, animal feeds and agriculture. Overall economic<br />
performance will continue to be the best indicator of future demand for lignosulfonates. Demand in most downstream markets<br />
is greatly influenced by general economic conditions. As a result, demand largely follows the patterns of the leading world<br />
economies.<br />
Central and Eastern Europe is expected to become the largest market for lignosulfonates in 2013, accounting for 20% of world<br />
consumption, with increased demand in most applications including construction, oil drilling and mining.<br />
Overall economic performance will continue to be the best indicator of future demand for lignosulfonates. The major end-use<br />
markets include construction, mining, animal feeds and agriculture. The market is at risk for further consolidation. Additional<br />
plant closures are expected in the next five years; this should help increase world capacity utilization since demand is growing<br />
at an average annual rate of 1.4%.<br />
(For the complete marketing research report on LIGNOSULFONATES, visit this report’s home page or see p. 671.5000 A of the <strong>Chemical</strong><br />
Economics Handbook.)<br />
4 Visit us at www.sriconsulting.com
<strong>Chemical</strong> <strong>Industries</strong> Newsletter January <strong>2009</strong><br />
CEH Marketing Research Report Abstract<br />
NATURAL RUBBER<br />
By Emanuel Ormonde and Thomas Kälin<br />
Asia has continued to dominate the world supply of natural rubber, averaging more than 90% of total world production. The<br />
largest natural rubber–producing countries include Thailand, Indonesia and Malaysia. Thailand is currently the largest<br />
producer, but Indonesia is closing in on Thai NR production, and is expected to overtake Thai NR production in coming years.<br />
Malaysia, which accounted for 32% of world production in 1988, shifted emphasis to other crops and nonagricultural<br />
investments and produced only 8% of the world total by 1998. Today, Malaysia represents roughly 12% of total world natural<br />
rubber production. Other regions that are expecting high growth in natural rubber production in the forecast period include<br />
Brazil, Guatemala, India, Sri Lanka, the Philippines and Vietnam.<br />
The shift toward radial tires, which use a higher percentage of natural rubber than bias-ply tires, has resulted in an increase in<br />
natural rubber consumption over the past twenty years. Natural rubber accounted for about 30% of the total world<br />
consumption of rubber in 1981; the share had increased to about 42–43% in 2007. Increased rubber consumption in the<br />
natural rubber–producing countries has also been a factor, as well as the greatly increasing demand for natural rubber in<br />
China and India. In the latter part of 2008, there was a slowdown in natural rubber demand as a result of the world economic<br />
downturn.<br />
The following pie chart shows world production of natural rubber:<br />
World Production of Natural Ruber—2008p<br />
Philippines<br />
Liberia<br />
Brazil<br />
Sri Lanka<br />
Côte d’Ivoire<br />
Vietnam<br />
Other Asia<br />
Other Africa<br />
Other Latin America<br />
Thailand<br />
China<br />
India<br />
Malaysia<br />
Indonesia<br />
Two long-standing development programs—one involving epoxidized natural rubber (ENR) and the other involving<br />
thermoplastic natural rubber (TPNR)—offer potentially increased use of natural rubber. ENR is aimed primarily at tire<br />
applications and TPNR at nontire uses, such as weather seals for vehicles, window seals for buildings, shoe sole applications<br />
and certain automotive components. However, developments to date have not flourished. Most industry observers do not<br />
expect either product to have a significant impact on natural rubber consumption.<br />
In contrast, there have been recent developments in the United States and Australia regarding guayule. Guayule rubber,<br />
developed first in the United States and now Australia, is currently at the forefront of commercial viability. In the current<br />
situation, guayule latex would impact substantially on natural rubber for medical devices and consumer markets, and may<br />
eventually impact the industrial markets for tires during 2015–2020.<br />
(For the complete marketing research report on NATURAL RUBBER, visit this report’s home page or see p. 525.2000 A of the <strong>Chemical</strong><br />
Economics Handbook.)<br />
Visit us at www.sriconsulting.com 5
January <strong>2009</strong><br />
<strong>Chemical</strong> <strong>Industries</strong> Newsletter<br />
CEH Industry Overview Abstract<br />
PAINT AND COATINGS OVERVIEW<br />
By Eric Linak with Akihiro Kishi<br />
The paint and coatings industry in the United States, Western Europe and Japan is mature and generally correlates with the<br />
health of the economy, especially housing and construction and transportation. Overall demand from 2007 to 2012 will<br />
increase at average annual rates of 2% in the United States and 1.5% in Western Europe. In Japan, however, consumption of<br />
paints and coatings will experience relatively slow growth during this period (0.3%) as a result of no growth in major markets<br />
such as automotive OEM, machinery and appliances.<br />
In the less industrialized world, coatings are growing at a much faster rate. The best prospects for growth are in Asia Pacific<br />
(10–15% growth per year in the near future), Eastern Europe (6%) and Latin America (6%). Growth of coatings in China is<br />
expected to continue at 8–10% per year. Most of the major multinational paint producers, including PPG, Akzo Nobel, Kansai<br />
Paint, Nippon Paint, BASF, DuPont, Chugoku Marine Paint, and Hempel, have production in China.<br />
The following chart shows consumption of paints and coatings by country and market:<br />
Percent Market Share in Each Region<br />
Consumption of Paints and Coatings by Market—2007<br />
Architectural Coatings<br />
Automotive OEM<br />
Other Product Finishes<br />
Special-Purpose Coatings<br />
Australia Brazil Canada China France Germany India Japan<br />
Korea,<br />
Rep. of Mexico Spain<br />
United<br />
Kingdom<br />
United<br />
States<br />
The major change that has taken place in the coatings industry during the last twenty years has been the adoption of new<br />
coating technologies. Until the early 1970s, most of the coatings were conventional low-solids, solvent-based formulations;<br />
waterborne (latex) paints, used in architectural applications, accounted for 30–35% of the total. In the late 1970s, however,<br />
impending government regulations on air pollution control focusing on industrial coating operations stimulated the<br />
development of low-solvent and solventless coatings that could reduce the emission of volatile organic compounds (VOCs).<br />
Energy conservation and rising solvent costs were also contributing factors. These new coating technologies include<br />
waterborne (thermosetting emulsion, colloidal dispersion, water-soluble) coatings, high-solids coatings, two-component<br />
systems, powder coatings and radiation-curable coatings.<br />
(For the complete industry overview, PAINT AND COATINGS OVERVIEW, visit this report’s home page or see p. 592.5100 A of the <strong>Chemical</strong><br />
Economics Handbook.)<br />
6 Visit us at www.sriconsulting.com
<strong>Chemical</strong> <strong>Industries</strong> Newsletter January <strong>2009</strong><br />
CEH Marketing Research Report Abstract<br />
UNSATURATED POLYESTER RESINS<br />
By Henry Chinn with Uwe Löchner and Yosuke Ishikawa<br />
Unsaturated polyester resins are produced by the polycondensation of saturated and unsaturated dicarboxylic acids with<br />
glycols. Unsaturated polyester resins form highly durable structures and coatings when they are cross-linked with a vinylic<br />
reactive monomer, most commonly styrene. The properties of the cross-linked unsaturated polyester resins depend on the<br />
types of acids and glycols used and their relative proportions.<br />
This report also covers vinyl ester resins, since the industry and government have chosen to group them with unsaturated<br />
polyester when collecting their statistics; however, vinyl esters are actually hybrids between unsaturated polyester and<br />
epoxies. They are linear reaction products of bisphenol A and epichlorohydrin that are terminated with an unsaturated (vinylic)<br />
acid such as methacrylic acid. This product is dissolved in styrene and is applied and cross-linked in the same way as<br />
unsaturated polyesters.<br />
On their own, cross-linked unsaturated polyester and vinyl ester resins have limited structural integrity, so they are often<br />
combined with fiberglass or mineral fillers before cross-linking to enhance their mechanical strength. Resins combined with<br />
fiberglass are referred to as fiberglass-reinforced plastic (FRP). Both lightweight and durable, FRP composites are consumed<br />
primarily by the construction, marine and land transportation industries, although they find use in a variety of other<br />
applications. Nonreinforced cross-linked unsaturated polyester resin is used to make cultured marble and solid surface counter<br />
tops, gel coats, automotive repair putty and filler, and other items such as bowling balls and buttons.<br />
The following pie chart shows world consumption of unsaturated polyester resins:<br />
World Consumption of Unsaturated Polyester Resins—2007<br />
Mexico<br />
Central/South America<br />
Central/Eastern Europe<br />
Japan<br />
Middle East/Africa<br />
Canada<br />
Oceania<br />
China<br />
Other Asia<br />
Western<br />
Europe<br />
United<br />
States<br />
Sales in the primary end markets for unsaturated polyester resin—construction, automotive and marine—depend on the<br />
performance of the general economy and unsaturated polyester resin consumption tends to swing dramatically with any<br />
change in gross domestic product (GDP).<br />
Future demand growth (2007–2012) in North America is forecast to slow somewhat especially in the United States. U.S.<br />
demand had a considerable drop in 2008, with a decline of 15–16% and will continue to decline in <strong>2009</strong>.<br />
Future demand growth (2007–2012) in Europe, the Middle East and Africa is forecast to slow somewhat. The industry will not<br />
be able to decouple from the financial crises, which reached the producing sector in 2008. In fact in 2008, demand was down,<br />
especially in the last quarter of 2008. Highest growth in the region is forecast for Russia, which will continue to grow at a<br />
double-digit rate, albeit from a small base. Future demand growth (2007–2012) in Asia is forecast to slow somewhat.<br />
Visit us at www.sriconsulting.com 7
January <strong>2009</strong><br />
<strong>Chemical</strong> <strong>Industries</strong> Newsletter<br />
The most dynamic market for unsaturated polyester resins is wind energy. On a global basis, new installed wind energy<br />
capacity in 2007 was about 20 gigawatts (GW). For 2012, new installed capacity worldwide is forecast to grow to almost<br />
approximately 53 GW (or 18% per year). Unsaturated polyester resins (including vinyl ester) are used to produce rotor blades,<br />
engine housings and gel coats in competition with epoxy resins. In Western Europe, annual growth of 7–8% is forecast for<br />
unsaturated polyester resin consumption in this segment during 2007–2012. Growth will be higher in Asia.<br />
(For the complete marketing research report on UNSATURATED POLYESTER RESINS, visit this report’s home page or see p. 580.1200 A of<br />
the <strong>Chemical</strong> Economics Handbook.)<br />
PEP Review Abstract<br />
ADVANCED PYROLYSIS GASOLINE UPGRADING<br />
By Abe Gelbein<br />
One of the major sources of BTX aromatics is the pyrolysis gasoline by-product from steam cracking of naphtha for olefins<br />
production. A variety of process schemes is available to recover the aromatics as well as other value-added products from the<br />
feedstock. One such process is the subject of a recent U.S. patent assigned to the SK Corporation. The process is named<br />
Advanced Pygas Upgrading or APU and is exclusively licensed by Axens. It involves the use of a catalytic hydrodealkylation<br />
step that produces a BTX mixture with low ethylbenzene content, LPG and fuel gas from the C 6+ fraction derived from<br />
stabilized pygas. Simple distillation is used to recover the pure aromatics fractions and the LPG.<br />
This review compares the relative economics of the APU process with that of an alternative conventional scheme that<br />
incorporates C 7 /C 8 splitting, extractive distillation to separate the aromatics and nonaromatics from the C 7 fraction and thermal<br />
hydrodealkylation to convert the C 8+ fraction to additional benzene and toluene. Products are benzene, toluene, C 6 - raffinate<br />
and fuel gas. Conceptual process designs, capital estimates and production cost estimates are developed for both processes<br />
for plants processing ~22,000 bbls/day of pygas.<br />
The estimated CAPEX and OPEX for both processes are roughly the same. The relative economics hinge on the value of the<br />
product slates. Based on third-quarter 2008 Gulf Coast prices the APU process has very favorable economics (B/T ROI >25%)<br />
and has a distinct advantage over the conventional route (B/T ROI
<strong>Chemical</strong> <strong>Industries</strong> Newsletter January <strong>2009</strong><br />
Specifically, this report examines the production of the biomonomer lactic acid by fermentation for the production of PLA and<br />
the production of the biomonomer PDO by fermentation and its reaction with terephthalic acid to produce the bioplastic PTT.<br />
For those engaged in the production of biopolymers and their petroleum-based competitors, it is useful for its comparative<br />
economics and understanding of the importance of feedstock costs to the overall economics of biopolymers.<br />
(For the complete December 2008 Report 265 on BIO-BASED POLYMERS, visit this report’s home page .)<br />
PEP Report Abstract<br />
BIOBUTANOL<br />
By Greg Bohlmann and Ron Bray<br />
World production of biofuels has experienced phenomenal growth. Various drivers for this phenomenon include high fuel<br />
prices, concerns about the environment, energy security and rural development. The majority of the growth in biofuels has<br />
been in the production of ethanol. However, there are other biofuels, so-called second-generation biofuels, that may offer<br />
some advantages over ethanol. Second-generation biofuels include cellulosic ethanol, covered in PEP Report 263 Cellulosic<br />
Ethanol, and the subject of this report, biobutanol.<br />
Biobutanol has a number of advantages over ethanol—it has a higher heating value, it is more hydrophobic than ethanol and<br />
can be transported via pipeline integrated in the existing petroleum-based fuels infrastructure and it can be added to gasoline<br />
at higher levels without engine modification.<br />
Biobutanol has garnered the interest not only of early-stage companies such as Tetravitae and Gevo, whose technologies are<br />
covered in this report, but also of major oil and chemical companies. BP and DuPont have formed a JV to develop biobutanol<br />
as a gasoline additive.<br />
This report covers the technological and economic aspects of the production of biobutanol via two processes. The first one is a<br />
modification of the established acetone-butanol-ethanol (ABE) fermentation developed by the University of Illinois and licensed<br />
to Tetravitae. It incorporates an improved microorganism (Clostridium beijerincki BA101) and a gas stripping system for in situ<br />
product removal (ISPR), originally covered in PEP Review 2007-1. The second process is based on a combination of patents<br />
from Gevo and DuPont. It incorporates a novel pathway for the production of isobutanol by a solvent-tolerant microorganism<br />
developed by Gevo and a recovery process based on a DuPont patent application. Neither process is commercial but is in the<br />
process demonstration phase.<br />
We also include for reference a conventional corn dry mill for the production of ethanol because it is likely that if the biobutanol<br />
development program is successful some existing ethanol plants may be converted to biobutanol.<br />
(For the complete December 2008 Report 264 on BIOBUTANOL, visit this report’s home page .)<br />
PEP Report Abstract<br />
CARBON CAPTURE FROM COAL FIRED POWER GENERATION<br />
By Ron Smith with Dipti Dave<br />
Carbon dioxide (CO 2 ), which causes global warming, is emitted industrially from refineries, petrochemical plants, cement, iron<br />
and steel manufacturing plants, and electric power plants. The prospect of global climate change is a matter of genuine public<br />
and private concern. From the viewpoint of the amount of emission, power plant flue gases account for the largest portion.<br />
Thus, the recovery of CO 2 from power plant flue gas is very important for preserving the global environment by way of<br />
prevention of CO 2 emission.<br />
Visit us at www.sriconsulting.com 9
January <strong>2009</strong><br />
<strong>Chemical</strong> <strong>Industries</strong> Newsletter<br />
CO 2 capture and storage or sale offer a new set of options for reducing greenhouse gas emissions that can complement the<br />
current strategies of improving energy efficiency and increasing the use of low-carbon or nonfossil energy resources. In this<br />
report we intend to concentrate on evaluation of technology alternatives for capturing CO 2 from large-scale (200–400<br />
megawatt) gas turbine power plants of the type recently installed in California to deal with the Enron energy trading scheme<br />
fiasco.<br />
This report reviews and evaluates three principal technology alternatives for reducing gas turbine power plant CO 2 emissions<br />
including direct-fired postcombustion gas scrubbing; precombustion decarbonization of natural gas whereby natural gas is<br />
converted to hydrogen and CO 2 by reforming, with hydrogen used as a combustion fuel to drive turbo-generator sets; and the<br />
use of oxyfuel created by separating oxygen from air and burning hydrocarbons with oxygen to produce a turbine exhaust with<br />
high concentration of CO 2 for capture and storage.<br />
(For the complete December 2008 Report 180B on CARBON CAPTURE FROM COAL FIRED POWER GENERATION, visit this report’s<br />
home page .)<br />
PEP Review Abstract<br />
CARBON CAPTURE VIA OXYCOMBUSTION<br />
By Mike Arné<br />
One of the options currently being investigated for carbon capture from coal-fired electric power generation is to use a<br />
predominately oxygen feed to the boiler. This technique, sometimes called oxyfuel or oxycombustion, produces a flue gas that<br />
is mostly composed of carbon dioxide. In effect, it replaces a postcombustion scrubber with a precombustion air separation<br />
unit.<br />
In this review we present our estimate of the capital cost and heat rate/energy efficiency of a pulverized coal-fired power plant<br />
using oxycombustion with 99% oxygen. Net plant output is 550 megawatts. Total fixed capital cost on a December 2008 basis<br />
is $1.8 billion, or roughly $3,300/kW. Gross power output is 790 megawatts; over half of the parasitic load is due to the air<br />
separation unit. The net plant heat rate (HHV) is 11,916 Btu/kWh. Net plant efficiency (HHV) is 28.6%. The power plant<br />
presented is based in large part on information from a U.S. Department of Energy study, Pulverized Coal Oxycombustion<br />
Power Plants, DOE/NETL-2007/1291 published in revised form in October 2007.<br />
(For the complete December 2008 Review 2008-11 on CARBON CAPTURE VIA OXYCOMBUSTION, visit this report’s home page .)<br />
PEP Report Abstract<br />
HYDROCRACKING OF HEAVY OILS AND RESIDUA<br />
By Richard Nielsen<br />
Hydrocracking of heavy oils and residua is increasingly important to refiners because of the increased global production of<br />
heavy and extra-heavy crude oils coupled with increased demand worldwide for low-sulfur middle distillates and residual fuel<br />
oils. Upgrading bitumen into synthetic crude oil (SCO) is of great current and future interest because of the planned and<br />
forecast large expansion of Canadian tar sands production and subsequent bitumen upgrading by hydrocracking into SCO.<br />
Hydrocracking of residual oils mainly increases the production of high-quality middle distillates for blending into jet and diesel<br />
fuels while reducing the volume of low-value, high-sulfur residual fuel oil. Hydrocracking increases the degree of saturation of<br />
the products, which increases product quality, for example, the diesel fuel’s cetane number. Of recent interest is the integration<br />
of hydrocracking with hydrotreating of the hydrocracked products to produce either very-low-sulfur middle distillates or lowsulfur<br />
SCO valued at a premium to many conventional crude oils. Capital and operating costs of the integrated plant are lower<br />
than two separate plants.<br />
10 Visit us at www.sriconsulting.com
<strong>Chemical</strong> <strong>Industries</strong> Newsletter January <strong>2009</strong><br />
This PEP Report provides an overview of the heavy oil hydrocracking process, feed and product supply and demand,<br />
hydrocracking chemistry, catalysts and hardware technology since PEP Report 228, Refinery Residue Upgrading, issued in<br />
2000. We then develop process economics for two bitumen upgrading processes that both integrate hydrocracking with<br />
hydrotreating of the hydrocracked gas oil and lighter products to produce SCO. The first process hydrocracks vacuum residue<br />
in ebullated bed reactors in a single stage without heavy oil recycle. Bitumen-derived naphtha and gas oils from the crude unit<br />
are also charged to the single-stage hydrotreating section of this plant. The second process hydrocracks atmospheric residue<br />
in a slurry reactor with a portion of the residue oil recycled to the hydrocracking reactor. Two-stage hydrotreating is used. Both<br />
processes use conventional fixed-bed hydrotreating reactors.<br />
(For the complete December 2008 Report 211B on HYDROCRACKING OF HEAVY OILS AND RESIDUA, visit this report’s home page .)<br />
PEP Review Abstract<br />
PROCESSING ACIDIC CRUDE OILS<br />
By Richard H. Nielsen<br />
Acidic crude oils are grades of crude oil that contain substantial amounts of naphthenic acids (NAs) or other acids. They are<br />
also called high-TAN crudes after the most common measure of acidity, the total acid number (TAN). Crude oils with as little<br />
as 0.5 mg KOH/g acid or petroleum fractions greater than about 1.0 mg KOH/g oil usually qualify as a high-acid crude or oil. At<br />
the 1.0 mg/g level, crude oils begin to be heavily discounted in value. Other than acidity, there appear to be no distinguishing<br />
properties that characterize these oils, although most high-TAN crudes’ gravities are often less than 29 API and often are low<br />
in sulfur (except for Venezuelan grades) and frequently produce high yields of gas oil. Acidic oils can vary widely in most other<br />
properties.<br />
Refining acidic crude oils is of increasing interest because of their increased production and usually discounted value.<br />
According to one study, incremental high-TAN crude production will rise by 1.8 million B/D from 2005 to 2010. Output will<br />
continue to rise at least through 2015. Acidic crudes are produced in every oil producing region. China will dominate<br />
production, which is forecast to more than double from 2006 to 2015. Other locations historically noted for high-NA crudes<br />
include Venezuela, India, Russia and some fields in California. Newer regions include the North Sea, West Africa, Mexico and<br />
offshore Brazil.<br />
NAs are known to cause severe corrosion problems, especially when NAs are in low-sulfur crude oils. Potential corrosion<br />
occurs at temperatures between about 450° and 750°F (232° to 399°C). NAs and their salts stabilize the oil-water emulsion,<br />
creating problems during desalting and in downstream separators. Decomposition of NAs produces CO 2 that increases the<br />
level of heat-stable salts formation in the dry gas amine absorbers and cause foaming. CO 2 in the aqueous phase increases<br />
the tendency for carbonate stress corrosion cracking.<br />
Acidity in petroleum oils is due to two main sources: organic sulfur and naphthenic acids. This review emphasizes naphthenic<br />
acids since corrosion by sulfur compounds is much more well known and understood.<br />
(For the complete December 2008 Review 2008-14 on PROCESSING ACIDIC CRUDE OILS, visit this report’s home page .)<br />
PEP Review Abstract<br />
PROCESS WATER MANAGEMENT AND TREATMENT<br />
By Charles Butcher<br />
With increasing pressure on water resources worldwide, increasing numbers of facilities are now recycling water within their<br />
own boundaries. Rising costs of raw water and discharge permits, plus more-stringent rules on the quantity and quality of<br />
treated effluent, make water reuse and recycling increasingly attractive.<br />
Visit us at www.sriconsulting.com 11
January <strong>2009</strong><br />
<strong>Chemical</strong> <strong>Industries</strong> Newsletter<br />
At present, the trend is largely limited to recycling selected water streams within the plant boundaries, and to reusing treated<br />
municipal wastewater as a source of process water. “Zero liquid discharge” (ZLD) and reuse of plants’ own treated effluent<br />
remain generally uneconomic, though they will increasingly be required in dry or environmentally sensitive locations.<br />
Key to this convergence between process water and wastewater are the various membrane-based treatment processes,<br />
especially reverse osmosis (RO). Thanks in large part to their ability to desalinate seawater at reasonable cost, large-scale RO<br />
systems are forecast to see sales growth of up to 50% over the next four years. The resulting fall in costs will further<br />
encourage the use of RO in wastewater recycling. Membrane technologies have also significantly improved the economics of<br />
water treatment for demanding uses such as boiler feed, semiconductor manufacturing and pharmaceuticals.<br />
A landmark water reuse project is at Dow <strong>Chemical</strong>’s Terneuzen site in the Netherlands, where 30,000 m 3 /d, representing half<br />
the water used on site, now comes from recycled process water, rainwater and household wastewater. Dow claims to have<br />
reduced its overall consumption of process water by 35% in thirteen years, and the company’s external water business<br />
proposes to cut the cost of water reuse by 35% by 2015.<br />
All water and wastewater treatment processes are sensitive to site conditions, and this is especially true of membranes. Done<br />
correctly, however, water reuse and recycling bring significant economic, environmental and social benefits.<br />
(For the complete December 2008 Review 2008-15 on PROCESS WATER MANAGEMENT AND TREATMENT, visit this report’s home<br />
page .)<br />
PEP Review Abstract<br />
SOIL REMEDIATION<br />
By Jamie Lacson<br />
Soil remediation is a significant part of environmental services. Most remediation projects have been conducted by<br />
environmental service companies, including engineering and environmental consulting firms. The technologies employed for<br />
soil remediation include traditional technologies such as incineration and solidification/stabilization, and emerging/innovative<br />
technologies such as air sparging, soil-vapor extraction, thermal desorption, and bioremediation. From a cost estimation<br />
perspective, soil remediation technologies can be broadly classified into two groups: in situ technologies and ex situ<br />
technologies. These two groups of treatment methods entail major differences in the provision of remedial services and,<br />
consequently, in the methods used to estimate costs. In this PEP Review, we discuss the process and economics of two ex<br />
situ soil remediation technologies: solvent extraction and soil washing.<br />
(For the complete December 2008 Review 2008-4 on SOIL REMEDIATION, visit this report’s home page .)<br />
PEP Review Abstract<br />
TOLUENE DIISOCYANATE FROM TOLUENE DIAMINE BY BAYER GAS PHASE<br />
PHOSGENATION PRELIMINARY EVALUATION<br />
By P.D. Pavlechko<br />
The recent developments announced by Bayer MaterialScience concerning technology to make toluene diisocyanate by gasphase<br />
phosgenation seem to offer new technology to a fairly mature industry. Announcements of scale, performance, energy<br />
savings and actions to build a new facility in China imply that there is something to the new technology. The patent literature is<br />
somewhat sparse on the subject and few details are published, so there are a lot of questions to be answered before an<br />
analysis of the potential design can be completed. Initial attempts wandered down several erroneous paths because of the<br />
lack of hard data and definitive route information. The limited information has raised questions about the scale of the project,<br />
12 Visit us at www.sriconsulting.com
<strong>Chemical</strong> <strong>Industries</strong> Newsletter January <strong>2009</strong><br />
and whether it is a single line or parallel lines. Since comments seem to suggest that the scale is a single process line, this<br />
analysis attempted to build a design based on the maximum scale announced.<br />
The effort managed to compile a plausible design as a single process line to the 300 kmt/yr scale. Limitations in costing<br />
correlation functions limit that design though by forcing parallel equipment and disallowing extrapolation beyond the limit of the<br />
correlations. However, extrapolation of refrigeration costs introduced an anomaly to the analysis too. Therefore, this analysis is<br />
limited to a preliminary assessment that shows promise, but will require more data and analysis to improve the result.<br />
(For the complete December 2008 Review 2008-1 on TOLUENE DIISOCYANATE FROM TOLUENE DIAMINE BY BAYER GAS PHASE<br />
PHOSGENATION PRELIMINARY EVALUATION, visit this report’s home page .)<br />
SCUP REPORTS SCHEDULED FOR <strong>2009</strong><br />
Report Title Author Status<br />
Specialty <strong>Chemical</strong>s Overview Uwe Fink In preparation<br />
Mining <strong>Chemical</strong>s Patricia Thiers In preparation<br />
Synthetic Lubricants Stefan Müller In preparation<br />
Rubber Processing <strong>Chemical</strong>s Fred Hajduk In preparation<br />
Specialty Paper <strong>Chemical</strong>s Ray Will In preparation<br />
Antioxidants Fred Hajduk In preparation<br />
Printing Inks Ray Will In preparation<br />
Corrosion Inhibitors Stefan Müller In preparation<br />
Imaging <strong>Chemical</strong>s: Inkjet Technologies Uwe Fink In preparation<br />
Plastics Additives Stefan Müller In preparation<br />
Specialty Films Fred Hajduk In preparation<br />
Adhesives and Sealants Ray Will In preparation<br />
To view a list of SCUP reports for sale separately, please see our website at<br />
http://www.sriconsulting.com/SCUP/Public/Reports/ . For additional information, please contact:<br />
Ralf Gubler, Acting Director<br />
Specialty <strong>Chemical</strong>s Update Program<br />
SRI Consulting<br />
4300 Bohannon Drive, Suite 200<br />
Menlo Park, CA 94025<br />
Tel. (650) 384-4300 Fax: (650) 330-1149<br />
Visit us at www.sriconsulting.com 13
January <strong>2009</strong><br />
<strong>Chemical</strong> <strong>Industries</strong> Newsletter<br />
CEH REPORTS AND PRODUCT<br />
REVIEWS IN PREPARATION<br />
Report Title<br />
Aromatic Ketone Polymers<br />
Chlorinated Polyethylene Resins<br />
Detergent Alcohols<br />
Diisocyanates<br />
Explosives<br />
Fumigants/Nematicides<br />
Gasoline Octane Improvers<br />
Hydrogen<br />
Liquid Crystal Polymers<br />
Maleic Anhydride<br />
Natural Fatty Acids<br />
Phosphorus <strong>Chemical</strong>s<br />
Plasticizer Alcohols<br />
Plastics Recycling<br />
Polyester Film<br />
Polyether Polyols for Urethanes<br />
Polystyrene<br />
Polysulfide Elastomers<br />
Polyurethane Foams<br />
Polyvinyl Alcohol<br />
Sulfone Polymers<br />
Surfactants, Household Detergents<br />
Zeolites<br />
Author<br />
Thomas Kälin<br />
Emanuel Ormonde<br />
Robert Modler<br />
Henry Chinn<br />
Stefan Schlag<br />
Mike Malveda<br />
Eric Linak<br />
Stefan Schlag<br />
Thomas Kälin<br />
Elvira Greiner<br />
Sebastian Bizzari<br />
Bala Suresh<br />
Sebastian Bizzari<br />
Jim Glauser<br />
Barbara Sesto<br />
Henry Chinn<br />
Koon-Ling Ring<br />
Emanuel Ormonde<br />
Henry Chinn<br />
Henry Chinn<br />
Eric Linak<br />
Bob Modler<br />
Sean Davis<br />
This list is provided for the benefit of <strong>Chemical</strong> Economics<br />
Handbook users who may simultaneously be undertaking their<br />
own studies in these areas. Clients are welcome to write or call<br />
us in order to discuss the work in progress.<br />
CEH REPORTS AVAILABLE SEPARATELY<br />
To obtain a list of CEH marketing research reports or product<br />
reviews for sale separately, please see our website at<br />
http://www.sriconsulting.com/CEH/Public/Reports/ or<br />
contact:<br />
Koon-Ling Ring, Director<br />
<strong>Chemical</strong> Economics Handbook Program<br />
SRI Consulting<br />
4300 Bohannon Drive, Suite 200<br />
Menlo Park, CA 94025<br />
Tel. (650) 384-4300 Fax: (650) 330-1149<br />
14 Visit us at www.sriconsulting.com
<strong>Chemical</strong> <strong>Industries</strong> Newsletter January <strong>2009</strong><br />
Need to know what’s happening with plant capital expenditures and construction activity<br />
Now you can get the latest data easily through <strong>Chemical</strong> Week’s Engineering &<br />
Construction Database. This database, updated weekly, has the most recent data on<br />
engineering and construction projects in the chemical industry, and has worldwide coverage.<br />
Go to www.chemweek.com/ECPD to see for yourself!<br />
This data is garnered from exclusive news items discovered via the editorial expertise at<br />
<strong>Chemical</strong> Week, so it is proprietary and unique among other information on the market.<br />
As a matter of fact, you won’t find it available to just ANYBODY—access is being given to<br />
only a select few people at this time, so once you register, you’ll have to bookmark<br />
www.chemweek.com/ecprojects to get to the data on a regular basis.<br />
When you get access, you can search by a variety of categories, including:<br />
• Producer<br />
• Location<br />
• Product Capacity (thousands of metric tons per year, unless otherwise specified)<br />
• Start-up Date<br />
• Contracting Services Provider<br />
• Technology Provider<br />
Also, you can search by region or look for a specific company.<br />
This database is updated weekly, so you get the latest data to stay on top of the biggest<br />
project investment activity in the industry!<br />
Go to www.chemweek.com/ECPD NOW to register for your free trial.<br />
Then, to make sure you continue to get access, bookmark<br />
www.chemweek.com/ecprojects to get our exclusive data.<br />
<strong>Chemical</strong> Week<br />
A Publication of Access Intelligence, LLC<br />
4 Choke Cherry Road, Suite 200<br />
Rockville, MD 20850<br />
www.chemweek.com<br />
Visit us at www.sriconsulting.com 15
January <strong>2009</strong><br />
<strong>Chemical</strong> <strong>Industries</strong> Newsletter<br />
CHEMICAL INDUSTRIES NEWSLETTER<br />
The <strong>Chemical</strong> <strong>Industries</strong> Newsletter is published monthly by SRI Consulting. The contents of the Newsletter are drawn from current research<br />
and publications of SRIC’s multiclient programs. Readers are welcome to call or write for more information about the subjects and programs<br />
mentioned (see addresses and telephone/fax numbers below).<br />
SRI Consulting offers the world’s most comprehensive ongoing multiclient databases on the chemical industry. The major multiclient programs<br />
include<br />
<strong>Chemical</strong> Economics Handbook<br />
Directory of <strong>Chemical</strong> Producers<br />
The China Report Canada Mexico<br />
Process Economics Program China Middle East<br />
Specialty <strong>Chemical</strong>s Update Program East Asia South/Central America<br />
World Petrochemicals Europe United States<br />
India<br />
Companies may participate in these continuing programs for the chemical industry through annual subscriptions or by purchasing individual<br />
reports. Each program is supported by inquiry and consulting privileges; electronic access is also available for all of these products.<br />
SRI Consulting.......................................................John Pearson, President and CEO<br />
George Intille, Senior Vice President<br />
Ralf Gubler, Vice President<br />
Russell Heinen, Vice President<br />
Linda Henderson, Vice President<br />
<strong>Chemical</strong> Economics Handbook..................................Koon-Ling Ring, Director<br />
Directory of <strong>Chemical</strong> Producers....................................Carolyn Read, Director<br />
Process Economics Program...............................................R.J. Chang, Director<br />
Production/Databases ...................................................Steven F. Read, Director<br />
Specialty <strong>Chemical</strong>s Update Program ...................Ralf Gubler, Acting Director<br />
World Petrochemicals............................................................Ed Gartner, Director<br />
About SRI Consulting<br />
SRI Consulting provides the world’s most comprehensive ongoing databases on the chemical industries. We offer an array of research-based<br />
programs designed to provide clients with specific market intelligence and analysis. These programs, combined with strategic information<br />
services, help clients define new market opportunities, identify and communicate future challenges, formulate and implement business<br />
strategies, and develop innovative products, processes and services. SRIC provides creative yet practical strategies, supported by renowned<br />
industry and technology expertise and delivered by multidisciplinary teams working closely with clients to ensure implementation. SRI<br />
Consulting is a division of Access Intelligence, LLC.<br />
About Access Intelligence, LLC<br />
Access Intelligence, LLC is a full-service global information and marketing solutions provider of competitive business-to-business information.<br />
The company publishes daily news services, premium-value newsletters, subscription-based websites, magazines, directories, and<br />
databases.<br />
SRI Consulting<br />
Headquarters<br />
_________________________ International Offices ____________________________<br />
Menlo Park, CA Europe, Middle East and Africa Beijing<br />
John Pearson, President and CEO Alfred-Escher-Strasse 34 Suite 1606, Tower B, Global Trade Center<br />
4300 Bohannon Drive, Suite 200 CH 8002 Zürich, Switzerland 36 North Third Ring Road East<br />
Menlo Park, CA 94025 Telephone: 41 44 283 63 33 Dongcheng District, Beijing 100013<br />
Telephone: (650) 384-4300 Fax: 41 44 283 63 30 China<br />
Fax: (650) 330-1149 zurich@sriconsulting.com Telephone: 8610-58256826<br />
menlopark@sriconsulting.com Fax: 8610-58256830<br />
beijing@sriconsulting.com<br />
U.S. Offices<br />
East Asia<br />
Houston<br />
Takeda Honcho Building, 8th Floor<br />
2002 Timberloch Place, Suite 110 2-1-7 Nihonbashi Honcho<br />
The Woodlands, TX 77380<br />
Chuoku, Tokyo 103-0023, Japan<br />
Telephone: (281) 203-6280 Telephone: 81 3 5202-7320<br />
Fax: (281) 203-6287 Fax: 81 3 5202-7333<br />
houston@sriconsulting.com<br />
tokyo@sriconsulting.com<br />
<strong>Chemical</strong> <strong>Industries</strong> Newsletter<br />
Ellen Blue, Editor<br />
© <strong>2009</strong> by SRI Consulting.<br />
All rights reserved. Unauthorized reproduction prohibited.<br />
16 Visit us at www.sriconsulting.com
December 2007<br />
<strong>Chemical</strong> <strong>Industries</strong> Newsletter<br />
(For the complete December 2007 report on CATALYSTS: EMISSION CONTROL CATALYSTS, visit this report’s home page or see vol. 5 of<br />
Specialty <strong>Chemical</strong>s—Strategies for Success.)<br />
SCUP Report Abstract<br />
CATALYSTS: PETROLEUM AND CHEMICAL PROCESS<br />
By Masahiro Yoneyama with Uwe Fink, Fred Hajduk, and Wei Yang<br />
This report focuses on petroleum and chemical process catalysts. See the SCUP Catalysts: Emission Control<br />
Catalysts report for information on that area.<br />
Process catalysts, a multibillion-dollar-per-year business worldwide, play a vital role in the economy. The value<br />
of products dependent on process catalysts, including petroleum products, chemicals, pharmaceuticals, synthetic<br />
rubber and plastics, and many others, is said to be in the hundreds of billions of dollars per year. About 90% of<br />
chemical manufacturing processes and more than 20% of all industrial products employ underlying catalytic<br />
steps. Petroleum refining, for example, which is the source of by far the largest share of industrial products,<br />
consists almost entirely of catalytic processes.<br />
For a number of catalysts, the strongest growth in demand through 2011 will occur in regions other than North<br />
America, Western Europe and Japan. Assuming no new economic crises prior to 2011, industrialized and<br />
developing countries in the Asia Pacific region and Latin America will become important markets for process<br />
catalysts. Rising incomes will drive demand for motor vehicles and transportation fuels in Asia and Latin<br />
America. Industrial chemical production, particularly of petrochemicals, is growing faster in Asia and the Middle<br />
East than in North America and Europe. This growth will be reflected in increased demand for a number of<br />
catalysts in the refinery segment (such as for hydroprocessing), for polymerization, and for hydrogen production.<br />
Low-sulfur mandates are also becoming more widespread in these regions.<br />
Legislation is driving growth in catalyst consumption in the developed countries in North America, Europe and<br />
Japan, while economic growth is the major driving force for developing countries of Asia. These regions are<br />
covered in detail in this report. More-stringent vehicle emissions standards are resulting in the development of<br />
advanced automotive catalysts that require low-sulfur fuel, thus driving demand for hydroprocessing catalysts<br />
(and refinery hydrogen). Increased use of hydroprocessing catalysts is also forecast for Western Europe. Overall<br />
catalyst demand growth in Japan will be more modest because of the continued shift of the manufacturing base<br />
overseas to other Asian countries. Catalyst consumption in both petroleum refining and chemical processing will<br />
grow fast reflecting high GDP growth in China.<br />
As the global refining industry moves to cleaner fuels, refiners are being squeezed on hydrogen availability and<br />
octane requirements. Gasoline desulfurization technology has advanced to limit hydrogen consumption and<br />
octane loss, but globally, the octane-barrel position of refiners will deteriorate. On the diesel side of the clean fuels<br />
challenge, a significant increase in hydrogen consumption is forecast to attain ultra-low-sulfur diesel (ULSD)<br />
from straight-run and cracked stocks containing refractory sulfur species. Increasingly, isomerization of light<br />
naphtha will be one of the preferred solutions to add octane to the gasoline pool, triggered by new catalyst<br />
formulations and optimized processes. Catalytic reforming is the technology of choice for the production of highoctane<br />
gasoline and is usually the main source of refinery hydrogen. Catalytic reforming and isomerization<br />
continue to grow because of their role in removing lead from gasoline in the developing world. Hydroprocessing<br />
is probably growing the most, in response to lower sulfur levels in gasoline and diesel.<br />
Major market segments for polymerization catalysts include polyethylene, polypropylene, polyethylene<br />
terephthalate, polyvinyl chloride and polystyrene. Polyolefin catalysts are the largest single market sector.<br />
Polyolefin catalyst consumption is nearly flat. Growth in polyolefin production is compensated mostly by the<br />
development and use of higher-efficiency catalysts.<br />
Technical improvements have reduced the cost of metallocene-produced polymers to levels more competitive<br />
with those produced with conventional Ziegler-Natta polymerization catalysts. Polymers based on single-site<br />
catalysts have unique properties and are expected to create substantial new markets; however, they will not<br />
18 Visit us at www.sriconsulting.com
<strong>Chemical</strong> <strong>Industries</strong> Newsletter December 2007<br />
displace conventional commodity polymers in existing markets. The initial slow growth of SSCs can also be<br />
attributed to intellectual property barriers.<br />
Advanced Ziegler-Natta catalysts have been developed that reportedly can produce polyolefins with properties<br />
similar to those produced by metallocenes, thereby resisting replacement. It is expected that Ziegler-Natta<br />
catalysts will remain the dominating technology because of its cost benefits.<br />
(For the complete December 2007 report on CATALYSTS: PETROLEUM AND CHEMICAL PROCESS, visit this report’s home page or see<br />
vol. 5 of Specialty <strong>Chemical</strong>s—Strategies for Success.)<br />
SCUP Report Abstract<br />
CONSTRUCTION CHEMICALS<br />
By Stefan Müller with Xiamong Ma and Yosuke Ishikawa<br />
In this report construction chemicals are defined as chemical compounds that are added as such or in<br />
formulations to or on construction materials at the construction site in order to improve workability, enhance<br />
performance, add functionality or protect the construction material or the finished structure made out of it. They<br />
undergo chemical reactions (e.g., cross-linking) or physical changes (e.g., solidification from melt) during their<br />
application. The following groups of chemicals will be discussed:<br />
• Concrete admixtures<br />
• Asphalt additives<br />
• Adhesives and sealants<br />
• Protective coatings<br />
Worldwide, the construction industry contributes significantly to the global GDP, and is one of the most<br />
important elements of every economy. Today’s demands on buildings, roads, bridges, tunnels and dams could<br />
not be met without construction chemicals. The strength of concrete has risen dramatically due to the<br />
development of construction chemicals. The diameter of a pillar needed to carry 100 tons was reduced from 100<br />
cm to 10 cm between 1920 and 2004. The cross section of such a pillar is one-hundredth of what was needed in<br />
1920. High-rise buildings must provide maximum space on minimum ground.<br />
The raw materials needed for the production of construction chemicals are manufactured by the large chemical<br />
producers. Polymers are the most important group of raw materials and are found in virtually every construction<br />
chemical formulation ranging from adhesives to waterproofing treatments. The development of new construction<br />
chemicals in many cases requires interaction of the chemical producer, construction chemical manufacturer and<br />
end user.<br />
Protective coatings are the most important group of construction chemicals, followed by adhesives and sealants,<br />
concrete admixtures and asphalt additives.<br />
Construction chemicals will certainly gain importance in the future. While in some regions, the construction of<br />
new buildings will predominate, the focus will shift to renovation in the older economies. This will directly<br />
influence the usage patterns—concrete admixtures are predominately used for new buildings while more<br />
adhesives and sealants are consumed during renovation.<br />
(For the complete December 2007 report on CONSTRUCTION CHEMICALS, visit this report’s home page or see vol. 6 of Specialty<br />
<strong>Chemical</strong>s—Strategies for Success.)<br />
Visit us at www.sriconsulting.com 19
December 2007<br />
<strong>Chemical</strong> <strong>Industries</strong> Newsletter<br />
SCUP Report Abstract<br />
ELECTRONIC CHEMICALS: PART 2<br />
PRINTED CIRCUIT BOARD (PCB) CHEMICALS<br />
AND SEMICONDUCTOR PACKAGING MATERIALS<br />
By Yoshio Inoguchi, Larisa Dorfman, Vivien Yang and Yosuke Ishikawa<br />
Specialty as well as commodity chemicals are used in virtually every step of the manufacture of printed circuit<br />
boards (PCB) and semiconductor packaging materials. This report covers the major chemicals that are consumed<br />
in the production of these PCB and semiconductor packaging.<br />
This study presents an overview of the PCB chemical and semiconductor packaging material markets worldwide<br />
with regional coverage and a focus on regions with rapid technological changes. Coverage includes the three<br />
major regional markets—the United States, Western Europe and Japan—as well as the Republic of Korea, Taiwan,<br />
China and ASEAN countries, where available.<br />
In 2006, the global market for electronic chemicals for the production of printed circuit boards (PCBs) and<br />
semiconductor packaging was valued in the billions of dollars. This diverse, complicated, technology-driven<br />
global market is projected to grow at a robust average annual rate through 2011. Key market trends fuel this<br />
engine and multiple industries come together to deliver electronic products to the marketplace.<br />
Market forces drive the demand for materials, wafers, equipment, IC devices, services, software and components,<br />
as well as packaging and PCBs. Materials are used in various applications of this continuous loop. The major<br />
macroeconomic drivers that influence this industry include:<br />
• Globalization. A product can be designed in one country and manufactured in other.<br />
• The rise of the consumer. Globalization has brought wealth to emerging economies.<br />
• The communication and information age. Businesses and people are spread out all over the globe.<br />
• The cost/performance paradox. Moore’s Law is still in effect.<br />
• The rise of Asia. This global trend cannot be overemphasized.<br />
Currently, the global growth of PCBs is being driven by the increased use of multilayered, flexible PCBs. The<br />
board density and design complexity keep increasing as electronic companies try to add more features to the<br />
product. The electronic designers are trying to design products with clock speeds in excess of 250 MHz. At these<br />
speeds, speed and power dissipation become an issue requiring the use of advanced materials that can maintain<br />
their physical properties under even more stressful conditions.<br />
The chemical markets for PCB fabrication and semiconductor packaging are greatly influenced by the demand for<br />
products in the key markets. Some of the fastest-growing electronic markets are high-definition televisions; small<br />
wireless devices, including mobile phones, PDAs, and GPS units; and integrated devices like the Apple iPhone ® .<br />
These markets are projected to grow at extremely rapid average annual rates through 2011.<br />
(For the complete December 2007 report on ELECTRONIC CHEMICALS: PART 2, PRINTED CIRCUIT BOARD [PCB] CHEMICALS AND<br />
SEMICONDUCTOR PACKAGING MATERIALS, visit this report’s home page or see vol. 7 of Specialty <strong>Chemical</strong>s—Strategies for Success.)<br />
20 Visit us at www.sriconsulting.com
<strong>Chemical</strong> <strong>Industries</strong> Newsletter December 2007<br />
SCUP REPORTS SCHEDULED FOR 2007<br />
Report Title Author Status<br />
Specialty <strong>Chemical</strong>s Industry Overview Uwe Fink Published<br />
Cosmetic <strong>Chemical</strong>s Stefan Müller Published<br />
Textile <strong>Chemical</strong>s Tad Sasano Published<br />
Flavors and Fragrances Laslo Somogyi Published<br />
Water-Soluble Polymers Ray Will Published<br />
Compounding of Engineering Thermoplastics Fred Hajduk Published<br />
Imaging <strong>Chemical</strong>s: Electrophotography Uwe Fink Published<br />
Process Catalysts Masahiro Yoneyama Published<br />
Surfactants Hossein Janshekar Published<br />
Construction <strong>Chemical</strong>s Stefan Müller Published<br />
Electronic <strong>Chemical</strong>s: Printed Circuit Boards Uwe Fink Published<br />
Emission Control Catalysts Masahiro Yoneyama Published<br />
To view a list of SCUP reports for sale separately, please see our website at<br />
http://www.sriconsulting.com/SCUP/Public/Reports/. For additional information, please contact:<br />
R. J. Chang, Assistant Director<br />
Specialty <strong>Chemical</strong>s Update Program<br />
SRI Consulting<br />
4300 Bohannon Drive, Suite 200<br />
Menlo Park, CA 94025<br />
Tel. (650) 384-4300 Fax: (650) 330-1149<br />
Visit us at www.sriconsulting.com 21
December 2007<br />
CEH REPORTS AND PRODUCT<br />
REVIEWS IN PREPARATION<br />
Report Title<br />
Acetonitrile<br />
Acetylene<br />
Acrylonitrile<br />
Aluminum <strong>Chemical</strong>s<br />
Boron<br />
Carbon Black<br />
Carbon Disulfide<br />
Dimethylformamide<br />
Dyes<br />
Elastomers Overview<br />
Ethyl Alcohol<br />
Ethyl Ether<br />
Furfural<br />
Furfuryl Alcohol<br />
Glycerin<br />
Helium<br />
High-Density Polyethylene<br />
Inorganic Pigments<br />
Isoprene<br />
Lithium, Lithium Minerals and<br />
Lithium <strong>Chemical</strong>s<br />
Natural Gas Liquids<br />
Nonene and Tetramer<br />
PET Solid-State Resins<br />
Polybutadiene Elastomers<br />
Polyimides<br />
Polyisoprene<br />
Polyvinyl Acetate<br />
Propylene Glycols<br />
Vinyl Acetate<br />
Author<br />
Barbara Sesto<br />
Sean Davis<br />
Barbara Sesto<br />
Bala Suresh<br />
Stefan Schlag<br />
Jim Glauser<br />
Milen Blagoev<br />
Sebastian Bizzari<br />
Yosuke Ishikawa<br />
RJ Chang<br />
Eric Linak<br />
Vimala Francis<br />
Ralf Gubler<br />
Ralf Gubler<br />
Ralf Gubler<br />
Bala Suresh<br />
Andrea Borruso<br />
Ray Will<br />
Emanuel Ormonde<br />
Jim Glauser<br />
Sean Davis<br />
Bob Modler<br />
Elvira Greiner<br />
Emanuel Ormonde<br />
Uwe Löchner<br />
Emanuel Ormonde<br />
Henry Chinn<br />
Henry Chinn<br />
Henry Chinn<br />
This list is provided for the benefit of <strong>Chemical</strong> Economics<br />
Handbook users who may simultaneously be undertaking their<br />
own studies in these areas. Clients are welcome to write or call<br />
us in order to discuss the work in progress.<br />
CEH REPORTS AVAILABLE SEPARATELY<br />
To obtain a list of CEH marketing research reports or product<br />
reviews for sale separately, please see our website at<br />
http://www.sriconsulting.com/CEH/Public/Reports/ or<br />
contact:<br />
Koon-Ling Ring, Director<br />
<strong>Chemical</strong> Economics Handbook Program<br />
SRI Consulting<br />
4300 Bohannon Drive, Suite 200<br />
Menlo Park, CA 94025<br />
Tel. (650) 384-4300 Fax: (650) 330-1149<br />
<strong>Chemical</strong> <strong>Industries</strong> Newsletter<br />
Sign Up for a Free<br />
Issue of<br />
Chlor-Alkali<br />
Report<br />
From<br />
Harriman Chemsult Limited<br />
As an SRIC client, you are eligible for a free<br />
issue of Harriman Chemsult Ltd’s Chlor-Alkali<br />
Report—the most reliable, comprehensive,<br />
and authoritative resource for current chloralkali<br />
market trends and pricing. Monthly<br />
analysis of supply, operating rates, demand,<br />
inventory, and import/export trends in the<br />
caustic soda markets includes:<br />
• Contract, spot and market pricing<br />
• Coverage of all international markets,<br />
including American, European and<br />
Asia/Pacific regions<br />
• Detailed tables, graphs, charts<br />
• In-depth coverage of caustic soda<br />
(liquid and solid), chlorine, soda ash,<br />
sodium sulfate, caustic potash and<br />
major chlorinated derivatives.<br />
• Plant and project news<br />
Pricing and market insights gathered by<br />
experts in the U.S., Europe, Southeast Asia,<br />
China, and South America. Subscription<br />
includes monthly publication, weekly<br />
indicators report, market alerts, and access to<br />
experts with historical database support.<br />
Receive a free one month trial of Chlor-Alkali<br />
Report services by visiting:<br />
http://www.harriman.co.uk.<br />
Harriman Chemsult Ltd. * 24-25 Scala Street * London<br />
W1T 2HP* U.K.<br />
Tel: +44 (0) 20 7462 1860 * Fax: +44 (0)20 7462 1861<br />
www.harriman.co.uk<br />
22 Visit us at www.sriconsulting.com
<strong>Chemical</strong> <strong>Industries</strong> Newsletter December 2007<br />
CHEMICAL INDUSTRIES NEWSLETTER<br />
The <strong>Chemical</strong> <strong>Industries</strong> Newsletter is published monthly by SRI Consulting. The contents of the Newsletter are drawn from current research<br />
and publications of SRIC’s multiclient programs. Readers are welcome to call or write for more information about the subjects and programs<br />
mentioned (see addresses and telephone/fax numbers below).<br />
SRI Consulting offers the world’s most comprehensive ongoing multiclient databases on the chemical industry. The major multiclient programs<br />
include<br />
<strong>Chemical</strong> Economics Handbook<br />
Directory of <strong>Chemical</strong> Producers<br />
The China Report Canada Mexico<br />
Process Economics Program China Middle East<br />
Specialty <strong>Chemical</strong>s Update Program East Asia South/Central America<br />
World Petrochemicals Europe United States<br />
India<br />
Companies may participate in these continuing programs for the chemical industry through annual subscriptions or by purchasing individual<br />
reports. Each program is supported by inquiry and consulting privileges; electronic access is also available for all of these products.<br />
SRI Consulting.......................................................John Pearson, President and CEO<br />
George Intille, Senior Vice President<br />
Ralf Gubler, Vice President<br />
Russell Heinen, Vice President<br />
Linda Henderson, Vice President<br />
<strong>Chemical</strong> Economics Handbook..................................Koon-Ling Ring, Director<br />
Directory of <strong>Chemical</strong> Producers....................................Carolyn Read, Director<br />
Process Economics Program.....................Greg Bohlmann, Assistant Director<br />
Production/Databases ...................................................Steven F. Read, Director<br />
Specialty <strong>Chemical</strong>s Update Program ................RJ Chang, Assistant Director<br />
World Petrochemicals............................................................Ed Gartner, Director<br />
About SRI Consulting<br />
SRI Consulting provides the world’s most comprehensive ongoing databases on the chemical industries. We offer an array of research-based<br />
programs designed to provide clients with specific market intelligence and analysis. These programs, combined with strategic information<br />
services, help clients define new market opportunities, identify and communicate future challenges, formulate and implement business<br />
strategies, and develop innovative products, processes and services. SRIC provides creative yet practical strategies, supported by renowned<br />
industry and technology expertise and delivered by multidisciplinary teams working closely with clients to ensure implementation. SRI<br />
Consulting is a division of Access Intelligence, LLC.<br />
About Access Intelligence, LLC<br />
Access Intelligence, LLC is a full-service global information and marketing solutions provider of competitive business-to-business information.<br />
The company publishes daily news services, premium-value newsletters, subscription-based websites, magazines, directories, and<br />
databases.<br />
SRI Consulting<br />
Headquarters<br />
_________________________ International Offices ____________________________<br />
Menlo Park, CA Europe, Middle East and Africa Beijing<br />
John Pearson, President and CEO Toedistrasse 23, 1st Floor Suite 1606, Tower B, Global Trade Center<br />
4300 Bohannon Drive, Suite 200 8002 Zürich, Switzerland 36 North Third Ring Road East<br />
Menlo Park, CA 94025 Telephone: 41 44 283 63 33 Dongcheng District, Beijing 100013<br />
Telephone: (650) 384-4300 Fax: 41 44 283 63 30 China<br />
Fax: (650) 330-1149 zürich@sriconsulting.com Telephone: 8610-58256826<br />
menlopark@sriconsulting.com Fax: 8610-58256830<br />
beijing@sriconsulting.com<br />
U.S. Offices<br />
East Asia<br />
Houston<br />
Takeda Honcho Building, 8th Floor<br />
2002 Timberloch Place, Suite 110 2-1-7 Nihonbashi Honcho<br />
The Woodlands, TX 77380<br />
Chuoku, Tokyo 103-0023, Japan<br />
Telephone: (281) 203-6280 Telephone: 81 3 5202-7320<br />
Fax: (281) 203-6287 Fax: 81 3 5202-7333<br />
houston@sriconsulting.com<br />
tokyo@sriconsulting.com<br />
<strong>Chemical</strong> <strong>Industries</strong> Newsletter<br />
Ellen Blue, Editor<br />
© 2007 by SRI Consulting.<br />
All rights reserved. Unauthorized reproduction prohibited.<br />
Visit us at www.sriconsulting.com 23