Direct methanol fuel cells â ready to go commercial?
Direct methanol fuel cells â ready to go commercial?
Direct methanol fuel cells â ready to go commercial?
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
FEATURE<br />
<strong>Direct</strong> <strong>methanol</strong><br />
<strong>fuel</strong> <strong>cells</strong> – <strong>ready</strong><br />
<strong>to</strong> <strong>go</strong> <strong>commercial</strong><br />
By George Apanel (SRI Consulting, Hous<strong>to</strong>n, Texas, USA) and Eric Johnson<br />
(Atlantic Consulting, Zürich, Switzerland)<br />
DMFCs are about <strong>to</strong> come of age as transportable power supplies. A mo<strong>to</strong>r<br />
scooter is al<strong>ready</strong> headed <strong>to</strong>wards serial production; next in line will be cell<br />
phone power packs, portable genera<strong>to</strong>rs and perhaps even au<strong>to</strong>mobiles. This<br />
article reviews the development of direct <strong>methanol</strong> <strong>fuel</strong> <strong>cells</strong>, current players in<br />
the market, key technical challenges and the current cost picture.<br />
and the presumption is that it will stay that<br />
way or even increase.<br />
• For stationary applications, hydrogen or natural<br />
gas appear <strong>to</strong> be the best <strong>fuel</strong>s (if a lowcost<br />
distribution infrastructure is in place),<br />
but liquids are more desirable for mobile<br />
applications. Methanol is attractive: first,<br />
because it is cheap, and secondly, because it<br />
converts directly <strong>to</strong> electricity without the<br />
need for a bulky reformer in the middle.<br />
Reflect for a moment, if you will, on the title of<br />
this article. To some it will seem completely<br />
mundane: ‘Of course DMFCs are <strong>ready</strong> <strong>to</strong> <strong>go</strong><br />
<strong>commercial</strong>!’ To others, especially those who<br />
have seen energy crises and fads come and <strong>go</strong>,<br />
the title might seem more provocative.<br />
Remember, only a few decades a<strong>go</strong> it was<br />
thought that by now we would eat pills for<br />
meals and drive flying cars.<br />
Besides, <strong>fuel</strong> <strong>cells</strong> have been waiting in the<br />
wings for a long time. It was back in 1839<br />
when William Grove, a Welsh judge, constructed<br />
the first known <strong>fuel</strong> cell. His device<br />
electrochemically reacted hydrogen with oxygen.<br />
But it <strong>to</strong>ok more than another century<br />
for the first practical application of <strong>fuel</strong> <strong>cells</strong><br />
<strong>to</strong> occur. This was for US outer-space exploration<br />
in the early 1960s. The need for an<br />
efficient, long-lasting, non-polluting, compact<br />
power source within the cramped confines<br />
of a space vehicle made <strong>fuel</strong> <strong>cells</strong> the<br />
right choice, despite their high initial cost of<br />
materials and development.<br />
All well and <strong>go</strong>od for technological progress,<br />
but this was still a long ways off from <strong>commercial</strong><br />
viability. The US space program, with its<br />
famed $640 <strong>to</strong>ilet seats, is hardly a conventional,<br />
for-profit market.<br />
Nonetheless, our research suggests that<br />
DMFCs’ time is nearly come. They could soon<br />
be used on a competitive, <strong>commercial</strong> basis <strong>to</strong><br />
power cell phones, portable electric genera<strong>to</strong>rs<br />
and vehicles. Indeed, a DMFC-driven mo<strong>to</strong>r<br />
scooter is about <strong>to</strong> see its market debut.<br />
Why should we be any more optimistic than<br />
the Welsh judge There are two main reasons:<br />
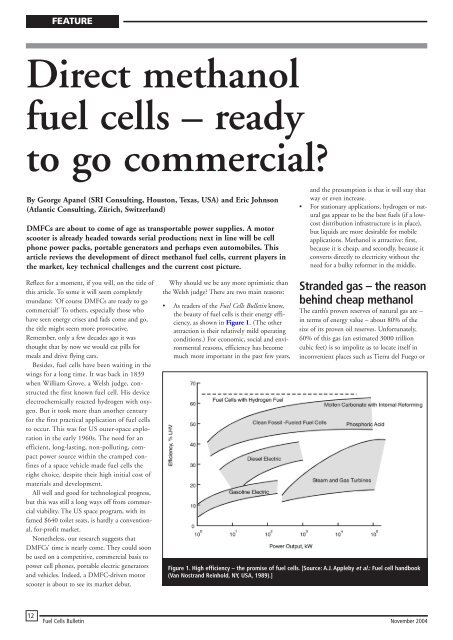
• As readers of the Fuel Cells Bulletin know,<br />
the beauty of <strong>fuel</strong> <strong>cells</strong> is their energy efficiency,<br />
as shown in Figure 1. (The other<br />
attraction is their relatively mild operating<br />
conditions.) For economic, social and environmental<br />
reasons, efficiency has become<br />
much more important in the past few years,<br />
Stranded gas – the reason<br />
behind cheap <strong>methanol</strong><br />
The earth’s proven reserves of natural gas are –<br />
in terms of energy value – about 80% of the<br />
size of its proven oil reserves. Unfortunately,<br />
60% of this gas (an estimated 3000 trillion<br />
cubic feet) is so impolite as <strong>to</strong> locate itself in<br />
inconvenient places such as Tierra del Fue<strong>go</strong> or<br />
Figure 1. High efficiency – the promise of <strong>fuel</strong> <strong>cells</strong>. [Source: A.J. Appleby et al.: Fuel cell handbook<br />
(Van Nostrand Reinhold, NY, USA, 1989).]<br />
12<br />
Fuel Cells Bulletin November 2004
FEATURE<br />
on the North Slope of Alaska. This so-called<br />
‘stranded’ gas can be drilled and brought <strong>to</strong> the<br />
surface, but it is still <strong>to</strong>o remote <strong>to</strong> be transported<br />
economically <strong>to</strong> its potential markets.<br />
Sometimes stranded gas is brought <strong>to</strong> the<br />
surface anyway, because it is locked in underground<br />
formations <strong>to</strong>gether with oil that can be<br />
(and is) extracted economically. This stranded,<br />
‘associated’ gas is either re-injected in<strong>to</strong> the<br />
earth (11 trillion cubic feet in 1998), burnt<br />
near the wellhead (known as ‘flaring’) or even<br />
just vented <strong>to</strong> the atmosphere. If the 3.8 trillion<br />
cubic feet that were flared and vented in 1998<br />
were instead reacted with water at the right<br />
temperature and pressure, the result would be<br />
approximately 74 million <strong>to</strong>nnes of <strong>methanol</strong>.<br />
This is about three times the 25.7 million<br />
<strong>to</strong>nnes actually produced that year, so the<br />
potential here is huge. Moreover, burning or<br />
venting <strong>fuel</strong> is terribly ‘politically incorrect’<br />
these days, not least thanks <strong>to</strong> concerns about<br />
global warming and sustainability. Finally, the<br />
spike in oil prices of the past few years has<br />
shifted the economics of stranded gas – some of<br />
those far-off places are looking closer than they<br />
used <strong>to</strong>.<br />
One option, of course, is <strong>to</strong> compress the gas<br />
in<strong>to</strong> liquefied natural gas (LNG), and then ship<br />
it <strong>to</strong> market. This involves construction of a<br />
costly liquefaction plant, as well as specialized<br />
shipping and terminal facilities, and in some<br />
cases captive power plants bound by long-term<br />
supply contracts. A typical grassroots LNG project<br />
liquefying approximately 980 million standard<br />
cubic feet (SCF) per day of natural gas for<br />
producing about 6 million <strong>to</strong>nnes per year of<br />
LNG could cost US$5–10 billion if the cost of<br />
the power plant is included with the production<br />
facilities.<br />
Another option is a gas-<strong>to</strong>-liquids (GTL)<br />
plant, where the natural gas is converted by a<br />
series of reactions in<strong>to</strong> a <strong>fuel</strong> similar <strong>to</strong> diesel.<br />
Shell is building a major gas-<strong>to</strong>-liquids unit <strong>to</strong><br />
liberate stranded gas in Qatar, and other such<br />
projects are on the drawing board.<br />
Yet another option is <strong>to</strong> use stranded gas <strong>to</strong><br />
make either of two petrochemicals: ammonia or<br />
<strong>methanol</strong>. (Indeed, most recent additions <strong>to</strong><br />
capacity of these products are in remote locations.)<br />
A major attraction of these is that the<br />
capital investment required for such projects is<br />
an order of magnitude lower than for LNG or<br />
gas-<strong>to</strong>-liquids. Furthermore, a world-scale<br />
<strong>methanol</strong> plant can operate on a gas field that is<br />
<strong>to</strong>o small <strong>to</strong> justify an LNG or gas-<strong>to</strong>-liquids<br />
operation. Indeed, such <strong>methanol</strong> plants can<br />
even be mounted on barges, and then moved<br />
from field <strong>to</strong> field. On the other hand, pricing<br />
in chemical markets is no<strong>to</strong>riously volatile.<br />
Great ideas can be – and often are – washed<br />
away in a flood of overcapacity or a shift in<br />
demand.<br />
If there were a stable <strong>fuel</strong> market for<br />
<strong>methanol</strong>, so much the better. (Currently there<br />
are <strong>fuel</strong> markets for it, but they are anything<br />
but stable.) Rest assured, <strong>methanol</strong> producers<br />
are watching <strong>fuel</strong> cell markets anxiously.<br />
Indeed, one of the major producers, Methanex,<br />
is co-sponsoring the development of a DMFCpowered<br />
mo<strong>to</strong>r scooter.<br />
Choose me, choose me –<br />
<strong>methanol</strong>’s mobility<br />
advantage<br />
In mobile applications, liquid <strong>fuel</strong>s are usually<br />
preferable <strong>to</strong> gaseous ones, and often <strong>to</strong> solid<br />
ones as well. So, not surprisingly, researchers<br />
have long been on the lookout for a fluid that<br />
would also be a suitable <strong>fuel</strong>.<br />
Methanol was an obvious candidate early on,<br />
because it:<br />
• can be readily made via a well known manufacturing<br />
process from plentiful raw materials;<br />
• remains liquid under normal s<strong>to</strong>rage conditions<br />
(unlike, say, butane, which tends <strong>to</strong><br />
evaporate much more easily);<br />
• is compatible with the existing <strong>fuel</strong> distribution<br />
infrastructure;<br />
• is relatively hydrogen-dense, i.e. four of the<br />
six a<strong>to</strong>ms in <strong>methanol</strong> (CH 3 OH) are hydrogen;<br />
and<br />
• is environmentally acceptable.<br />
Figure 2. First through a reformer – an indirect <strong>methanol</strong> <strong>fuel</strong> cell.<br />
In the 1990s, development began of an indirect<br />
<strong>methanol</strong> <strong>fuel</strong> cell (Figure 2). A <strong>fuel</strong> processor<br />
(also known as a reformer) was used <strong>to</strong> convert<br />
<strong>methanol</strong> in<strong>to</strong> hydrogen-rich gas for pro<strong>to</strong>nexchange<br />
membrane (PEM) <strong>fuel</strong> <strong>cells</strong>.<br />
Reforming <strong>to</strong> produce hydrogen from<br />
<strong>methanol</strong> is well known on an industrial scale.<br />
At temperatures of 250–300°C, <strong>methanol</strong> is<br />
reacted with steam over a copper-zinc or copper-chromium<br />
catalyst in<strong>to</strong> hydrogen and CO 2<br />
via the following reaction:<br />
CH 3 OH + H 2 O → CO 2 + 3H 2<br />
Methanol enters the <strong>fuel</strong> processor where it is<br />
vaporized, combined with water and converted<br />
in<strong>to</strong> moist, hydrogen-rich reformate. The carbon<br />
monoxide byproduct is removed from the<br />
reformate via a gas purification system.<br />
The purified, hydrogen-rich reformate is fed<br />
<strong>to</strong> the <strong>fuel</strong> cell’s anode chamber, where the<br />
hydrogen is oxidized while oxygen (in the<br />
form of air) is reduced at the cathode. Air is<br />
supplied <strong>to</strong> the cell via a compressor. The<br />
compressed cathode effluent gas passes<br />
through an expander <strong>to</strong> recover some of the<br />
energy from the compression step. The spent<br />
<strong>fuel</strong> from the anode chamber contains some<br />
residual hydrogen, and is fed <strong>to</strong> a catalytic<br />
burner <strong>to</strong> provide heat for <strong>methanol</strong> vaporization<br />
and the endothermic steam <strong>methanol</strong><br />
reformer.<br />
The downsides here are:<br />
• because this reaction consumes energy, the<br />
reformer needs supplemental heat; and<br />
• the reformer adds sizable capital costs, and it<br />
can be bulky.<br />
Nevertheless, DaimlerChrysler has shown that<br />
it can work. In 2001 its Necar 5 pro<strong>to</strong>type –<br />
which used a <strong>methanol</strong> reformer based system<br />
– was driven in a pilot test right across the<br />
United States.<br />
November 2004<br />
Fuel Cells Bulletin<br />
13
FEATURE<br />
Of course, other liquid hydrocarbons can<br />
also be reformed <strong>to</strong> hydrogen. In fact, the same<br />
features that make <strong>methanol</strong> a <strong>go</strong>od <strong>fuel</strong> candidate<br />
also apply generally <strong>to</strong> gasoline, although<br />
the gasoline reformer would be even more complicated<br />
and less efficient. What sets <strong>methanol</strong><br />
apart from those hydrocarbon <strong>fuel</strong>s is its ability<br />
<strong>to</strong> be fed directly in<strong>to</strong> a <strong>fuel</strong> cell, which cuts out<br />
the reformer with its associated costs and<br />
headaches. This is possible because, like all alcohols,<br />
<strong>methanol</strong> contains oxygen: this makes it<br />
more reactive than plain hydrocarbons.<br />
This was first recognized in 1922 by the<br />
German researcher E. Müller, who studied<br />
electro-oxidation of <strong>methanol</strong>. By 1951, Karl<br />
Kordesch and Adolf Marko had begun construction<br />
of a direct <strong>methanol</strong> <strong>fuel</strong> cell based on<br />
Müller’s early concept.<br />
DMFC interest fired up again about a decade<br />
later, this time focused on military applications.<br />
Alkaline electrolytes were tested by researchers at<br />
Allis-Chalmers in 1963. DMFCs based on aqueous<br />
acid electrolytes, which do not react with<br />
CO 2 produced at the anode, were developed by<br />
researchers at Esso and Shell in 1965. Esso<br />
developed a 100 We DMFC for powering communication<br />
equipment. Shell found Pt-Ru <strong>to</strong> be<br />
most effective as an anode electrocatalyst, and<br />
developed a 300 We system pro<strong>to</strong>type in 1968.<br />
Progress then mostly hibernated until 1992,<br />
when DuPont’s Nafion solid polymer electrolyte<br />
membrane was found <strong>to</strong> be far superior<br />
<strong>to</strong> the old medium of sulfuric acid (Figure 3).<br />
In 1994, the Jet Propulsion Labora<strong>to</strong>ry in<br />
California demonstrated a Nafion-based<br />
DMFC, and since then, a new wave of development<br />
has been rolling.<br />
Key players in DMFCs<br />
In our research, we have identified 15 companies<br />
that are leading the development of<br />
DMFCs. They are listed here in alphabetical<br />
order, with a brief discussion of each.<br />
Ball Aerospace & Technologies<br />
20 W electrical output for medical devices, cell<br />
www.ball.com/aerospace<br />
phones, PDAs, lap<strong>to</strong>ps and other wireless<br />
The Colorado-based company is developing a devices. Giner Electrochemical Systems, which<br />
handheld 20 We DMFC power system for the is a joint venture between Giner Inc and<br />
US Defense Advanced Research Projects General Mo<strong>to</strong>rs, has also delivered a complete<br />
Agency (DARPA) Palm Power Program. This liquid-feed direct <strong>methanol</strong> <strong>fuel</strong> cell system providing<br />
a 50 We/12 V output <strong>to</strong> the US Army<br />
compact electrical power source is primarily<br />
intended for foot soldiers and robotics under Research Labora<strong>to</strong>ry.<br />
battlefield conditions, but may find other<br />
emerging, non-military applications. If this<br />
1.5 lb (700 g) device were used <strong>to</strong> power a lap<strong>to</strong>p<br />
computer, it could run continuously for<br />
three days between <strong>methanol</strong> refills. While the<br />
DMFC device is similar in size <strong>to</strong> three videocassette<br />
tapes, 22 lbs (10 kg) of conventional<br />
batteries would otherwise be required for such a<br />
continuous service interval.<br />
<strong>Direct</strong> Methanol Fuel Cell<br />
Lynntech Industries (nowFideris)<br />
Corporation<br />
www.dmfcc.com<br />
This company, founded in California in 2002,<br />
aims <strong>to</strong> <strong>commercial</strong>ize DMFC knowhow developed<br />
at the California Institute of Technology/<br />
NASA Jet Propulsion Labora<strong>to</strong>ry (JPL) and the<br />
University of Southern California. The company<br />
has acquired the rights <strong>to</strong> 26 issued and<br />
more than 40 pending US and foreign patents<br />
on JPL’s DMFC technology.<br />
Figure 3. The power (and voltage) of Nafion. [Source: S. Srinivasan et al.: Techno-economic challenges<br />
for PEMFCs and DMFCs entering energy sec<strong>to</strong>r. First International Conference on Fuel Cell<br />
Science, Engineering & Technology, Rochester, NY, USA, April 2003.]<br />
Giner Electrochemical Systems<br />
www.ginerinc.com<br />
Massachusetts-based GES has announced its<br />
demonstration of a simplified DMFC, based on<br />
a novel method of supplying <strong>methanol</strong> <strong>to</strong> the<br />
<strong>fuel</strong> cell. This is particularly attractive <strong>to</strong> micro<br />
and low-power <strong>fuel</strong> <strong>cells</strong> of up <strong>to</strong> approximately<br />
H Power/Plug Power<br />
www.plugpower.com<br />
US-based H Power (subsequently acquired by<br />
Plug Power) was in a joint development agreement<br />
with DuPont Fluoroproducts <strong>to</strong> develop<br />
DMFCs in the range from 100 <strong>to</strong> 1000 We for<br />
portable and mobile applications.<br />
www.fideris.com<br />
A new generation of test systems for DMFCs<br />
has been launched by this company, which<br />
recently changed its name and relocated <strong>to</strong><br />
Massachusetts. The company has also delivered<br />
four pro<strong>to</strong>type, self-contained DMFC 15 W/<br />
12 V power supplies <strong>to</strong> the US Army Research<br />
Labora<strong>to</strong>ry.<br />
Manhattan Scientifics<br />
www.mhtx.com<br />
NY-based Manhattan Scientifics is <strong>commercial</strong>izing<br />
DMFC technology developed by former<br />
Los Alamos National Labora<strong>to</strong>ry researcher<br />
Bob Hockaday at Energy Related Devices in<br />
Arizona. The company has a proprietary<br />
method for the use and manufacture of<br />
MicroFuel Cell power devices, which are<br />
aimed at portable electronics such as cell<br />
phones, lap<strong>to</strong>p computers, camcorders and<br />
power <strong>to</strong>ols. Manhattan Scientifics has used<br />
MicroFuel Cell technology <strong>to</strong> create its Power<br />
Holster, a portable charger/cell phone carrier.<br />
Fueled by disposable ampoules containing<br />
<strong>methanol</strong>, the <strong>fuel</strong> <strong>cells</strong> can also use cartridges<br />
that provide hydrogen gas generated by a sodium<br />
borohydride/water reaction. Generating<br />
approximately 1 W of electric power, the<br />
MicroFuel Cell array in the unit consists of 10<br />
<strong>cells</strong> in a 4.7 inch long × 1.8 inch wide × 0.02<br />
inch thick (120 × 46 × 0.5 mm) package. The<br />
<strong>cells</strong> are formed on paper-thin plastic sheets<br />
intended for roll-fed mass production. The<br />
14<br />
Fuel Cells Bulletin November 2004
FEATURE<br />
specific energy density of its MicroFuel Cell is<br />
940 Wh/kg, which is between six and nine<br />
times that of <strong>commercial</strong> Li-ion batteries.<br />
Mechanical Technology Inc/<br />
MTI Micro Fuel Cells<br />
Medis Technologies<br />
www.medistechnologies.com<br />
US/Israeli-based Medis has completed the first<br />
phase in the development of its direct liquid Technology<br />
<strong>methanol</strong> (DLM) <strong>fuel</strong> cell for portable electronic<br />
devices. The 1 × 1 × 3/8 inch (25 × 25 × 10 In Korea, Samsung has demonstrated a 40 We<br />
www.sait.samsung.co.kr<br />
mm) DLM <strong>fuel</strong> cell module delivers 0.24 We at DMFC (12 V/3.4 A) that operates a notebook<br />
0.9 V. The company plans <strong>to</strong> package its <strong>fuel</strong> PC for more than 6 h without charging. Core<br />
<strong>cells</strong> as individual modules (‘chips’) for manufacturers<br />
of portable electronic<br />
technologies for miniature DMFC cell packs<br />
devices.<br />
Mo<strong>to</strong>rola Labs<br />
NEC<br />
up the PDA. Thus, it is anticipated that the<br />
www.labs.nec.co.jp/Eng/innovative/E1/<strong>to</strong>p.html<br />
battery will be replaced by an ultracapaci<strong>to</strong>rtype<br />
device before the DMFC is <strong>commercial</strong>-<br />
Japanese electronics giant NEC has announced<br />
a miniature DMFC based on applied nanotechnology.<br />
The power output is claimed <strong>to</strong> be market a DMFC for notebook PCs.<br />
ized. Toshiba has also announced plans <strong>to</strong><br />
about 10 times that of a Li-ion battery with the<br />
Samsung Advanced Institute of<br />
are under development that will power cell<br />
phones and PDAs. The company has also<br />
developed a credit-card-sized DMFC which<br />
www.mo<strong>to</strong>rola-labs.com<br />
would fit in<strong>to</strong> a cell phone, which it hopes <strong>to</strong><br />
US giant Mo<strong>to</strong>rola has demonstrated a pro<strong>to</strong>type<br />
of a miniature integrated, ceramic-based, power density of 50 mW/cm 2 , and a <strong>to</strong>tal<br />
<strong>commercial</strong>ize by 2005. It has a maximum<br />
reformed <strong>methanol</strong>-<strong>to</strong>-hydrogen <strong>fuel</strong> cell electric power output of 2000 mW.<br />
(RHFC). The device incorporates an integrated<br />
<strong>methanol</strong> vaporizer and steam reformer.<br />
The company has also demonstrated a pro<strong>to</strong>type<br />
ceramic-based DMFC which could power<br />
www.smart<strong>fuel</strong>cell.com<br />
German-based Smart Fuel Cell is developing<br />
a cell phone for a month between <strong>methanol</strong><br />
compact DMFC systems for the portable electronics<br />
and small portable genera<strong>to</strong>r markets. A<br />
refills. The <strong>fuel</strong> cell assembly fits in<strong>to</strong> a belt<br />
holster, with <strong>methanol</strong> s<strong>to</strong>red in a small cartridge.<br />
The device is designed <strong>to</strong> either power<br />
portable DMFC power supply unit, <strong>fuel</strong>ed by a<br />
replaceable <strong>methanol</strong> cartridge, will soon be<br />
a cell phone directly, or <strong>to</strong> recharge a conventional<br />
battery pack for powering the phone.<br />
<strong>commercial</strong>ly available on a limited basis <strong>to</strong><br />
industrial cus<strong>to</strong>mers. SFC has demonstrated a<br />
The assembly measures about 2 × 4 × 0.5<br />
inches (50 × 100 × 12 mm). The device integrates<br />
miniature <strong>methanol</strong> concentration sensors,<br />
liquid-gas separation for CO 2 release,<br />
pumps and control electronics. A highly simplified<br />
and miniaturized passive air design www.<strong>to</strong>shiba.co.jp/rdc<br />
Toshiba<br />
eliminates the need for condensers, heatexchangers<br />
and other complex devices. The <strong>to</strong>type DMFC for powering a PDA. It has a<br />
The Japanese company has demonstrated a pro-<br />
company appears <strong>to</strong> be planning <strong>commercial</strong> power density of 30 mW/cm 2 at a constant<br />
<strong>fuel</strong> cell product introductions for cell phones average power output of 5 We. However, an<br />
by 2005–2006.<br />
auxiliary rechargeable battery is required <strong>to</strong> start<br />
Yuasa Corporation<br />
same volume. The company says it plans <strong>commercial</strong><br />
product introductions by 2005 for<br />
www.mtimicro<strong>fuel</strong><strong>cells</strong>.com<br />
MTI Micro in Latham, NY is using technology<br />
www.yuasa.co.jp<br />
notebook computers and cell phones.<br />
licensed from Los Alamos National Labora<strong>to</strong>ry,<br />
This Japanese battery manufacturer has demonstrated<br />
DMFC pro<strong>to</strong>types with electric power<br />
from where some of its key staff (including its<br />
chief technology officer, Dr Shimshon PolyFuel<br />
levels of 100 and 300 W, and says it intends<br />
Gottesfeld) have moved. The company has www.poly<strong>fuel</strong>.com <strong>to</strong> introduce <strong>commercial</strong> products in the near<br />
partnered with DuPont <strong>to</strong> develop Nafionbased<br />
DMFCs, and plans <strong>to</strong> <strong>commercial</strong>ize a from independent research consultancy SRI<br />
The California-based company – a spinoff future.<br />
DMFC for cell phone applications in 2005. International – appears <strong>to</strong> be targeting <strong>commercial</strong><br />
product introductions by 2005. In late<br />
Run DMFC – the key<br />
The planned DMFC product would be about<br />
the same size as a conventional cell phone battery,<br />
but have a considerably longer operational reportedly ruled that a PolyFuel DMFC The two main barriers <strong>to</strong> economic DMFCs<br />
2002 the US Department of Transportation technical challenges<br />
time span between <strong>methanol</strong> cartridge replacements<br />
as well as being more environmentally allowed on board <strong>commercial</strong> aircraft. This ini-<br />
designed <strong>to</strong> power lap<strong>to</strong>p computers could be are <strong>methanol</strong> crossover and slow anode kinetics.<br />
• Methanol crossover is the diffusion of<br />
acceptable.<br />
tiated the process of regula<strong>to</strong>ry acceptance for<br />
<strong>methanol</strong> through the membrane from<br />
DMFCs as an alternative <strong>to</strong> conventional bat-<br />
the anode side <strong>to</strong> the cathode. This<br />
teries in such applications.<br />
reduces DMFC efficiency, primarily<br />
because (1) the crossed-over <strong>methanol</strong> is<br />
essentially wasted, and (2) the cathode’s<br />
catalyst can be poisoned by the carbon<br />
a<strong>to</strong>ms in the <strong>methanol</strong>. This generally<br />
limits <strong>methanol</strong> concentrations at the<br />
anode compartment <strong>to</strong> around 2–5 wt%,<br />
and may require recirculation of water<br />
produced in the cathode compartment<br />
back <strong>to</strong> the anode compartment in order<br />
<strong>to</strong> keep the <strong>methanol</strong> dilute. Such constraints<br />
generally limit the overall DMFC<br />
efficiency <strong>to</strong> around 15–20% and the<br />
power density <strong>to</strong> around 30 mW/cm 2 .<br />
Methanol crossover losses can be 30%<br />
or higher of the <strong>methanol</strong> <strong>fuel</strong> for some<br />
DMFC membranes and design configurations.<br />
• DMFC anode kinetics are slower than<br />
those of PEM <strong>fuel</strong> <strong>cells</strong> running on<br />
hydrogen. This means in turn that either<br />
power densities are lower, or more expensive<br />
platinum catalysts are needed. On<br />
the other hand, DMFCs do not require<br />
cooling plates or supplemental membrane<br />
hydration systems, which could ultimately<br />
make them more compact than hydrogen<br />
PEM <strong>fuel</strong> <strong>cells</strong>.<br />
SFC Smart Fuel Cell GmbH<br />
DMFC that can power a lap<strong>to</strong>p for an entire<br />
The key <strong>to</strong> resolving <strong>methanol</strong> crossover lies<br />
day on a single <strong>methanol</strong> cartridge.<br />
in the membrane, whereas the key <strong>to</strong> faster<br />
kinetics lies in the catalyst and the membrane-electrode<br />
assembly (MEA). The development<br />
of a DMFC membrane that is also<br />
capable of operation at elevated temperatures<br />
would have particularly profound significance,<br />
by also enhancing kinetics while<br />
reducing catalyst requirements. All are the<br />
subjects of intense research.<br />
November 2004<br />
Fuel Cells Bulletin<br />
15
FEATURE<br />
Membranes<br />
Nafion was a great advance in its day, but for<br />
DMFCs there are better materials. Research<br />
has focused on three types: perfluorinated<br />
(which includes Nafion), partially fluorinated<br />
and non-fluorinated.<br />
Perfluorinated membranes currently cost<br />
well upwards of US$800 per m 2 for DMFC<br />
applications, which could be as much as $300<br />
per kW of electric power output. The major<br />
offerings are:<br />
• Micro-reinforced perfluorinated ionomer<br />
composite membranes – such as those<br />
marketed by W.L. Gore under the trade<br />
name Gore-Select. They are microporous<br />
stretched PTFE filled with perfluorinated<br />
ionomer. The thickness of the membrane<br />
can be reduced <strong>to</strong> 20–35 µm. Another<br />
membrane of this type is the Aciplex<br />
range from Asahi Glass in Japan.<br />
• Perfluorinated membranes/inorganic<br />
particle composites – these are produced<br />
by recasting Nafion solution and introducing<br />
inorganic additives (such as<br />
nanoporous, highly hydrophilic silica<br />
particles) or heteropolyacids, such as<br />
phosphotungstic acid or phosphomolybdenic<br />
acid. The principal problem with<br />
such membranes is the hydrosolubility<br />
of the heteropolyacids. Improved performances<br />
have been reported for DMFCs<br />
in some cases because of reduced<br />
<strong>methanol</strong> crossover, with a maximum<br />
power density of 240 mW/cm 2 at 0.4 V.<br />
However, the claims of reduced <strong>methanol</strong><br />
crossover for such membranes appear <strong>to</strong><br />
be controversial.<br />
• Other Nafion-based composites – impregnating<br />
a Nafion membrane with poly-(1-<br />
methyl pyrrole) via in situ polymerization<br />
using UV radiation or H 2 O 2 results in<br />
reduced <strong>methanol</strong> permeation. Unfortunately,<br />
the ionic conductivity was also<br />
reduced. A plasma processing technique<br />
has been devised <strong>to</strong> deposit a thin plasma<br />
polymerized film on Nafion membranes,<br />
<strong>to</strong> improve their barrier properties. The<br />
use of layered membrane structures has<br />
been investigated. Another approach<br />
involves a three-layered laminar electrolyte<br />
system, consisting of a dense, <strong>methanol</strong>impermeable<br />
pro<strong>to</strong>nic conduc<strong>to</strong>r sandwiched<br />
between electronically insulating<br />
layers.<br />
Partially fluorinated membranes under<br />
active investigation include the grafting of fluorinated<br />
base polymer films by various techniques<br />
with select materials. Other partially<br />
fluorinated membranes under investigation<br />
Figure 4. Cell phone with DMFC – external view (left) and exploded internal view (right).<br />
include poly(α, β, β-trifluorostyrene) and<br />
copolymers:<br />
• Grafted ionomer membranes – this involves<br />
bathing base polymer films in gamma radiation<br />
<strong>to</strong> generate free radicals for subsequent<br />
grafting. One developer claims <strong>to</strong><br />
have reduced <strong>methanol</strong> crossover relative <strong>to</strong><br />
Nafion 117 by up <strong>to</strong> 66%, adding that the<br />
membrane can be manufactured at a small<br />
fraction of the cost of Nafion.<br />
• Ionomer membranes based on poly(α, β,<br />
β-trifluorostyrene) and copolymers – these<br />
have been pioneered by Ballard Power<br />
Systems, which has developed several partially<br />
fluorinated pro<strong>to</strong>n-conducting membranes<br />
of sulfonated or phosphonated polymers<br />
plus either poly(phenyl quinoxaline):<br />
BAM 1G (2, 6-diphenyl 1,4-phenylene<br />
oxide), BAM 2G, or a trifluoro styrene<br />
based membrane, BAM 3G.<br />
A variety of novel non-fluorinated ionomer<br />
membranes have been reported in the general<br />
literature. These include ionomers based on<br />
phosphazene, arylene main chains, and<br />
poly(benzimidazole) or PBI. One developer is<br />
making impressive claims not only about reducing<br />
<strong>methanol</strong> crossover, but also operating at<br />
higher temperatures and <strong>methanol</strong> concentrations<br />
– these are critical hurdles for DMFCs, if<br />
they are <strong>to</strong> be economic in au<strong>to</strong>mobiles.<br />
Catalysts and MEAs<br />
Catalyst preparation and MEA manufacturing<br />
techniques are major areas of research.<br />
There are two general catalyst preparation<br />
approaches:<br />
• Impregnation – this is typically a deposition<br />
step of the Pt and Ru precursors (for<br />
example H 2 PtCl 6 , RhCl 3 , Pt(NH 3 ) 2 (OH) 2 ,<br />
Ru 3 CO 2 , Pt(NH 3 ) 2 (NO) 2 etc.) followed<br />
by a reduction step. Reduction can be via<br />
the chemical reduction of the electrocatalyst<br />
slurry in aqueous solution using N 2 H 4 ,<br />
NaS 2 O 5 , NaS 2 O 3 or NaBH 4 , or via the gas<br />
phase reduction of the impregnated carbon<br />
black using a hydrogen gas stream. A drawback<br />
of this procedure is that high dispersions<br />
may not be achievable in the presence<br />
of high metal loadings.<br />
• Colloidal – the advantage here is higher<br />
surface areas with high metal loading. The<br />
main disadvantages are the complexity of<br />
the preparation, the need for organic compounds/solvents,<br />
and generally higher production<br />
costs.<br />
MEA manufacturing research includes experimentation<br />
in:<br />
• Incorporation of PTFE/carbon composite<br />
ducts.<br />
• Modification of the hydrophobichydrophilic<br />
properties of the ionomer in<br />
the electrocatalyst layer.<br />
• Use of pore formers <strong>to</strong> increase the porosity<br />
of the cathode active layer.<br />
• Sputter deposition fabrication.<br />
• Bipolar flow-field plates.<br />
DMFC costs –<br />
competitive in small<br />
mobile applications<br />
Based on a detailed review of patents and<br />
technical papers, we have designed a DMFC<br />
16<br />
Fuel Cells Bulletin November 2004
RESEARCH TRENDS<br />
Conventional Power Cost of DMFC power<br />
Device power source output Base case Optimistic case<br />
Cell phone Rechargeable 2 We 43% lower 82% lower<br />
batteries<br />
Small portable Internal combustion Under 22% lower 67% lower<br />
genera<strong>to</strong>r engine 500 We<br />
Au<strong>to</strong>mobile Internal combustion 75 kWe 188% higher 17% higher<br />
engine<br />
Source: Process Economics Program Report 243A, <strong>Direct</strong> Methanol Fuel Cells, <strong>to</strong> be published in Oc<strong>to</strong>ber 2004<br />
by SRI Consulting.<br />
Table 1. Cost of DMFC power output compared <strong>to</strong> conventional power sources (on a $/kWe basis).<br />
engineering concept for three mobile applications:<br />
cell phones (Figure 4), small portable<br />
genera<strong>to</strong>rs, and au<strong>to</strong>mobiles. Some of these<br />
applications may well be competitive with<br />
conventional power systems (Table 1).<br />
DMFCs for cell phones and small portable<br />
genera<strong>to</strong>rs are now below the cost of conventional<br />
power technologies. For au<strong>to</strong>mobiles,<br />
DMFCs are not yet cost-competitive, but<br />
on<strong>go</strong>ing progress is dramatic, and the technology<br />
is readily scalable. If environmental concerns<br />
keep increasing the costs of conventional<br />
engines and <strong>fuel</strong>s, then it is only a matter of<br />
time until DMFC au<strong>to</strong>mobiles become <strong>commercial</strong>ly<br />
viable.<br />
Convinced At least one company is. Parker-<br />
Hannifin has begun selling its Vectrix DMFC<br />
mo<strong>to</strong>r scooter on a pilot basis. The aim is <strong>to</strong> <strong>go</strong><br />
fully <strong>commercial</strong> in 2006.<br />
For more information, contact: George Apanel,<br />
SRI Consulting, 16945 Northchase Drive, Suite 1910,<br />
Hous<strong>to</strong>n, TX 77060, USA. Tel: +1 281 876 6929,<br />
Email:gapanel@sriconsulting.com,<br />
Web:www.sriconsulting.com<br />
Or contact: Eric Johnson, Atlantic Consulting,<br />
Obstgartenstrasse 14, CH-8136 Gattikon, Switzerland.<br />
Tel:+41 1 772 1079,Email:ejohnson@ecosite.co.uk<br />
Research Trends<br />
Performance of highly dispersed<br />
Pt/C catalysts for PEM and DMFCs<br />
P.K. Shen and Z. Tian: Electrochimica Acta<br />
49(19) 3107–3111 (15 August 2004).<br />
Zirconia-bridged hydrocarbon/<br />
phosphotungstic acid hybrid materials<br />
for high-temperature PEMFCs<br />
J.-D. Kim and I. Honma: Electrochimica Acta<br />
49(19) 3179–3183 (15 August 2004).<br />
Preparation and characterization<br />
of solution-cast Nafion ionomer<br />
membranes<br />
R.F. Silva, M. De Francesco and A. Pozio: Electrochimica<br />
Acta 49(19) 3211–3219 (15 Aug. 2004).<br />
Palladinized Nafion for DMFCs<br />
Y.J. Kim et al.: Electrochimica Acta 49(19) 3227–<br />
3234 (15 August 2004).<br />
3D carbon nanotube network based<br />
on hierarchical structure grown on<br />
carbon paper, for PEMFC electrodes<br />
X. Sun et al.: Chemical Physics Letters 394(4–6)<br />
266–270 (21 August 2004).<br />
Pro<strong>to</strong>n-conducting PDMS/metal<br />
oxide hybrid membranes with<br />
phosphotungstic acid, for hightemperature<br />
PEMFCs<br />
J.-D. Kim and I. Honma: Electrochimica Acta<br />
49(20) 3429–3433 (30 August 2004).<br />
XRD/XPS analysis of as-prepared<br />
and conditioned DMFC array MEAs<br />
R.R. Díaz-Morales et al.: J. Electrochem. Soc.<br />
151(9) A1314–1318 (September 2004).<br />
SOFC anodes based on Cu-Ni and<br />
Cu-Co bimetallics in CeO 2<br />
-YSZ<br />
S.-I. Lee, J.M. Vohs and R.J. Gorte: J. Electrochem.<br />
Soc. 151(9) A1319–1323 (Sep. 2004).<br />
Temperature-dependent kinetics of<br />
DMFC anode<br />
J. Nordlund and G. Lindbergh: J. Electrochem.<br />
Soc. 151(9) A1357–1362 (September 2004).<br />
High-temperature, pro<strong>to</strong>n-conducting<br />
polytetramethylene oxide/zirconia<br />
hybrid membranes<br />
J.-D. Kim and I. Honma: J. Electrochem. Soc.<br />
151(9) A1396–1401 (September 2004).<br />
LaNi(Fe)O 3<br />
as SOFC cathode<br />
H. Orui et al.: J. Electrochem. Soc. 151(9)<br />
A1412–1417 (September 2004).<br />
Effect of segregation <strong>to</strong> interface<br />
on H 2<br />
–H 2<br />
O–Ni–YSZ electrode<br />
performance<br />
K. Vels Hansen, K. Norrman and M. Mogensen:<br />
J. Electrochem. Soc. 151(9) A1436–1444 (September<br />
2004).<br />
Model and procedure <strong>to</strong> optimize<br />
PEMFC internal structure and external<br />
shape, for maximum net power<br />
J.V.C. Vargas, J.C. Ordonez and A. Bejan: Int. J.<br />
Heat & Mass Transfer 47(19/20) 4177–4193<br />
(September 2004).<br />
Preparation of Ni/YSZ anode for IT-<br />
SOFCs by coating precipitation<br />
F.H. Wang et al.: Materials Letters 58(24)<br />
3079–3083 (September 2004).<br />
Ionic, electronic transport in stabilized<br />
β-La 2<br />
Mo 2<br />
O 9<br />
SOFC electrolytes<br />
I.P. Marozau et al.: Electrochimica Acta 49(21)<br />
3517–3524 (1 September 2004).<br />
Subsolidus phase equilibria in Al 2<br />
O 3<br />
–<br />
CeO 2<br />
–PbO and Al 2<br />
O 3<br />
–CeO 2<br />
–RuO 2<br />
systems<br />
M. Hrovat et al.: Materials Research Bulletin<br />
39(11) 1723–1728 (1 September 2004).<br />
Hydrogen generation by hydrolysis<br />
reaction of lithium borohydride<br />
Y. Kojima et al.: Int. J. Hydrogen Energy 29(12)<br />
1213–1217 (September 2004).<br />
H 2<br />
s<strong>to</strong>rage in wind turbine <strong>to</strong>wers<br />
R. Kottenstette and J. Cotrell: Int. J. Hydrogen<br />
Energy 29(12) 1277–1288 (September 2004).<br />
High-temperature membranes in<br />
power generation with CO 2<br />
capture<br />
R. Bredesen, K. Jordal and O. Bolland: Chem.<br />
Eng. & Processing 43(9) 1129–1158 (Sep. 2004).<br />
November 2004<br />
Fuel Cells Bulletin<br />
17