Ion-Selective Electrodes With Ionophore-Doped Sensing Membranes
Ion-Selective Electrodes With Ionophore-Doped Sensing Membranes
Ion-Selective Electrodes With Ionophore-Doped Sensing Membranes
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
<strong>Ion</strong>ophore-doped sensing membranes 2549<br />
EMF (mV)<br />
59.2 mV<br />
(a)<br />
−12<br />
−11.5 −11 −10.5 −10 −9.5 −9 −8.5<br />
log a Ag<br />
+<br />
Sample phase<br />
Membrane phase<br />
Inner filling phase<br />
I +<br />
LI + R − I +<br />
Nernstian<br />
layer, d<br />
I + layer, d<br />
I + I +<br />
Nernstian<br />
Thickness<br />
Concentration of I +<br />
(b)<br />
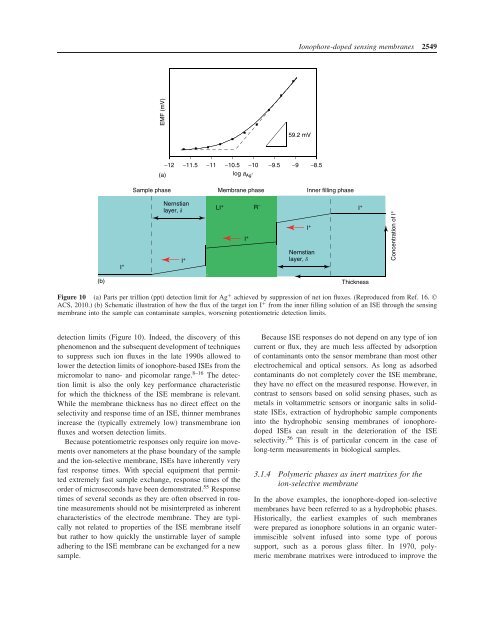
Figure 10 (a) Parts per trillion (ppt) detection limit for Ag + achieved by suppression of net ion fluxes. (Reproduced from Ref. 16. ©<br />
ACS, 2010.) (b) Schematic illustration of how the flux of the target ion I + from the inner filling solution of an ISE through the sensing<br />
membrane into the sample can contaminate samples, worsening potentiometric detection limits.<br />
detection limits (Figure 10). Indeed, the discovery of this<br />
phenomenon and the subsequent development of techniques<br />
to suppress such ion fluxes in the late 1990s allowed to<br />
lower the detection limits of ionophore-based ISEs from the<br />
micromolar to nano- and picomolar range. 8–16 The detection<br />
limit is also the only key performance characteristic<br />
for which the thickness of the ISE membrane is relevant.<br />
While the membrane thickness has no direct effect on the<br />
selectivity and response time of an ISE, thinner membranes<br />
increase the (typically extremely low) transmembrane ion<br />
fluxes and worsen detection limits.<br />
Because potentiometric responses only require ion movements<br />
over nanometers at the phase boundary of the sample<br />
and the ion-selective membrane, ISEs have inherently very<br />
fast response times. <strong>With</strong> special equipment that permitted<br />
extremely fast sample exchange, response times of the<br />
order of microseconds have been demonstrated. 55 Response<br />
times of several seconds as they are often observed in routine<br />
measurements should not be misinterpreted as inherent<br />
characteristics of the electrode membrane. They are typically<br />
not related to properties of the ISE membrane itself<br />
but rather to how quickly the unstirrable layer of sample<br />
adhering to the ISE membrane can be exchanged for a new<br />
sample.<br />
Because ISE responses do not depend on any type of ion<br />
current or flux, they are much less affected by adsorption<br />
of contaminants onto the sensor membrane than most other<br />
electrochemical and optical sensors. As long as adsorbed<br />
contaminants do not completely cover the ISE membrane,<br />
they have no effect on the measured response. However, in<br />
contrast to sensors based on solid sensing phases, such as<br />
metals in voltammetric sensors or inorganic salts in solidstate<br />
ISEs, extraction of hydrophobic sample components<br />
into the hydrophobic sensing membranes of ionophoredoped<br />
ISEs can result in the deterioration of the ISE<br />
selectivity. 56 This is of particular concern in the case of<br />
long-term measurements in biological samples.<br />
3.1.4 Polymeric phases as inert matrixes for the<br />
ion-selective membrane<br />
In the above examples, the ionophore-doped ion-selective<br />
membranes have been referred to as a hydrophobic phases.<br />
Historically, the earliest examples of such membranes<br />
were prepared as ionophore solutions in an organic waterimmiscible<br />
solvent infused into some type of porous<br />
support, such as a porous glass filter. In 1970, polymeric<br />
membrane matrixes were introduced to improve the