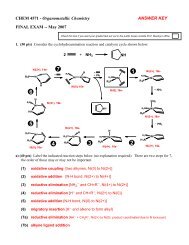
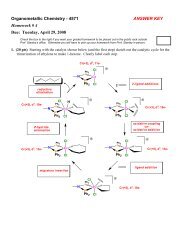
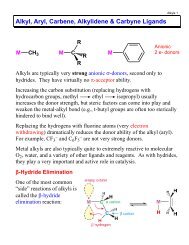
Acids & Bases - Chemistry
Acids & Bases - Chemistry
Acids & Bases - Chemistry
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
<strong>Acids</strong> & <strong>Bases</strong> 20It is very important to remember thatthere are almost always two steps inconverting from [OH − ] to pH:1) convert [OH − ] to [H+]:K[ H+ w] =[ OH − =]2) then convert [H + ] to pH.---- or ----1) convert [OH − ] to pOH2) then convert pOH to pH:−1×10[ OH− ]pH = 14 − pOH14