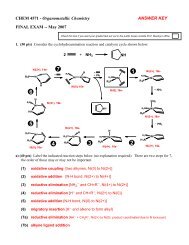
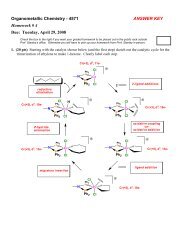
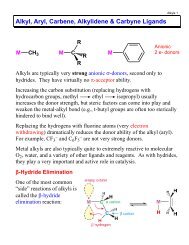
Acids & Bases - Chemistry
Acids & Bases - Chemistry
Acids & Bases - Chemistry
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
Ka=[ NH3][H+[ NH ]4+]<strong>Acids</strong> & <strong>Bases</strong> 44But note that the reciprocal of this expression isalready in the Kb relationship:Ka=[ NH3][H+[ NH ]4+] NH+4Kb=[ ] K[ NH ] [ HTherefore, K a and K b are related via K w .Here are the very important K a / K b relationshipsyou need to know/understand:KwK b = -or- Ka=Ka3w+]KKwbK = K × Kwab